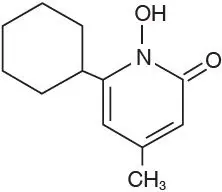
Adverse Reactions/Side Effects
In the vehicle-controlled clinical trials conducted in the US, 9% (30/327) of patients treated with Ciclopirox Topical Solution, 8%, (Nail Lacquer), and 7% (23/328) of patients treated with vehicle reported treatment-emergent adverse events (TEAE) considered by the investigator to be causally related to the test material.
The incidence of these adverse events, within each body system, was similar between the treatment groups except for Skin and Appendages: 8% (27/327) and 4% (14/328) of subjects in the ciclopirox and vehicle groups reported at least one adverse event, respectively. The most common were rash-related adverse events: periungual erythema and erythema of the proximal nail fold were reported more frequently in patients treated with Ciclopirox Topical Solution, 8%, (Nail Lacquer), (5% [16/327]) than in patients treated with vehicle (1% [3/328]). Other TEAEs thought to be causally related included nail disorders such as shape change, irritation, ingrown toenail, and discoloration.
The incidence of nail disorders was similar between the treatment groups (2% [6/327] in the Ciclopirox Topical Solution, 8%, (Nail Lacquer), group and 2% [7/328] in the vehicle group). Moreover, application site reactions and/or burning of the skin occurred in 1% of patients treated with Ciclopirox Topical Solution, 8%, (Nail Lacquer), (3/327) and vehicle (4/328).
A 21-Day Cumulative Irritancy study was conducted under conditions of semi-occlusion. Mild reactions were seen in 46% of patients with the Ciclopirox Topical Solution, 8%, (Nail Lacquer), 32% with the vehicle and 2% with the negative control, but all were reactions of mild transient erythema. There was no evidence of allergic contact sensitization for either the Ciclopirox Topical Solution, 8%, (Nail Lacquer), or the vehicle base. In a separate study of the photosensitization potential of Ciclopirox Topical Solution, 8%, (Nail Lacquer), in a maximized test design that included the occluded application of sodium lauryl sulfate, no photoallergic reactions were noted. In four subjects localized allergic contact reactions were observed. In the vehicle-controlled studies, one patient treated with Ciclopirox Topical Solution, 8%, (Nail Lacquer), discontinued treatment due to a rash, localized to the palm (causal relation to test material undetermined).
Use of Ciclopirox Topical Solution, 8%, (Nail Lacquer), for 48 additional weeks was evaluated in an open-label extension study conducted in patients previously treated in the vehicle-controlled studies. Three percent (9/281) of subjects treated with Ciclopirox Topical Solution, 8%, (Nail Lacquer), experienced at least one TEAE that the investigator thought was causally related to the test material. Mild rash in the form of periungual erythema (1% [2/281]) and nail disorders (1% [4/281]) were the most frequently reported. Four patients discontinued because of TEAEs. Two of the four had events considered to be related to test material: one patient's great toenail "broke away" and another had an elevated creatine phosphokinase level on Day 1 (after 48 weeks of treatment with vehicle in the previous vehicle-controlled study).
To report SUSPECTED ADVERSE REACTIONS, contact Cosette Pharmaceuticals, Inc. at 1-800-922-1038 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.