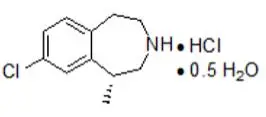
Drug Detail:Belviq (Lorcaserin [ lor-kas-er-in ])
Drug Class: Anorexiants
Highlights of Prescribing Information
BELVIQ® (lorcaserin hydrochloride) tablets, for oral use, CIV
BELVIQ XR® (lorcaserin hydrochloride) extended-release tablets for oral use, CIV
Initial U.S. Approval: 2012
Indications and Usage for Belviq
BELVIQ/BELVIQ XR is a serotonin 2C receptor agonist indicated as an adjunct to a reduced-calorie diet and increased physical activity for chronic weight management in adults with an initial body mass index (BMI) of:
- 30 kg/m2 or greater (obese) (1) or
- 27 kg/m2 or greater (overweight) in the presence of at least one weight-related comorbid condition, (e.g., hypertension, dyslipidemia, type 2 diabetes) (1)
Limitations of Use:
- The safety and efficacy of coadministration with other products for weight loss have not been established (1)
- The effect of BELVIQ/BELVIQ XR on cardiovascular morbidity and mortality has not been established (1)
Belviq Dosage and Administration
- BELVIQ 10 mg one tablet twice daily
- BELVIQ XR 20 mg one tablet once daily
- Do not chew, crush or divide BELVIQ XR tablets (2)
- BELVIQ/BELVIQ XR can be taken with or without food (2)
- Discontinue if 5% weight loss is not achieved by week 12 (2)
Dosage Forms and Strengths
Tablets: 10 mg film-coated (3)
Extended-release Tablets: 20 mg film-coated (3)
Contraindications
- Pregnancy (4)
- Hypersensitivity (4)
Warnings and Precautions
- Serotonin Syndrome or Neuroleptic Malignant Syndrome (NMS)-like Reactions: The safety of coadministration with other serotonergic or antidopaminergic agents has not been established. Manage with immediate BELVIQ/BELVIQ XR discontinuation and provide supportive treatment. (5.1)
- Valvular heart disease: If signs or symptoms develop consider BELVIQ/BELVIQ XR discontinuation and evaluate the patient for possible valvulopathy. (5.2)
- Cognitive Impairment: May cause disturbances in attention or memory. Caution with use of hazardous machinery when starting BELVIQ/BELVIQ XR treatment. (5.3)
- Psychiatric Disorders, including euphoria and dissociation: Do not exceed recommended dose of BELVIQ 10 mg twice daily or BELVIQ XR 20 mg once daily. (5.4)
- Monitor for depression or suicidal thoughts. Discontinue if symptoms develop. (5.4)
- Use of Antidiabetic Medications: weight loss may cause hypoglycemia. Monitor blood glucose. BELVIQ/BELVIQ XR has not been studied in patients taking insulin. (5.5)
- Priapism: Patients should seek emergency treatment if an erection lasts >4 hours. Use BELVIQ/BELVIQ XR with caution in patients predisposed to priapism. (5.6)
Adverse Reactions/Side Effects
Most common adverse reactions (greater than 5%) in non-diabetic patients are headache, dizziness, fatigue, nausea, dry mouth, and constipation, and in diabetic patients are hypoglycemia, headache, back pain, cough, and fatigue. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Eisai Inc. at 1-888-274-2378 or FDA at 1-800-FDA-1088 or at www.fda.gov/medwatch.
Drug Interactions
Serotonergic drugs (selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), monoamine oxidase inhibitors (MAOIs), triptans, bupropion, dextromethorphan, St. John’s Wort): use with extreme caution due to the risk of serotonin syndrome. (7.1)
Use In Specific Populations
- Lactation: BELVIQ/BELVIQ XR is not recommended when breastfeeding. (8.2)
- Pediatric Use: Safety and effectiveness not established and use not recommended. (8.4)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 4/2018
Related/similar drugs
Ozempic, Wegovy, phentermine, semaglutide, Saxenda, AlliFull Prescribing Information
1. Indications and Usage for Belviq
BELVIQ/BELVIQ XR is indicated as an adjunct to a reduced-calorie diet and increased physical activity for chronic weight management in adult patients with an initial body mass index (BMI) of:
- 30 kg/m2 or greater (obese), or
- 27 kg/m2 or greater (overweight) in the presence of at least one weight related comorbid condition (e.g., hypertension, dyslipidemia, type 2 diabetes)
[see Dosage and Administration (2)]
Limitations of Use:
- The safety and efficacy of coadministration of BELVIQ/BELVIQ XR with other products intended for weight loss including prescription drugs (e.g., phentermine), over-the-counter drugs, and herbal preparations have not been established.
- The effect of BELVIQ/BELVIQ XR on cardiovascular morbidity and mortality has not been established.
2. Belviq Dosage and Administration
- The recommended dose for BELVIQ is one 10 mg tablet administered orally twice daily.
- The recommended dose for BELVIQ XR is one 20 mg tablet administered orally once daily.
- Do not exceed recommended dose [see Warnings and Precautions (5.4)].
- BELVIQ/BELVIQ XR can be taken with or without food.
- Swallow BELVIQ XR tablets whole and do not chew, crush, or divide.
- Response to therapy should be evaluated by week 12. If a patient has not lost at least 5% of baseline body weight, discontinue BELVIQ/BELVIQ XR, as it is unlikely that the patient will achieve and sustain clinically meaningful weight loss with continued treatment [see Clinical Studies (14)].
- BMI is calculated by dividing weight (in kg) by height (in meters) squared. A BMI chart for height in inches and weight in pounds is provided below:
| Weight | (lb) | 125 | 130 | 135 | 140 | 145 | 150 | 155 | 160 | 165 | 170 | 175 | 180 | 185 | 190 | 195 | 200 | 205 | 210 | 215 | 220 | 225 | |
| (kg) | 56. 8 | 59. 1 | 61. 4 | 63. 6 | 65. 9 | 68. 2 | 70. 5 | 72. 7 | 75. 0 | 77. 3 | 79. 5 | 81. 8 | 84. 1 | 86. 4 | 88. 6 | 90. 9 | 93. 2 | 95. 5 | 97. 7 | 100. 0 | 102. 3 | ||
| Height | |||||||||||||||||||||||
| (in) | (cm) | ||||||||||||||||||||||
| 58 | 147. 3 | 26 | 27 | 28 | 29 | 30 | 31 | 32 | 34 | 35 | 36 | 37 | 38 | 39 | 40 | 41 | 42 | 43 | 44 | 45 | 46 | 47 | |
| 59 | 149. 9 | 25 | 26 | 27 | 28 | 29 | 30 | 31 | 32 | 33 | 34 | 35 | 36 | 37 | 38 | 39 | 40 | 41 | 43 | 44 | 45 | 46 | |
| 60 | 152. 4 | 24 | 25 | 26 | 27 | 28 | 29 | 30 | 31 | 32 | 33 | 34 | 35 | 36 | 37 | 38 | 39 | 40 | 41 | 42 | 43 | 44 | |
| 61 | 154. 9 | 24 | 25 | 26 | 27 | 27 | 28 | 29 | 30 | 31 | 32 | 33 | 34 | 35 | 36 | 37 | 38 | 39 | 40 | 41 | 42 | 43 | |
| 62 | 157. 5 | 23 | 24 | 25 | 26 | 27 | 27 | 28 | 29 | 30 | 31 | 32 | 33 | 34 | 35 | 36 | 37 | 38 | 38 | 39 | 40 | 41 | |
| 63 | 160. 0 | 22 | 23 | 24 | 25 | 26 | 27 | 28 | 28 | 29 | 30 | 31 | 32 | 33 | 34 | 35 | 36 | 36 | 37 | 38 | 39 | 40 | |
| 64 | 162. 6 | 22 | 22 | 23 | 24 | 25 | 26 | 27 | 28 | 28 | 29 | 30 | 31 | 32 | 33 | 34 | 34 | 35 | 36 | 37 | 38 | 39 | |
| 65 | 165. 1 | 21 | 22 | 23 | 23 | 24 | 25 | 26 | 27 | 28 | 28 | 29 | 30 | 31 | 32 | 33 | 33 | 34 | 35 | 36 | 37 | 38 | |
| 66 | 167. 6 | 20 | 21 | 22 | 23 | 23 | 24 | 25 | 26 | 27 | 27 | 28 | 29 | 30 | 31 | 32 | 32 | 33 | 34 | 35 | 36 | 36 | |
| 67 | 170. 2 | 20 | 20 | 21 | 22 | 23 | 24 | 24 | 25 | 26 | 27 | 27 | 28 | 29 | 30 | 31 | 31 | 32 | 33 | 34 | 35 | 35 | |
| 68 | 172. 7 | 19 | 20 | 21 | 21 | 22 | 23 | 24 | 24 | 25 | 26 | 27 | 27 | 28 | 29 | 30 | 30 | 31 | 32 | 33 | 34 | 34 | |
| 69 | 175. 3 | 18 | 19 | 20 | 21 | 21 | 22 | 23 | 24 | 24 | 25 | 26 | 27 | 27 | 28 | 29 | 30 | 30 | 31 | 32 | 33 | 33 | |
| 70 | 177. 8 | 18 | 19 | 19 | 20 | 21 | 22 | 22 | 23 | 24 | 24 | 25 | 26 | 27 | 27 | 28 | 29 | 29 | 30 | 31 | 32 | 32 | |
| 71 | 180. 3 | 17 | 18 | 19 | 20 | 20 | 21 | 22 | 22 | 23 | 24 | 24 | 25 | 26 | 27 | 27 | 28 | 29 | 29 | 30 | 31 | 31 | |
| 72 | 182. 9 | 17 | 18 | 18 | 19 | 20 | 20 | 21 | 22 | 22 | 23 | 24 | 24 | 25 | 26 | 27 | 27 | 28 | 29 | 29 | 30 | 31 | |
| 73 | 185. 4 | 17 | 17 | 18 | 19 | 19 | 20 | 20 | 21 | 22 | 22 | 23 | 24 | 24 | 25 | 26 | 26 | 27 | 28 | 28 | 29 | 30 | |
| 74 | 188. 0 | 16 | 17 | 17 | 18 | 19 | 19 | 20 | 21 | 21 | 22 | 23 | 23 | 24 | 24 | 25 | 26 | 26 | 27 | 28 | 28 | 29 | |
| 75 | 190. 5 | 16 | 16 | 17 | 18 | 18 | 19 | 19 | 20 | 21 | 21 | 22 | 23 | 23 | 24 | 24 | 25 | 26 | 26 | 27 | 28 | 28 | |
| 76 | 193. 0 | 15 | 16 | 16 | 17 | 18 | 18 | 19 | 20 | 20 | 21 | 21 | 22 | 23 | 23 | 24 | 24 | 25 | 26 | 26 | 27 | 27 | |
3. Dosage Forms and Strengths
- BELVIQ 10 mg tablets: blue, film-coated, round, biconvex, debossed with “A” on one side and “10” on the other side.
- BELVIQ XR 20 mg extended-release tablets: orange, film-coated, round, biconvex, debossed with “A” on one side and “20” on the other side.
4. Contraindications
● Pregnancy: Weight loss in a pregnant woman offers no benefit and may result in fetal harm [see Use in Specific Populations (8.1)].
● Hypersensitivity: BELVIQ/BELVIQ XR is contraindicated in patients with prior hypersensitivity reactions to lorcaserin or to any of the product components. Hypersensitivity reactions have been reported [see Adverse Reactions (6.2)].
5. Warnings and Precautions
5.1 Serotonin Syndrome or Neuroleptic Malignant Syndrome (NMS)-like Reactions
BELVIQ/BELVIQ XR is a serotonergic drug. The development of a potentially life-threatening serotonin syndrome or Neuroleptic Malignant Syndrome (NMS)-like reactions have been reported during use of serotonergic drugs, including, but not limited to, selective serotonin-norepinephrine reuptake inhibitors (SNRIs) and selective serotonin reuptake inhibitors (SSRIs), tricyclic antidepressants (TCAs), bupropion, triptans, dietary supplements such as St. John’s Wort and tryptophan, drugs that impair metabolism of serotonin (including monoamine oxidase inhibitors [MAOIs]), dextromethorphan, lithium, tramadol, antipsychotics or other dopamine antagonists, particularly when used in combination [see Drug Interactions (7.1)].
Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, coma), autonomic instability (e.g., tachycardia, labile blood pressure, hyperthermia), neuromuscular aberrations (e.g.,hyperreflexia, incoordination) and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). Serotonin syndrome, in its most severe form, can resemble neuroleptic malignant syndrome, which includes hyperthermia, muscle rigidity, autonomic instability with possible rapid fluctuation of vital signs, and mental status changes. Patients should be monitored for the emergence of serotonin syndrome or NMS-like signs and symptoms.
The safety of BELVIQ/BELVIQ XR when coadministered with other serotonergic or antidopaminergic agents, including antipsychotics, or drugs that impair metabolism of serotonin, including MAOIs, has not been systematically evaluated and has not been established.
If concomitant administration of BELVIQ/BELVIQ XR with an agent that affects the serotonergic neurotransmitter system is clinically warranted, extreme caution and careful observation of the patient is advised, particularly during treatment initiation and dose increases. Treatment with BELVIQ/BELVIQ XR and any concomitant serotonergic or antidopaminergic agents, including antipsychotics, should be discontinued immediately if the above events occur and supportive symptomatic treatment should be initiated [see Adverse Reactions (6.1) and Drug Interactions (7.1)].
5.2 Valvular Heart Disease
Regurgitant cardiac valvular disease, primarily affecting the mitral and/or aortic valves, has been reported in patients who took serotonergic drugs with 5-HT2B receptor agonist activity. The etiology of the regurgitant valvular disease is thought to be activation of 5-HT2B receptors on cardiac interstitial cells. At therapeutic concentrations, lorcaserin is selective for 5-HT2C receptors as compared to 5-HT2B receptors. In clinical trials of 1-year duration, 2.4% of patients receiving BELVIQ and 2.0% of patients receiving placebo developed echocardiographic criteria for valvular regurgitation at one year (mild or greater aortic regurgitation and/or moderate or greater mitral regurgitation): none of these patients was symptomatic [see Adverse Reactions (6.1) and Clinical Pharmacology (12.1)].
BELVIQ/BELVIQ XR has not been studied in patients with congestive heart failure or hemodynamically-significant valvular heart disease. Preliminary data suggest that 5HT2B receptors may be overexpressed in congestive heart failure; therefore, BELVIQ/BELVIQ XR should be used with caution in patients with congestive heart failure.
BELVIQ/BELVIQ XR should not be used in combination with serotonergic and dopaminergic drugs that are potent 5-HT2B receptor agonists and are known to increase the risk for cardiac valvulopathy (e.g., cabergoline).
Patients who develop signs or symptoms of valvular heart disease, including dyspnea, dependent edema, congestive heart failure, or a new cardiac murmur while being treated with BELVIQ/BELVIQ XR should be evaluated and discontinuation of BELVIQ/BELVIQ XR should be considered.
5.3 Cognitive Impairment
In clinical trials of at least one year in duration, impairments in attention and memory were reported adverse reactions associated with 1.9% of patients treated with BELVIQ and 0.5% of patients treated with placebo, and led to discontinuation in 0.3% and 0.1% of these patients, respectively. Other reported adverse reactions associated with BELVIQ in clinical trials included confusion, somnolence, and fatigue [see Adverse Reactions (6.1)].
Since BELVIQ/BELVIQ XR have the potential to impair cognitive function, patients should be cautioned about operating hazardous machinery, including automobiles, until they are reasonably certain that BELVIQ/BELVIQ XR therapy does not affect them adversely [see Patient Counseling Information (17)].
5.4 Psychiatric Disorders
Events of euphoria, hallucination, and dissociation were seen with BELVIQ at supratherapeutic doses in short- term studies [see Adverse Reactions (6.1), Drug Abuse and Dependence (9.2), and Overdosage (10)]. In clinical trials of at least 1-year in duration, 6 patients (0.2%) treated with BELVIQ developed euphoria, as compared with 1 patient (<0.1%) treated with placebo. Doses should not exceed BELVIQ 10 mg twice daily or BELVIQ XR 20 mg once daily.
Some drugs that target the central nervous system have been associated with depression or suicidal ideation. Patients treated with BELVIQ/BELVIQ XR should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior. Discontinue BELVIQ/BELVIQ XR in patients who experience suicidal thoughts or behaviors [see Adverse Reactions (6.1)].
5.5 Potential Risk of Hypoglycemia in Patients with Type 2 Diabetes Mellitus on Anti- diabetic Therapy
Weight loss may increase the risk of hypoglycemia in patients with type 2 diabetes mellitus treated with insulin and/or insulin secretagogues (e.g., sulfonylureas); hypoglycemia was observed in clinical trials with BELVIQ. BELVIQ/BELVIQ XR has not been studied in combination with insulin. Measurement of blood glucose levels prior to starting BELVIQ/BELVIQ XR and during BELVIQ/BELVIQ XR treatment is recommended in patients with type 2 diabetes. Decreases in medication doses for anti-diabetic medications which are non-glucose-dependent should be considered to mitigate the risk of hypoglycemia. If a patient develops hypoglycemia after starting BELVIQ/BELVIQ XR, appropriate changes should be made to the anti-diabetic drug regimen [see Adverse Reactions (6.1)].
5.6 Priapism
Priapism (painful erections greater than 6 hours in duration) is a potential effect of 5-HT2C receptor agonism. If not treated promptly, priapism can result in irreversible damage to the erectile tissue. Men who have an erection lasting greater than 4 hours, whether painful or not, should immediately discontinue the drug and seek emergency medical attention.
BELVIQ/BELVIQ XR should be used with caution in men who have conditions that might predispose them to priapism (e.g., sickle cell anemia, multiple myeloma, or leukemia), or in men with anatomical deformation of the penis (e.g., angulation, cavernosal fibrosis, or Peyronie's disease). There is limited experience with the combination of BELVIQ/BELVIQ XR and medication indicated for erectile dysfunction (e.g., phosphodiesterase type 5 inhibitors). Therefore, the combination of BELVIQ/BELVIQ XR and these medications should be used with caution.
5.7 Heart Rate Decreases
In clinical trials of at least 1-year in duration, the mean change in heart rate (HR) was -1.2 beats per minute (bpm) in BELVIQ and -0.4 bpm in placebo-treated patients without diabetes and -2.0 beats per minute (bpm) in BELVIQ and -0.4 bpm in placebo-treated patients with type 2 diabetes. The incidence of HR less than 50 bpm was 5.3% in BELVIQ and 3.2% in placebo-treated patients without diabetes and 3.6% in BELVIQ and 2.0% in placebo-treated patients with type 2 diabetes. In the combined population, adverse reactions of bradycardia occurred in 0.3% of BELVIQ and 0.1% of placebo-treated patients. Use BELVIQ/BELVIQ XR with caution in patients with bradycardia or a history of heart block greater than first degree.
5.8 Hematological Changes
In clinical trials of at least one year in duration, adverse reactions of decreases in white blood cell count (including leukopenia, lymphopenia, neutropenia, and decreased white cell count) were reported in 0.4% of patients treated with BELVIQ as compared to 0.2% of patients treated with placebo. Adverse reactions of decreases in red blood cell count (including anemia and decreases in hemoglobin and hematocrit) were reported by 1.3% of patients treated with BELVIQ as compared to 1.2% treated with placebo [see Adverse Reactions (6.1)]. Consider periodic monitoring of complete blood count during treatment with BELVIQ/BELVIQ XR.
5.9 Prolactin Elevation
Lorcaserin moderately elevates prolactin levels. In a subset of placebo-controlled clinical trials of at least one year in duration, elevations of prolactin greater than the upper limit of normal, two times the upper limit of normal, and five times the upper limit of normal, measured both before and 2 hours after dosing, occurred in 6.7%, 1.7%, and 0.1% of BELVIQ-treated patients and 4.8%, 0.8%, and 0.0% of placebo-treated patients, respectively [see Adverse Reactions (6.1)]. Prolactin should be measured when symptoms and signs of prolactin excess are suspected (e.g., galactorrhea, gynecomastia). There was one patient treated with BELVIQ who developed a prolactinoma during the trial. The relationship of BELVIQ/BELVIQ XR to the prolactinoma in this patient is unknown.
6. Adverse Reactions/Side Effects
The following important adverse reactions are described below and elsewhere in labeling:
- Serotonin Syndrome or NMS-like Reactions [see Warnings and Precautions (5.1)]
- Valvular Heart Disease [see Warnings and Precautions (5.2)]
- Cognitive Impairment [see Warnings and Precautions (5.3)]
- Psychiatric Disorders [see Warnings and Precautions (5.4)]
- Hypoglycemia [see Warnings and Precautions (5.5)]
- Heart Rate Decreases [see Warnings and Precautions (5.7)]
- Hematological Changes [see Warnings and Precautions (5.8)]
- Prolactin Elevation [see Warnings and Precautions (5.9)]
6.1 Clinical Trials Experience
In the BELVIQ placebo-controlled clinical database of trials of at least one year in duration, of 6888 patients (3451 BELVIQ vs. 3437 placebo; age range 18-66 years, 79.3% women, 66.6% Caucasians, 19.2% Blacks, 11.8% Hispanics, 2.4% other, 7.4% type 2 diabetics), a total of 1969 patients were exposed to BELVIQ 10 mg twice daily for 1 year and 426 patients were exposed for 2 years.
In clinical trials of at least one year in duration, 8.6% of patients treated with BELVIQ prematurely discontinued treatment due to adverse reactions, compared with 6.7% of placebo-treated patients. The most common adverse reactions leading to discontinuation more often among BELVIQ treated patients than placebo were headache (1.3% vs. 0.8%), depression (0.9% vs. 0.5%), and dizziness (0.7% vs. 0.2%).
Most Common Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most common adverse reactions for non-diabetic patients (greater than 5% and more commonly than placebo) treated with BELVIQ compared to placebo were headache, dizziness, fatigue, nausea, dry mouth, and constipation. The most common adverse reactions for diabetic patients were hypoglycemia, headache, back pain, cough, and fatigue. Adverse reactions that were reported by greater than or equal to 2% of patients and were more frequently reported by patients taking BELVIQ compared to placebo are summarized in Table 2 (non-diabetic subjects) and Table 3 (subjects with type 2 diabetes mellitus).
| Number of patients (%) | ||
| Adverse Reaction | BELVIQ
N=3195 | Placebo
N=3185 |
| Gastrointestinal Disorders | ||
| Nausea | 264 (8.3) | 170 (5.3) |
| Diarrhea | 207 (6.5) | 179 (5.6) |
| Constipation | 186 (5.8) | 125 (3.9) |
| Dry mouth | 169 (5.3) | 74 (2.3) |
| Vomiting | 122 (3.8) | 83 (2.6) |
| General Disorders And Administration Site Conditions | ||
| Fatigue | 229 (7.2) | 114 (3.6) |
| Infections And Infestations | ||
| Upper respiratory tract infection | 439 (13.7) | 391 (12.3) |
| Nasopharyngitis | 414 (13.0) | 381 (12.0) |
| Urinary tract infection | 207 (6.5) | 171 (5.4) |
| Musculoskeletal And Connective Tissue Disorders | ||
| Back pain | 201 (6.3) | 178 (5.6) |
| Musculoskeletal pain | 65 (2.0) | 43 (1.4) |
| Nervous System Disorders | ||
| Headache | 537 (16.8) | 321 (10.1) |
| Dizziness | 270 (8.5) | 122 (3.8) |
| Respiratory, Thoracic And Mediastinal Disorders | ||
| Cough | 136 (4.3) | 109 (3.4) |
| Oropharyngeal pain | 111 (3.5) | 80 (2.5) |
| Sinus congestion | 93 (2.9) | 78 (2.4) |
| Skin And Subcutaneous Tissue Disorders | ||
| Rash | 67 (2.1) | 58 (1.8) |
| Number of patients (%) | ||
| Adverse Reaction | BELVIQ
N=256 | Placebo
N=252 |
| Gastrointestinal Disorders | ||
| Nausea | 24 (9.4) | 20 (7.9) |
| Toothache | 7 (2.7) | 0 |
| General Disorders And Administration Site Conditions | ||
| Fatigue | 19 (7.4) | 10 (4.0) |
| Peripheral edema | 12 (4.7) | 6 (2.4) |
| Immune System Disorders | ||
| Seasonal allergy | 8 (3.1) | 2 (0.8) |
| Infections And Infestations | ||
| Nasopharyngitis | 29 (11.3) | 25 (9.9) |
| Urinary tract infection | 23 (9.0) | 15 (6.0) |
| Gastroenteritis | 8 (3.1) | 5 (2.0) |
| Metabolism And Nutrition Disorders | ||
| Hypoglycemia | 75 (29.3) | 53 (21.0) |
| Worsening of diabetes mellitus | 7 (2.7) | 2 (0.8) |
| Decreased appetite | 6 (2.3) | 1 (0.4) |
| Musculoskeletal And Connective Tissue Disorders | ||
| Back pain | 30 (11.7) | 20 (7.9) |
| Muscle spasms | 12 (4.7) | 9 (3.6) |
| Nervous System Disorders | ||
| Headache | 37 (14.5) | 18 (7.1) |
| Dizziness | 18 (7.0) | 16 (6.3) |
| Psychiatric Disorders | ||
| Anxiety | 9 (3.5) | 8 (3.2) |
| Insomnia | 9 (3.5) | 6 (2.4) |
| Stress | 7 (2.7) | 3 (1.2) |
| Depression | 6 (2.3) | 5 (2.0) |
| Respiratory, Thoracic And Mediastinal Disorders | ||
| Cough | 21 (8.2) | 11 (4.4) |
| Vascular Disorders | ||
| Hypertension | 13 (5.1) | 8 (3.2) |
Other Adverse Reactions
Serotonin-associated Adverse Reactions
SSRIs, SNRIs, bupropion, tricyclic antidepressants, and MAOIs were excluded from the BELVIQ trials. Triptans and dextromethorphan were permitted: 2% and 15%, respectively, of patients without diabetes and 1% and 12%, respectively, of patients with type 2 diabetes experienced concomitant use at some point during the trials. Two patients treated with BELVIQ in the clinical program experienced a constellation of symptoms and signs consistent with serotonergic excess, including one patient on concomitant dextromethorphan who reported an event of serotonin syndrome. Some symptoms of possible serotonergic etiology that are included in the criteria for serotonin syndrome were reported by patients treated with BELVIQ and placebo during clinical trials of at least 1 year in duration. In both groups, chills were the most frequent of these events (1.0% vs. 0.2%, respectively), followed by tremor (0.3% vs. 0.2%), confusional state (0.2% vs. less than 0.1%), disorientation (0.1% vs. 0.1%) and hyperhidrosis (0.1% vs. 0.2%). Because serotonin syndrome has a very low incidence, an association between BELVIQ/BELVIQ XR and serotonin syndrome cannot be excluded on the basis of clinical trial results [see Warnings and Precautions (5.1)].
Hypoglycemia in Patients with Type 2 Diabetes
In a clinical trial of patients with type 2 diabetes mellitus, severe hypoglycemia (requiring the assistance of another person, requiring intravenous glucose, or hospitalization) occurred in 4 (1.6%) of BELVIQ-treated patients and in 1 (0.4%) placebo-treated patient. Of these 4 BELVIQ-treated patients, all were concomitantly using a sulfonylurea (with or without metformin). BELVIQ/BELVIQ XR has not been studied in patients taking insulin. Hypoglycemia defined as blood sugar less than or equal to 65 mg/dL and with symptoms occurred in 19 (7.4%) BELVIQ-treated patients and 16 (6.3%) placebo-treated patients.
Cognitive Impairment
In clinical trials of at least 1-year duration, adverse reactions related to cognitive impairment (e.g., difficulty with concentration/attention, difficulty with memory, and confusion) occurred in 2.3% of patients taking BELVIQ and 0.7% of patients taking placebo.
Psychiatric Disorders
Psychiatric disorders leading to hospitalization or drug withdrawal occurred more frequently in patients treated with BELVIQ (2.2%) as compared to placebo (1.1%) in non-diabetic patients.
Euphoria. In short-term studies with healthy individuals, the incidence of euphoric mood following supratherapeutic doses of BELVIQ (40 and 60 mg) was increased as compared to placebo [see Drug Abuse and Dependence (9.2)]. In clinical trials of at least 1-year duration in obese patients, euphoria was observed in 0.17% of patients taking BELVIQ and 0.03% taking placebo.
Depression and Suicidality. In trials of at least one year in duration, reports of depression/mood problems occurred in 2.6% BELVIQ-treated vs. 2.4% placebo-treated and suicidal ideation occurred in 0.6% BELVIQ- treated vs. 0.4% placebo-treated patients. 1.3% of BELVIQ patients vs. 0.6% of placebo patients discontinued drug due to depression-, mood-, or suicidal ideation-related events.
Laboratory Abnormalities
Lymphocyte and Neutrophil Counts. In clinical trials of at least 1-year duration, lymphocyte counts were below the lower limit of normal in 12.2% of patients taking BELVIQ and 9.0% taking placebo, and neutrophil counts were low in 5.6% and 4.3%, respectively.
Hemoglobin. In clinical trials of at least 1-year duration, 10.4% of patients taking BELVIQ and 9.3% taking placebo had hemoglobin below the lower limit of normal at some point during the trials.
Prolactin. In clinical trials, elevations of prolactin greater than the upper limit of normal, two times the upper limit of normal, and five times the upper limit of normal, occurred in 6.7%, 1.7%, and 0.1% of BELVIQ-treated patients and 4.8%, 0.8%, and 0.0% of placebo-treated patients, respectively.
Eye disorders
More patients on BELVIQ reported an eye disorder than patients on placebo in clinical trials of patients without diabetes (4.5% vs. 3.0%) and with type 2 diabetes (5.9% vs. 1.6%). In the population without diabetes, events of blurred vision, dry eye, and visual impairment occurred in BELVIQ-treated patients at an incidence greater than that of placebo. In the population with type 2 diabetes, visual disorders, conjunctival infections, irritations, and inflammations, ocular sensation disorders, and cataract conditions occurred in BELVIQ-treated patients at an incidence greater than placebo.
Echocardiographic Safety Assessments
The possible occurrence of regurgitant cardiac valve disease was prospectively evaluated in 7794 patients in three clinical trials of at least one year in duration, 3451 of whom took BELVIQ. The primary echocardiographic safety parameter was the proportion of patients who developed echocardiographic criteria of mild or greater aortic insufficiency and/or moderate or greater mitral insufficiency from baseline to 1 year. At 1 year, 2.4% of patients who received BELVIQ and 2.0% of patients who received placebo developed valvular regurgitation. The relative risk for valvulopathy with BELVIQ is summarized in Table 4. BELVIQ/BELVIQ XR was not studied in patients with congestive heart failure or hemodynamically-significant valvular heart disease [see Warnings and Precautions (5.2)].
| Study 1 | Study 2 | Study 3 | ||||
| BELVIQ
N=1278 | Placebo
N=1191 | BELVIQ N=1208 | Placebo
N=1153 | BELVIQ
N=210 | Placebo
N=209 |
|
| FDA-defined
Valvulopathy, n (%) | 34 (2.7) | 28 (2.4) | 24 (2.0) | 23 (2.0) | 6 (2.9) | 1 (0.5) |
| Relative Risk (95% CI) | 1.13 (0.69, 1.85) | 1.00 (0.57, 1.75) | 5.97 (0.73, 49.17) | |||
| Pooled RR (95% CI) | 1.16 (0.81, 1.67) | |||||
1 Patients without valvulopathy at baseline who received study medication and had a post-baseline echocardiogram; ITT-intention-to-treat; LOCF-last observation carried forward
6.2 Post-Marketing Experience
The following adverse reactions have been identified during post approval use. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: drug hypersensitivity
7. Drug Interactions
7.1 Use with Other Agents that Affect Serotonin Pathways
Based on the mechanism of action of BELVIQ/BELVIQ XR and the theoretical potential for serotonin syndrome, use with extreme caution in combination with other drugs that may affect the serotonergic neurotransmitter systems, including, but not limited to, triptans, monoamine oxidase inhibitors (MAOIs, including linezolid, an antibiotic which is a reversible non-selective MAOI), selective serotonin reuptake inhibitors (SSRIs), selective serotonin- norepinephrine reuptake inhibitors (SNRIs), dextromethorphan, tricyclic antidepressants (TCAs), bupropion, lithium, tramadol, tryptophan, and St. John’s Wort [see Warnings and Precautions (5.1)].
8. Use In Specific Populations
8.1 Pregnancy
Risk Summary
BELVIQ/BELVIQ XR is contraindicated during pregnancy, because weight loss offers no benefit to a pregnant woman and may result in fetal harm [see Clinical Considerations]. Limited data on lorcaserin use in pregnant women are not sufficient to determine a drug-associated risk of major congenital malformations or miscarriage. No adverse developmental effects were observed when lorcaserin was administered to pregnant rats and rabbits during organogenesis at exposures up to 44- and 19-times the clinical dose of 20 mg daily, respectively. In rats, maternal exposure to lorcaserin in late pregnancy resulted in lower body weight in offspring which persisted to adulthood [see Data]. Advise pregnant women of the potential risk to a fetus.
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage of clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Clinical Considerations
Disease-associated maternal and/or embryofetal risk
Appropriate weight gain based on pre-pregnancy weight is currently recommended for all pregnant women, including those who are already overweight or obese, due to the obligatory weight gain that occurs in maternal tissues during pregnancy.
Data
Animal Data
Reproduction studies were performed in pregnant rats and rabbits that were administered lorcaserin hydrochloride during the period of embryofetal organogenesis. Plasma exposures up to 44 and 19 times the clinical dose of 20 mg daily in pregnant rats and rabbits, respectively, did not reveal evidence of teratogenicity or embryolethality with lorcaserin hydrochloride.
In a pre- and postnatal development study, maternal rats were dosed from gestation through post-natal day 21 at 5, 15, and 50 mg/kg lorcaserin hydrochloride; pups were indirectly exposed in utero and throughout lactation. Stillborns and lower pup viability was observed at 50 mg/kg, or 44 times the clinical dose of 20 mg daily, based on AUC. All other doses lowered pup body weight similarly at birth which persisted to adulthood; however, no developmental abnormalities were observed and reproductive performance was not affected.
8.2 Lactation
Risk Summary
There are no data on the presence of lorcaserin in human milk, the effects on the breastfed infant, or the effects on milk production. Because of the potential for serious adverse reactions in a breastfed infant, advise women that use of BELVIQ/BELVIQ XR is not recommended while breastfeeding.
8.4 Pediatric Use
The safety and effectiveness of BELVIQ/BELVIQ XR in pediatric patients below the age of 18 have not been established and the use of BELVIQ/BELVIQ XR is not recommended in pediatric patients.
8.6 Renal Impairment
No dose adjustment of BELVIQ/BELVIQ XR is required in patients with mild renal impairment. Use BELVIQ/BELVIQ XR with caution in patients with moderate renal impairment. Use of BELVIQ/BELVIQ XR in patients with severe renal impairment or end stage renal disease is not recommended [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
Dose adjustment is not required for patients with mild hepatic impairment (Child-Pugh score 5-6) to moderate hepatic impairment (Child-Pugh score 7-9). The effect of severe hepatic impairment on lorcaserin was not evaluated. Use BELVIQ/BELVIQ XR with caution in patients with severe hepatic impairment [see Clinical Pharmacology (12.3)].
9. Drug Abuse and Dependence
9.1 Controlled Substance
BELVIQ/BELVIQ XR is listed in Schedule IV of the Controlled Substances Act.
9.2 Abuse
In a human abuse potential study in recreational drug abusers, supratherapeutic oral doses of BELVIQ (40 and 60 mg) produced up to two- to six-fold increases on measures of “High”, “Good Drug Effects”, “Hallucinations” and “Sedation” compared to placebo. These responses were similar to those produced by oral administration of the positive control drugs, zolpidem (15 and 30 mg) and ketamine (100 mg). In this study, the incidence of the adverse reaction of euphoria following lorcaserin administration (40 and 60 mg; 19%) is similar to the incidence following zolpidem administration (13-16%), but less than the incidence following ketamine administration (50%). The duration of euphoria following lorcaserin administration persisted longer (> 9 hours) than that following zolpidem (1.5 hours) or ketamine (2.5 hours) administration.
Overall, in short-term studies with healthy individuals, the rate of euphoria following oral administration of lorcaserin was 16% following 40 mg (n = 11 of 70) and 19% following 60 mg (n = 6 of 31). However, in clinical studies with obese patients with durations of 4 weeks to 2 years, the incidence of euphoria and hallucinations following oral doses of lorcaserin up to 40 mg was low (< 1.0%).
10. Overdosage
No experience with overdose of BELVIQ/BELVIQ XR is available. In clinical studies that used doses that were higher than the recommended dose, the most frequent adverse reactions associated with BELVIQ were headache, nausea, abdominal discomfort, and dizziness. Single 40- and 60-mg doses of BELVIQ caused euphoria, altered mood, and hallucination in some subjects. Treatment of overdose should consist of BELVIQ/BELVIQ XR discontinuation and general supportive measures in the management of overdosage. Lorcaserin is not eliminated to a therapeutically significant degree by hemodialysis.
12. Belviq - Clinical Pharmacology
12.1 Mechanism of Action
Lorcaserin is believed to decrease food consumption and promote satiety by selectively activating 5-HT2C receptors on anorexigenic pro-opiomelanocortin neurons located in the hypothalamus. The exact mechanism of action is not known.
Lorcaserin at the recommended daily dose selectively interacts with 5-HT2C receptors as compared to 5-HT2A and 5-HT2B receptors (see Table 5), other 5-HT receptor subtypes, the 5-HT receptor transporter, and 5-HT reuptake sites.
| Serotonin Receptor Subtype | EC50, nM | Ki, nM |
| 5HT2C | 39 | 13 |
| 5HT2B | 2380 | 147 |
| 5HT2A | 553 | 92 |
12.3 Pharmacokinetics
Absorption
BELVIQ
Lorcaserin is absorbed from the gastrointestinal tract with peak plasma concentration occurring 1.5 - 2 hours after oral dosing. The absolute bioavailability of lorcaserin has not been determined. Steady state is reached within 3 days after twice daily dosing, and accumulation is estimated to be approximately 70%.
Effect of Food. Twelve adult volunteers (6 men and 6 women) were given a single 10 mg oral dose of BELVIQ in a fasted state and after administration of a high fat (approximately 50% of total caloric content of the meal) and high-calorie (approximately 800–1000 calories) meal. The Cmax increased approximately 9% and exposure (AUC) increased approximately 5% under fed conditions. Tmax was delayed approximately 1 hour in the fed state. BELVIQ can be administered with or without food.
BELVIQ XR
In an open label, randomized, crossover clinical trial, single dose and steady state pharmacokinetics of BELVIQ XR 20 mg administered once daily were compared with BELVIQ 10 mg tablet administered twice daily under fasted conditions in 34 healthy subjects. At steady state, the time to reach peak plasma concentrations of lorcaserin (tmax) following BELVIQ XR 20 mg once daily was approximately 10 hours compared with 1.5 hours for BELVIQ 10 mg tablet twice daily. A single dose administration of BELVIQ XR 20 mg resulted in comparable total plasma exposure (AUC0-∞), but approximately 25% lower peak exposures (Cmax) relative to two doses of BELVIQ tablets administered 12 hours apart. At steady state, however, both Cmax,ss and area under the plasma concentration versus time curve (AUC0-24,ss) of BELVIQ XR 20 mg administered once daily were bioequivalent to BELVIQ 10 mg tablets administered twice daily under fasted conditions.
Effect of Food. Intake of high fat, high calorie breakfast before a single 20 mg oral dose of BELVIQ XR resulted in approximately 46% increase in Cmax and 17% increase in AUC0-∞ but no change in tmax. At steady state, however, there was no significant food effect on the rate or extent of absorption of BELVIQ XR.
Distribution
Lorcaserin distributes to the cerebrospinal fluid and central nervous system in humans. Lorcaserin hydrochloride is moderately bound (~70%) to human plasma proteins.
Metabolism
Lorcaserin is extensively metabolized in the liver by multiple enzymatic pathways. After oral administration of lorcaserin the major circulating metabolite is lorcaserin sulfamate (M1), with a plasma Cmax that exceeds lorcaserin Cmax by 1- to 5-fold. N-carbamoyl glucuronide lorcaserin (M5) is the major metabolite in urine; M1 is a minor metabolite in urine, representing approximately 3% of dose. Other minor metabolites excreted in urine were identified as glucuronide or sulfate conjugates of oxidative metabolites. The principal metabolites exert no pharmacological activity at serotonin receptors.
Elimination
Lorcaserin is extensively metabolized by the liver and the metabolites are excreted in the urine. In a human mass balance study in which healthy subjects ingested radiolabeled lorcaserin, 94.5% of radiolabeled material was recovered, with 92.3% and 2.2% recovered from urine and feces, respectively. The terminal phase half-life for BELVIQ/ BELVIQ XR is approximately 11 to 12 hours.
Specific Populations
Renal Impairment. The pharmacokinetics of lorcaserin was studied in patients with varying degrees of renal function. Creatinine clearance (CLcr) was calculated by Cockcroft-Gault equation based on ideal body weight (IBW). Impaired renal function decreased Cmax of lorcaserin, with no change in AUC.
Exposure of lorcaserin sulfamate metabolite (M1) was increased in patients with impaired renal function by approximately 1.7-fold in mild (CLcr = 50-80 mL/min), 2.3-fold in moderate (CLcr = 30-50 mL/min) and 10.5-fold in severe renal impairment (CLcr = <30 mL/min) compared to normal subjects (CLcr >80 mL/min).
Exposure of the N-carbamoyl-glucuronide metabolite (M5) was increased in patients with impaired renal function by approximately 1.5-fold in mild (CLcr = 50-80 mL/min), 2.5-fold in moderate (CLcr = 30-50 mL/min) and 5.1-fold in severe renal impairment (CLcr = <30 mL/min) compared to normal subjects (CLcr >80 mL/min).
The terminal half-life of M1 is prolonged by 26%, 96%, and 508% in mild, moderate, and severe renal impairment, respectively. The terminal half-life of M5 is prolonged by 0%, 26%, and 22% in mild, moderate, and severe renal impairment, respectively. The metabolites M1 and M5 accumulate in patients with severely impaired renal function.
Approximately 18% of metabolite M5 in the body was cleared from the body during a standard 4-hour hemodialysis procedure. Lorcaserin and M1 were not cleared by hemodialysis. Lorcaserin is not recommended for patients with severe renal impairment (CLcr <30 mL/min) or patients with end stage renal disease [see Use in Specific Populations (8.6)].
Estimate Ideal Body Weight (IBW) in (kg)
Males: IBW = 50 kg + 2.3 kg for each inch over 5 feet.
Females: IBW = 45.5 kg + 2.3 kg for each inch over 5 feet.
The Cockcroft-Gault calculation using the IBW:
female:
GFR (mL/min) = 0.85 x (140-age) x ideal body weight (kg)
72 x serum creatinine (mg/dL)
male:
GFR (mL/min) = (140-age) x ideal body weight (kg)
72 x serum creatinine (mg/dL)
Hepatic Impairment. The pharmacokinetics of lorcaserin was evaluated in patients with hepatic impairment and subjects with normal hepatic function. Lorcaserin Cmax was 7.8% and 14.3% lower, in subjects with mild (Child-Pugh score 5-6) and moderate (Child-Pugh score 7-9) hepatic impairment, respectively, than that in subjects with normal hepatic function. The half-life of lorcaserin is prolonged by 59% to 19 hours in patients with moderate hepatic impairment. Lorcaserin exposure (AUC) is approximately 22% and 30% higher in patients with mild and moderate hepatic impairment, respectively. Dose adjustment is not required for patients with mild to moderate hepatic impairment. The effect of severe hepatic impairment on lorcaserin was not evaluated [see Use in Specific Populations (8.7)].
Gender. No dosage adjustment based on gender is necessary. Gender did not meaningfully affect the pharmacokinetics of lorcaserin.
Geriatric. No dosage adjustment is required based on age alone. In a clinical trial of 12 healthy elderly (age greater than 65 years) subjects and 12 matched adult patients, lorcaserin exposure (AUC and Cmax) was equivalent in the two groups. Cmax was approximately 18% lower in the elderly group, and Tmax was increased from 2 hours to 2.5 hours in the elderly group as compared to the non-elderly adult group.
Race. No dosage adjustment based on race is necessary. Race did not meaningfully affect the pharmacokinetics of lorcaserin.
Drug-Drug Interactions
Lorcaserin inhibits CYP 2D6-mediated metabolism. In a clinical trial in 21 CYP 2D6 extensive metabolizers, concomitant administration of lorcaserin (10 mg BID for 4 days) increased dextromethorphan peak concentrations (Cmax) by approximately 76% and exposure (AUC) by approximately 2-fold [see Drug Interactions (7.2)].
14. Clinical Studies
The safety and efficacy of BELVIQ for chronic weight management in conjunction with reduced caloric intake and increased physical activity were evaluated in 3 randomized, double-blind, placebo-controlled trials with durations ranging from 52 to 104 weeks. Two trials in adults without type 2 diabetes mellitus (Study 1 and Study 2) and one study in adults with type 2 diabetes mellitus (Study 3) evaluated the effect of BELVIQ. The primary efficacy parameter in these studies was weight loss at 1 year, which was assessed by percent of patients achieving greater than or equal to 5% weight loss, percent of patients achieving greater than or equal to 10% weight loss, and mean weight change. All patients received one-on-one instruction for a reduced-calorie diet and exercise counseling that began with the first dose of study medication and continued every four weeks throughout the trial.
Study 1 was a 2-year study that enrolled 3182 patients who were obese (BMI 30-45 kg/m2), or who were overweight (BMI 27-29.9 kg/m2) and had at least one weight-related comorbid condition such as hypertension or dyslipidemia. In Year 2, placebo patients were continued on placebo and BELVIQ, patients were re- randomized in a 2:1 ratio to continue BELVIQ or to switch to placebo. The mean age was 44 (range 18-65); 83.5% were women. Sixty-seven percent were Caucasian, 19% were African American and 12% were Hispanic. Mean baseline body weight was 100.0 kg and mean BMI was 36.2 kg/m2.
Study 2 was a 1-year study that enrolled 4008 patients who were obese (BMI 30-45 kg/m2) or were overweight (BMI 27-29.9 kg/m2) with at least one comorbid condition such as hypertension or dyslipidemia. The mean age was 44 (range 18-65); 80% were women. Sixty-seven percent were Caucasian, 20% were African American and 11% were Hispanic. Mean baseline body weight was 100.2 kg and mean BMI was 35.9 kg/m2.
Study 3 was a 1-year study that enrolled 604 adult patients with BMI greater than or equal to 27 kg/m2 and inadequately controlled type 2 diabetes (HbA1c range 7-10%) being treated with metformin and/or a sulfonylurea. Mean age was 53 (range 21-65); 54% were women. Sixty-one percent were Caucasian, 21% African American and 14% were Hispanic. Mean BMI was 36 kg/m2 and mean HbA1C was 8.1%.
A substantial percentage of randomized subjects withdrew from each study prior to week 52: 50% in Study 1, 45% in Study 2 and 36% in Study 3.
One-Year Weight Management in Patients without Diabetes Mellitus
Weight loss at 1 year in Studies 1 and 2 is presented in Table 6. The pooled data are reflective of the individual study results.
Statistically significantly greater weight loss was achieved with BELVIQ compared to placebo at week 52. The Year 1 placebo-adjusted weight loss achieved in patients treated with BELVIQ was 3.3 kg by ITT/LOCF analysis. The time course of weight loss with BELVIQ and placebo through week 52 is depicted in Figure 1.
Patients who did not lose at least 5% of baseline body weight by week 12 were unlikely to achieve at least 5% weight loss at week 52.
| BELVIQ
N=3098 | Placebo
N=3038 |
|
| Weight (kg) | ||
| Baseline mean (SD) | 100.4 (15.7) | 100.2 (15.9) |
| Change from baseline (adjusted mean1) (SE) | -5.8 (0.1) | -2.5 (0.1) |
| Difference from placebo (adjusted mean1) (95% CI) | -3.3** (-3.6, -2.9) | |
| Percent change from baseline (adjusted mean1) (SE) | -5.8 (0.1) | -2.5 (0.1) |
| Difference from placebo (adjusted mean1) (95% CI) | -3.3** ( -3.6, -3.0) | |
| % of Patients losing greater than or equal to 5% body weight | 47.1 | 22.6 |
| Difference from placebo (95% CI) | 24.5** (22.2, 26.8) | |
| % of Patients losing greater than or equal to 10% body weight | 22.4 | 8.7 |
| Difference from placebo (95% CI) | 13.8** (12.0, 15.5) |
SD=Standard Deviation; SE=Standard Error; CI=Confidence Interval
Intent to Treat Population using last observation carried forward method; All patients who received study medication and had a post-baseline body weight. Forty-four percent (44%) of patients in BELVIQ and 51% in placebo dropped out before the 52-week endpoint.
1Least squares means adjusted for baseline value, treatment, study and treatment by study interaction.
**p<0.001 compared to placebo. Type 1 error was controlled across the three endpoints.
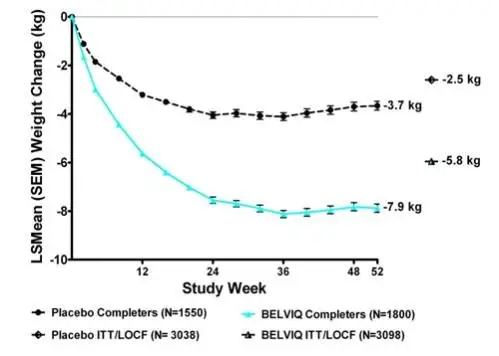
Figure 1. Longitudinal Weight Change (kg) in Completer Population: Studies 1 and 2
Two-Year Weight Management in Patients without Diabetes Mellitus
The safety and efficacy of BELVIQ for weight management during 2 years of treatment were evaluated in Study 1. Of the 3182 patients who were randomized in Year 1, 1553 (48.8%) were randomized in Year 2. Patients in all three Year 2 patient groups (BELVIQ Year 1/ BELVIQ Year 2, BELVIQ Year 1/placebo Year 2, and placebo Year 1/placebo Year 2) regained weight in Year 2 but remained below their Year 1 mean baseline weight (Figure 2).
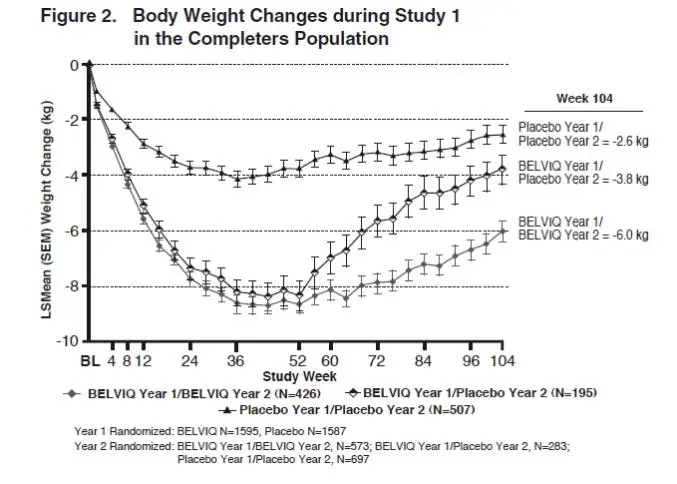
Effect of BELVIQ on Cardiometabolic Parameters and Anthropometry
Changes in lipids, fasting glucose, fasting insulin, waist circumference, heart rate, and blood pressure with BELVIQ are shown in Table 7.
In a substudy of 154 patients conducted as part of Study 2, DEXA analysis showed a 9.9% reduction in fat mass from a baseline of 44.5 kg in patients treated with BELVIQ compared to a 4.6% reduction from a baseline of 45.0 kg in patients treated with placebo. The placebo-adjusted reduction in fat mass achieved on BELVIQ was -5.3%. Reductions in lean body mass were 1.9% and 0.3% from baseline values of 48.0 kg and 51.0 kg, respectively, for BELVIQ- and placebo-treated patients.
| BELVIQ
N=3096 | Placebo
N=3039 | BELVIQ minus
Placebo (LSMean) |
|||
| Baseline mg/dL | % change from Baseline (LSMean1) | Baseline mg/dL | % change from Baseline (LSMean) | ||
| Total Cholesterol | 194.4 | -0.9 | 194.8 | 0.4 | -1.2* |
| LDL Cholesterol | 114.3 | 1.6 | 114.1 | 2.9 | -1.3* |
| HDL Cholesterol | 53.2 | 1.8 | 53.5 | 0.6 | 1.2* |
| Triglycerides | 135.4 | -5.3 | 137.0 | -0.5 | -4.8* |
| Baseline | change from Baseline (LSMean) | Baseline | change from Baseline (LSMean) | BELVIQ minus
Placebo (LSMean) |
|
| Systolic blood pressure (mmHg) | 121.4 | -1.8 | 121.5 | -1.0 | -0.7* |
| Diastolic blood pressure (mmHg) | 77.4 | -1.6 | 77.7 | -1.0 | -0.6* |
| Heart Rate (bpm) | 69.5 | -1.2 | 69.5 | -0.4 | -0.8 |
| Fasting glucose (mg/dL) | 92.1 | -0.2 | 92.4 | 0.6 | -0.8 |
| Fasting insulin2 (µIU/mL) | 15.9 | -3.3 | 15.8 | -1.3 | -2.1* |
| Waist Circumference (cm) | 109.3 | -6.5 | 109.6 | -4.0 | -2.5 |
1 Least squares means adjusted for baseline value, treatment, study and treatment by study interaction
2 Measured in Study 1 only (n=1538)
* Statistically significant versus placebo based on the pre-specified gatekeeping method for controlling Type I error in key secondary endpoints.
One-Year Weight Management in Patients with Type 2 Diabetes Mellitus
Weight loss among patients with type 2 diabetes mellitus who were treated with BELVIQ was statistically significantly greater than that among patients treated with placebo (Table 8).
| BELVIQ
N=251 | Placebo
N=248 |
|
| Weight loss (kg)
Baseline mean (SD) Change from baseline (adjusted mean1) (SE) Difference from placebo (adjusted mean1) (95% CI) | 103.5 (17.2) -4.7 (0.4) -3.1** (-4.0, -2.2) | 102.3 (18.0) -1.6 (0.4) |
| Percent change from baseline (adjusted mean1) (SE) Difference from placebo (adjusted mean1) (95% CI) | -4.5 (0.4) -3.1** (-3.9, -2.2) | -1.5 (0.4) |
| % of Patients losing greater than or equal to 5% body weight
Difference from placebo (95% CI) | 37.5 21.3** (13.8, 28.9) | 16.1 |
| % of Patients losing greater than or equal to 10% body weight
Difference from placebo (95% CI) | 16.3 11.9** (6.7, 17.1) | 4.4 |
SD=Standard Deviation; SE=Standard Error; CI=Confidence Interval
Intent to Treat Population using last observation carried forward method; All patients who received study medication and had a post-baseline body weight. Thirty-four percent (34%) of patients in Belviq and 38% in placebo dropped out before the 52-week endpoint.
1Least squares means adjusted for baseline value, baseline HbA1c stratum and prior antihyperglycemic medication stratum.
**p<0.001 compared to placebo. Type 1 error was controlled across the three endpoints.
Effect of BELVIQ on Cardiometabolic Parameters and Anthropometry in Patients with Type 2
Diabetes Mellitus
Patients in Study 3 were taking either metformin and/or a sulfonylurea at study start, and had inadequate glycemic control (HbA1c range 7-10%). Changes in HbA1c and fasting glucose with BELVIQ use are shown in Table 9.
| BELVIQ
N=256 | Placebo
N=252 | BELVIQ minus
Placebo (LSMean) |
|||
| Baseline | Change from
Baseline (LSMean1) | Baseline | Change from Baseline (LSMean) | ||
| HbA1C (%) | 8.1 | -0.9 | 8.0 | -0.4 | -0.5* |
| Fasting glucose (mg/dL) | 163.3 | -27.4 | 160.0 | -11.9 | -15.5* |
| Systolic blood pressure (mmHg) | 126.6 | -0.8 | 126.5 | -0.9 | 0.1 |
| Diastolic blood pressure (mmHg) | 77.9 | -1.1 | 78.7 | -0.7 | -0.4 |
| Heart Rate (bpm) | 72.3 | -2.0 | 72.7 | -0.4 | -1.6 |
| Waist Circumference (cm) | 115.8 | -5.5 | 113.5 | -3.3 | -2.2 |
| Baseline | % Change
from Baseline (LSMean) | Baseline | % Change
from Baseline (LSMean) | BELVIQ minus
Placebo (LSMean) |
|
| Total Cholesterol (mg/dL) | 173.5 | -0.7 | 172.0 | -0.1 | -0.5 |
| LDL Cholesterol (mg/dL) | 95.0 | 4.2 | 94.6 | 5.0 | -0.8 |
| HDL Cholesterol (mg/dL) | 45.3 | 5.2 | 45.7 | 1.6 | 3.6 |
| Triglycerides (mg/dL) | 172.1 | -10.7 | 163.5 | -4.8 | -5.9 |
Intent to Treat Population using last observation carried forward method; All patients who received study medication and
had a post-baseline measurement.
* Statistically significant versus placebo based on the pre-specified gatekeeping method for controlling Type I error in key secondary endpoints.
1Least squares means adjusted for baseline value, baseline HbA1c stratum and prior antihyperglycemic medication stratum.
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
- Inform patients that BELVIQ/BELVIQ XR is indicated for chronic weight management only in conjunction with a reduced-calorie diet and increased physical activity.
- Caution patients not to increase their dose of BELVIQ/BELVIQ XR.
- Instruct patients to discontinue use of BELVIQ/BELVIQ XR if they have not achieved 5% weight loss by 12 weeks of treatment.
- Instruct patients to tell their healthcare provider about all the medications, nutritional supplements and vitamins (including any weight loss products) that they may take while taking BELVIQ/BELVIQ XR.
- Inform patients of the possibility of serotonin syndrome or Neuroleptic Malignant Syndrome (NMS)-like reactions with the combined use of BELVIQ/BELVIQ XR with other serotonergic drugs, including selective serotonin-norepinephrine reuptake inhibitors (SNRIs) and selective serotonin reuptake inhibitors (SSRIs), triptans, drugs that impair metabolism of serotonin (including monoamine oxidase inhibitors [MAOIs]), dietary supplements such as St. John’s Wort and tryptophan, tramadol, or antipsychotics or other dopamine antagonists.
- Inform patients who develop signs or symptoms of valvular heart disease, including dyspnea or dependent edema to seek medical attention.
- Caution patients about operating hazardous machinery, including automobiles, until they are reasonably certain that BELVIQ/BELVIQ XR therapy does not affect them adversely.
- Instruct patients to seek medical attention in the event of emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
- Instruct men who have an erection lasting greater than 4 hours, whether painful or not, to immediately discontinue the drug and seek emergency medical attention.
- Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to inform their healthcare provider with a known or suspected pregnancy [see Contraindications (4), Use in Specific Populations (8.1)].
- Advise women to avoid use of BELVIQ/BELVIQ XR while breastfeeding [see Use in Specific Populations (8.2)].
Distributed by Eisai Inc., Woodcliff Lake, NJ 07677
© 2018
| BELVIQ
lorcaserin hydrochloride hemihydrate tablet |
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
|
||||||||||||||||||||||||
| BELVIQ XR
lorcaserin hydrochloride hemihydrate tablet, film coated, extended release |
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
| Labeler - Eisai Inc. (831600833) |