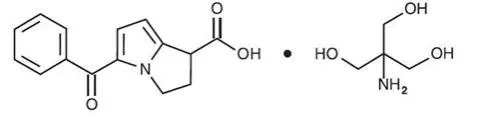
Drug Detail:Acuvail (Ketorolac ophthalmic [ kee-toe-role-ak ])
Drug Class: Ophthalmic anti-inflammatory agents
Highlights of Prescribing Information
These highlights do not include all the information needed to use ACUVAIL® safely and effectively. See full prescribing information for ACUVAIL®.
ACUVAIL® (ketorolac tromethamine ophthalmic solution) 0.45%
Initial U.S. Approval: 1991
Indications and Usage for Acuvail
ACUVAIL ophthalmic solution is a nonsteroidal, anti-inflammatory indicated for the treatment of pain and inflammation following cataract surgery. (1)
Acuvail Dosage and Administration
One drop of ACUVAIL should be applied by the patient to the affected eye twice daily beginning 1 day prior to cataract surgery, and continued through the first 2 weeks of the postoperative period. (2.1)
Dosage Forms and Strengths
ACUVAIL ophthalmic solution containing 4.5 mg/mL ketorolac tromethamine in a single-use vial. (3)
Contraindications
Hypersensitivity to any component of this product. (4)
Warnings and Precautions
- Delayed healing (5.1)
- Cross-sensitivity or hypersensitivity (5.2)
- Increased bleeding time due to interference with thrombocyte aggregation (5.3)
- Corneal effects including keratitis (5.4)
Adverse Reactions/Side Effects
Most common adverse reactions occurring in 1-6% of patients were increased intraocular pressure, conjunctival hemorrhage, and vision blurred. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Allergan at 1-800-678-1605 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 2/2019
Related/similar drugs
diclofenac ophthalmic, dexamethasone ophthalmic, ketorolac ophthalmic, prednisolone ophthalmic, Lotemax, DurezolFull Prescribing Information
1. Indications and Usage for Acuvail
ACUVAIL® ophthalmic solution is indicated for the treatment of pain and inflammation following cataract surgery.
2. Acuvail Dosage and Administration
3. Dosage Forms and Strengths
4.5 mg/mL ketorolac tromethamine solution (0.45%) in a single-use vial.
4. Contraindications
ACUVAIL solution is contraindicated in patients with previously demonstrated hypersensitivity to any of the ingredients in the formulation.
5. Warnings and Precautions
5.1 Delayed Healing
Topical nonsteroidal anti-inflammatory drugs (NSAIDs) may slow or delay healing. Topical corticosteroids are also known to slow or delay healing. Concomitant use of topical NSAIDs and topical steroids may increase the potential for healing problems.
5.2 Cross-Sensitivity or Hypersensitivity
There is the potential for cross-sensitivity to acetylsalicylic acid, phenylacetic acid derivatives, and other NSAIDs. There have been reports of bronchospasm or exacerbation of asthma associated with the use of ketorolac tromethamine ophthalmic solution in patients who either have a known hypersensitivity to aspirin/non-steroidal anti-inflammatory drugs, or a past medical history of asthma. Therefore, caution should be used when treating individuals who have previously exhibited sensitivities to these drugs.
5.3 Increased Bleeding Time
With some NSAIDs, there exists the potential for increased bleeding time due to interference with thrombocyte aggregation. There have been reports that ocularly applied nonsteroidal anti-inflammatory drugs may cause increased bleeding of ocular tissues (including hyphemas) in conjunction with ocular surgery.
It is recommended that ACUVAIL ophthalmic solution be used with caution in patients with known bleeding tendencies or who are receiving other medications, which may prolong bleeding time.
5.4 Corneal Effects
Use of topical NSAIDs may result in keratitis. In some susceptible patients, continued use of topical NSAIDs may result in epithelial breakdown, corneal thinning, corneal erosion, corneal ulceration, or corneal perforation. These events may be sight threatening. Patients with evidence of corneal epithelial breakdown should immediately discontinue use of topical NSAIDs and should be closely monitored for corneal health.
Postmarketing experience with topical NSAIDs suggests that patients with complicated ocular surgeries, corneal denervation, corneal epithelial defects, diabetes mellitus, ocular surface diseases (e.g., dry eye syndrome), rheumatoid arthritis, or repeat ocular surgeries within a short period of time may be at increased risk for corneal adverse events which may become sight threatening. Topical NSAIDs should be used with caution in these patients.
Postmarketing experience with topical NSAIDs also suggests that use more than 1 day prior to surgery or use beyond 14 days post-surgery may increase patient risk for the occurrence and severity of corneal adverse events.
6. Adverse Reactions/Side Effects
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to the rates in the clinical studies of another drug and may not reflect the rates observed in practice.
6.1 Clinical Studies Experience
The most common adverse reactions were reported in 1-6% of patients and included increased intraocular pressure, conjunctival hyperemia and/or hemorrhage, corneal edema, ocular pain, headache, tearing and vision blurred. Some of these reactions may be the consequence of the cataract surgical procedure.
| ACUVAIL
ketorolac tromethamine solution/ drops |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Allergan, Inc. (144796497) |