Drug Detail:Ciprofloxacin ophthalmic (Ciprofloxacin ophthalmic [ sip-roe-flox-a-sin-off-thal-mik ])
Drug Class: Ophthalmic anti-infectives
Ciprofloxacin Ophthalmic Solution Description
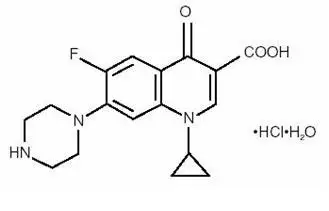
Each mL of Ciprofloxacin ophthalmic solution 0.3% contains: Active: ciprofloxacin HCl 3.5 mg equivalent to 3 mg base. Preservative: benzalkonium chloride 0.006%. Inactives: sodium acetate, acetic acid, mannitol 4.6%, edetate disodium 0.05%, hydrochloric acid and/or sodium hydroxide (to adjust pH) and water for injection. The pH is approximately 4.5 and the osmolality is approximately 300 mOsm.
Ciprofloxacin Ophthalmic Solution - Clinical Pharmacology
Microbiology: Ciprofloxacin has in vitro activity against a wide range of gram-negative and gram-positive organisms. The bactericidal action of ciprofloxacin results from interference with the enzyme DNA gyrase which is needed for the synthesis of bacterial DNA.
Aerobic gram-Positive microorganisms:
Staphylococcus aureus (methicillin-susceptible strains)
Staphylococcus epidermidis (methicillin-susceptible strains)
Streptococcus pneumoniae
Streptococcus (Viridans Group)
Aerobic gram-negative microorganisms:
Haemophilus influenzae
Pseudomonas aeruginosa
Serratia marcescens
Ciprofloxacin has been shown to be active in vitro against most strains of the following organisms, however, the clinical significance of these data is unknown:
Gram-Positive:
Enterococcus faecalis (Many strains are only moderately susceptible)
Staphylococcus haemolyticus
Staphylococcus hominis
Staphylococcus saprophyticus
Streptococcus pyogenes
Gram-Negative
Acinetobacter calcoaceticus subsp. anitratus
Aeromonas caviae
Aeromonas hydrophila
Brucella melitensis
Campylobacter coli
Campylobacter jejuni
Citrobacter diversus
Citrobacter freundii
Edwardsiella tarda
Enterobacter aerogenes
Enterobacter cloacae
Escherichia coli
Haemophilus ducreyi
Haemophilus parainfluenzae
Kiebsiella pneumoniae
Klebsiella oxytoca
Legionella pneumophila
Moraxella (Branhamella) catarrhalis
Morganella morganii
Neisseria gonorrhoeae
Neisseria meningitides
Pasteurella multocida
Proteus mirabilis
Proteus vulgaris
Providencia rettgeri
Providencia stuartii
Salmonella enteritidis
Salmonella typhi
Shigella sonneii
Shigella flexneri
Vibrio cholera
Vibrio parahaemolyticus
Vibrio vulnificus
Yersinia enterocolitica
Chlamydia trachomatis (only moderately susceptible) and Mycobacterium tuberculosis (only moderately susceptible).
Most strains of Pseudomonas cepacia and some strains of Pseudomonas maltophilia are resistant to ciprofloxacin as are most anaerobic bacteria, including Bacteroides fragilis and Clostridium difficile.
The minimal bactericidal concentration (MBC) generally does not exceed the minimal inhibitory concentration (MIC) by more than a factor of 2. Resistance to ciprofloxacin in vitro usually develops slowly (multiple-step mutation).
Ciprofloxacin does not cross-react with other antimicrobial agents such as beta-lactams or aminoglycosides; therefore, organisms resistant to these drugs may be susceptible to ciprofloxacin.
Indications and Usage for Ciprofloxacin Ophthalmic Solution
Corneal Ulcers:
Pseudomonas aeruginosa
Serratia marcescens *
Staphylococcus aureus
Staphylococcus epidermidis
Streptococcus pneumoniae
Streptococcus (Viridans Group) *
Conjunctivitis:
Haemophilus influenzae
Staphylococcus aureus
Staphylococcus epidermidis
Streptococcus pneumoniae
*Efficacy for this organism was studied in fewer than 10 infections.
Warnings and Precautions
NOT FOR INJECTION INTO THE EYE.
Serious and occasionally fatal hypersensitivity (anaphylactic) reactions, some following the first dose, have been reported in patients receiving systemic quinolone therapy. Some reactions were accompanied by cardiovascular collapse, loss of consciousness, tingling, pharyngeal or facial edema, dyspnea, urticaria, and itching. Only a few patients had a history of hypersensitivity reactions. Serious anaphylactic reactions require immediate emergency treatment with epinephrine and other resuscitation measures, including oxygen, intravenous fluids, intravenous antihistamines, corticosteroids, pressor amines and airway management, as clinically indicated.
Remove contact lenses before using.
Precautions
General:
Ciprofloxacin should be discontinued at the first appearance of a skin rash or any other sign of hypersensitivity reaction.
In clinical studies of patients with bacterial corneal ulcer, a white crystalline precipitate located in the superficial portion of the corneal defect was observed in 35 (16.6%) of 210 patients. The onset of the precipitate was within 24 hours to 7 days after starting therapy. In one patient, the precipitate was immediately irrigated out upon its appearance. In 17 patients, resolution of the precipitate was seen in 1 to 8 days (seven within the first 24 to 72 hours), in five patients, resolution was noted in 10 to 13 days. In nine patients, exact resolution days were unavailable; however, at follow-up examinations, 18 to 44 days after onset of the event, complete resolution of the precipitate was noted. In three patients, outcome information was unavailable. The precipitate did not preclude continued use of ciprofloxacin, nor did it adversely affect the clinical course of the ulcer or visual outcome. (see ADVERSE REACTIONS).
Information for patients:
Do not touch dropper tip to any surface, as this may contaminate the solution.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
- Salmonella /Microsome Test (Negative)
- E. coli DNA Repair Assay (Negative)
- Mouse Lymphoma Cell Forward Mutation Assay (Positive)
- Chinese Hamster V 79Cell HGPRT Test (Negative)
- Syrian Hamster Embryo Cell Transformation Assay (Negative)
- Saccharomyces cerevisiae Point Mutation Assay (Negative)
- Saccharomyces cerevisiae Mitotic Crossover and Gene Conversion Assay (Negative)
- Rat Hepatocyte DNA Repair Assay (Positive)
- Rat Hepatocyte DNA Repair Assay
- Micronucleus Test (Mice)
- Dominant Lethal Test (Mice)
Adverse Reactions/Side Effects
To report SUSPECTED ADVERSE REACTIONS, contact LEADING PHARMA AT 1-844-740-7500 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Ciprofloxacin Ophthalmic Solution Dosage and Administration
The recommended dosage regimen for the treatment of corneal ulcers is two drops into the affected eye every 15 minutes for the first six hours and then two drops into the affected eye every 30 minutes for the remainder of the first day. On the second day, instill two drops in the affected eye hourly. On the third through the fourteenth day, place two drops in the affected eye every four hours. Treatment may be continued after 14 days if corneal re-epithelialisation has not occurred.
Bacterial Conjunctivitis:
The recommended dosage regimen for the treatment of bacterial conjunctivitis is one or two drops instilled into the conjunctival sac(s) every two hours while awake for two days and one or two drops every four hours while awake for the next five days.
How is Ciprofloxacin Ophthalmic Solution supplied
2.5 mL - NDC 69315-308-02
5 mL – NDC 69315-308-05
10 mL – NDC 69315-308-10
STORAGE:
Store at 25°C (77°F); excursions permitted 15°C to 30°C (59°F to 86°F) [See USP controlled room temperature]. Retain in carton until contents are used and protect from light.
ANIMAL PHARMACOLOGY:
Ciprofloxacin and related drugs have been shown to cause arthropathy in immature animals of most species tested following oral administration. However, a one month topical ocular study using immature Beagle dogs did not demonstrate any articular lesions.
CLINICAL STUDIES:
Following therapy with Ciprofloxacin ophthalmic solution 0.3%, 76% of the patients with corneal ulcers and positive bacterial cultures were clinically cured and complete re-epithelialisation occurred in about 92% of the ulcers.
In 3 and 7 day multicenter clinical trials, 52% of the patients with conjunctivitis and positive conjunctival cultures were clinically cured and 70-80% had all causative pathogens eradicated by the end of treatment.
In a randomized, double-masked, multicenter, parallel-group clinical trial of pediatric patients with bacterial conjunctivitis, between birth and 31 days of age, patients were dosed with Ciprofloxacin ophthalmic solution or another anti-infective agent. Clinical outcomes for the trial demonstrated a clinical cure rate of 80% at Day 9 and a microbiological eradication success rate of 85% at Day 9.
Please note that microbiologic eradication does not always correlate with clinical outcome in anti-infective trials.
Rx Only
Distributed by:
Leading Pharma, LLC.
Fairfield, NJ07004
Manufactured By:
FDC Limited,
B-8, MIDC Industrial Area,
Waluj, Aurangabad - 431 136,
Maharashtra, India
Revision: 10/2019
STERILE OPHTHALMIC SOLUTION
CIPROFLOXACIN OPHTHALMIC SOLUTION USP 0.3%
INSTRUCTIONS FOR USE
Read this Instruction for Use before you start using Ciprofloxacin ophthalmic solution and each time you get a refill. There may be new information. This information does not take the place of talking to your doctor about your medical condition or your treatment.
Use Ciprofloxacin ophthalmic solution as prescribed by your doctor. Ask your doctor if you have questions about how to use Ciprofloxacin ophthalmic solution.
Important information:
- Ciprofloxacin ophthalmic solution is for use in the eye only. Ciprofloxacin ophthalmic solution should not be injected into the eye.
- If you use other medicines in your eye, wait at least 10 minutes between using Ciprofloxacin ophthalmic solution and your other eye medicines.
- If you wear contact lenses, remove them before using Ciprofloxacin ophthalmic solution. Wait at least 15 minutes after using Ciprofloxacin ophthalmic solution before placing your contact lenses back in your eyes.
- Do not touch your eye, fingers, or other surfaces with the tip of the Ciprofloxacin ophthalmic solution bottle. You may get bacteria on the tip of the bottle that can cause you to get an eye infection. This eye infection can lead to serious eye damage or vision loss. If you think you have gotten bacteria on the tip of the bottle or you have an eye infection, call your doctor right away.
- Wash your hands before each use.

Before you use a bottle of Ciprofloxacin ophthalmic solution for the first time:
- Check the expiration date on the bottle before use. Do not use Ciprofloxacin ophthalmic solution if the expiration date has passed.
- Check that the seal on the dust cover is not broken or missing. Do not use the medicine if the seal is broken or missing. Call your doctor or pharmacist.
Each time you use Ciprofloxacin ophthalmic solution:
Step 1: Turn the dust cover clockwise to break the seal.
Step 2: Remove the dust cover and throw it away.
Step 3: Turn the tan coloured cap anti-clockwise and remove. Place the cap on a clean flat surface.
Step 4: Tilt your head backwards.

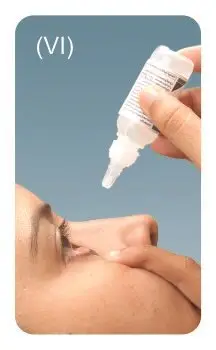
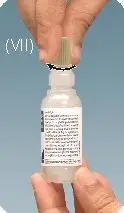
Step 6: Replace the tan coloured cap back on the bottle and turn it clockwise to close.
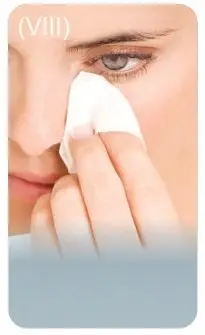
Step 7: Remove any extra solution from skin around the eyes with a tissue.
- If your doctor has told you to use drops in both eyes, repeat Step 5 for your other eye.
- The Ciprofloxacin ophthalmic solution bottle tip is made to give a certain amount of medicine in 1 drop. Do not make the opening of the bottle tip bigger or you may get too much Ciprofloxacin ophthalmic solution.
- After you use all of the doses of Ciprofloxacin ophthalmic solution that your doctor has prescribed, there will be some medicine left in the bottle. Do not use the extra medicine in the bottle. Throw the bottle away.
How should I store Ciprofloxacin ophthalmic solution?
- Store at 25°C (77°F); excursions permitted 15°C to 30°C (59°F to 86°F) [See USP controlled room temperature].
- Retain in carton until contents are used and protect from light
Keep Ciprofloxacin ophthalmic solution and all medicines out of the reach of children.
This Instruction for Use has been approved by the U.S. Food and Drug Administration.
Distributed by:
Leading Pharma, LLC.
Fairfield, NJ07004
Manufactured By:
FDC Limited,
B-8, MIDC Industrial Area,
Waluj, Aurangabad - 431 136,
Maharashtra, India
Revision: 10/2019
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC 69315-308-05
Ciprofloxacin Ophthalmic Solution USP 0.3% as base
Rx Only
5 mL Sterile
PRECAUTION: Do not touch dropper tip to any surface, as this may contaminate the solution.
USUAL DOSAGE: Read enclosed insert.
FOR TOPICAL OPHTHALMIC USE ONLY.
EACH mL CONTAINS:
Active: ciprofloxacin HCl 3.5 mg equivalent to 3 mg ciprofloxacin base.
Preservative: benzalkonium chloride 0.006%.
Inactives: sodium acetate, acetic acid, mannitol, edetate disodium, hydrochloric acid and/or sodium hydroxide (to adjust pH) and water for injection.
STORAGE: Store at 25°C (77°F); excursions permitted 15°C to 30°C (59°F to 86°F) [See USP controlled room temperature].Protect from light.

| CIPROFLOXACIN
ciprofloxacin solution |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Leading Pharma, LLC (079575060) |
| Registrant - FDC Limited (650078413) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| FDC Limited | 862267994 | analysis(69315-308) , manufacture(69315-308) | |




