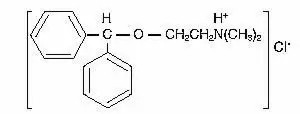
Drug Detail:Benadryl (Diphenhydramine [ dye-fen-hye-dra-meen ])
Drug Class: Anticholinergic antiemetics Anticholinergic antiparkinson agents Antihistamines Miscellaneous anxiolytics, sedatives and hypnotics
Related/similar drugs
acetaminophen, prednisone, ibuprofen, hydroxyzine, ondansetron, lorazepam, fluticasone nasalContraindications
Warnings
Antihistamines should be used with considerable caution in patients with narrow-angle glaucoma, stenosing peptic ulcer, pyloroduodenal obstruction, symptomatic prostatic hypertrophy, or bladder-neck obstruction.
Local necrosis has been associated with the use of subcutaneous or intradermal use of intravenous Benadryl.
Overdosage
Antihistamine overdosage reactions may vary from central nervous system depression to stimulation. Stimulation is particularly likely in pediatric patients. Atropine-like signs and symptoms; dry mouth; fixed, dilated pupils; flushing; and gastrointestinal symptoms may also occur.
Stimulants should not be used.
Vasopressors may be used to treat hypotension.
Benadryl Injection Dosage and Administration
THIS PRODUCT IS FOR INTRAVENOUS OR INTRAMUSCULAR ADMINISTRATION ONLY.
Benadryl in the injectable form is indicated when the oral form is impractical.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
DOSAGE SHOULD BE INDIVIDUALIZED ACCORDING TO THE NEEDS AND THE RESPONSE OF THE PATIENT.
| BENADRYL
diphenhydramine hydrochloride injection, solution |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| BENADRYL
diphenhydramine hydrochloride injection, solution |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Parke-Davis |