Drug Detail:Cystaran eye drops (Cysteamine ophthalmic [ sis-tee-a-meen-off-thal-mik ])
Drug Class: Miscellaneous ophthalmic agents
Highlights of Prescribing Information
CYSTARAN (cysteamine ophthalmic solution) 0.44%, for topical ophthalmic use
Initial U.S. Approval: 1994
Indications and Usage for Cystaran
CYSTARAN is a cystine-depleting agent indicated for the treatment of corneal cystine crystal accumulation in patients with cystinosis. (1)
Cystaran Dosage and Administration
Instill one drop of CYSTARAN in each eye, every waking hour. (2)
Dosage Forms and Strengths
Ophthalmic solution containing 6.5 mg/mL of cysteamine hydrochloride equivalent to 4.4 mg/mL of cysteamine (0.44%). (3)
Contraindications
None. (4)
Warnings and Precautions
To minimize the risk of contamination, do not touch the dropper tip to any surface. Keep bottle tightly closed when not in use. (5.1)
Adverse Reactions/Side Effects
The most common adverse reactions (incidence approximately 10% or greater) are sensitivity to light, redness, eye pain/irritation, headache and visual field defects. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Leadiant Biosciences, Inc. at 1-888-393-4584 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 2/2022
Full Prescribing Information
1. Indications and Usage for Cystaran
CYSTARAN is a cystine-depleting agent indicated for the treatment of corneal cystine crystal accumulation in patients with cystinosis.
2. Cystaran Dosage and Administration
Instill one drop of CYSTARAN in each eye, every waking hour.
Do not touch dropper tip to any surface, as this may contaminate the solution.
Discard after 1 week of use.
3. Dosage Forms and Strengths
Ophthalmic solution containing 6.5 mg/mL of cysteamine hydrochloride equivalent to 4.4 mg/mL of cysteamine (0.44%).
5. Warnings and Precautions
5.1 Contamination of Tip and Solution
To minimize contaminating the dropper tip and solution, care should be taken not to touch the eyelids or surrounding areas with the dropper tip of the bottle. Keep bottle tightly closed when not in use.
5.2 Benign Intracranial Hypertension
There have been reports of benign intracranial hypertension (or pseudotumor cerebri) associated with oral cysteamine treatment that has resolved with the addition of diuretic therapy.
There have also been reports associated with ophthalmic use of cysteamine; however, all of these patients were on concurrent oral cysteamine.
6. Adverse Reactions/Side Effects
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety data described below reflect exposure in controlled clinical trials of six months to 19 years duration in approximately 300 patients.
The most frequently reported ocular adverse reactions occurring in ≥10% of patients were sensitivity to light, redness, and eye pain/irritation, headache and visual field defects.
8. Use In Specific Populations
8.4 Pediatric Use
The safety and effectiveness of CYSTARAN (cysteamine ophthalmic solution) 0.44% have been established in pediatric patients.
8.5 Geriatric Use
When the clinical studies with CYSTARAN were conducted, the reduced life expectancy from cystinosis did not make it possible to include patients in the geriatric age range.
8.6 Renal Impairment
The effect of renal impairment on the pharmacokinetics of cysteamine following ophthalmic administration of cysteamine ophthalmic solution has not been evaluated because ophthalmic exposure compared to systemic exposure is negligible. The majority of the patients in the ophthalmic clinical studies are assumed to have had some degree of renal impairment due to their underlying systemic disease. The total daily ophthalmic dose is less than 2% of the recommended oral daily dose of cysteamine; thus, the systemic exposure following ophthalmic administration is expected to be negligible compared to oral administration.
11. Cystaran Description
CYSTARAN is a sterile ophthalmic solution containing 6.5 mg/mL of cysteamine hydrochloride, equivalent to 4.4 mg/mL of cysteamine (0.44%) as the active ingredient. Cysteamine is a cystine-depleting agent which lowers the cystine content of cells in patients with cystinosis.
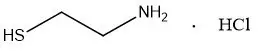
Molecular Formula: C2H7NS HCl
Molecular Weight: 113.61
Each milliliter of CYSTARAN contains: Active: cysteamine 4.4 mg (equivalent to cysteamine hydrochloride 6.5 mg); Preservative: benzalkonium chloride 0.1 mg; Inactive Ingredients: sodium chloride, hydrochloric acid and/or sodium hydroxide (to adjust pH to 4.1-4.5), and purified water.
12. Cystaran - Clinical Pharmacology
14. Clinical Studies
Clinical efficacy was evaluated in controlled clinical trials in approximately 300 patients. The primary efficacy end point was the response rate of eyes that had a reduction of at least 1 unit in the photo-rated Corneal Cystine Crystal Score (CCCS) at some time point during the study when baseline CCCS ≥1, or a lack of an increase of more than 1 unit in CCCS throughout the study when baseline CCCS <1.
Study 1 combined the data from three smaller studies. For eyes with a lower baseline of CCCS <1, the response rate was 13% (4/30) [95% CI: (4, 32)]. For eyes with a higher baseline of CCCS ≥1, the response rate was 32% (94/291) [95% CI: (27, 38)].
Study 2 evaluated ocular cystinosis patients who had a baseline of CCCS ≥1. The response rate was 67% (10/15) [95% CI: (38, 88)].
Study 3 also evaluated ocular cystinosis patients; for eyes with a baseline of CCCS ≥1, the response rate was 33% (3/9) [95% CI: (8, 70)].
Corneal crystals accumulate if CYSTARAN is discontinued.
16. How is Cystaran supplied
Fifteen (15) mL of CYSTARAN (cysteamine ophthalmic solution) 0.44% is supplied in the following bottles:
- an opaque, white, low-density polyethylene (LDPE) 70 mm tall bottle with a 15 mm white, LDPE controlled dropper tip and closed with a white, polypropylene screw cap. The bottle is stored in a carton. NDC 54482-020-01
- an opaque, white, LDPE 88 mm tall bottle with a 1.6 mm blue, silicone rubber flow-controlled dropper tip and closed with a white, high-density polyethylene (HDPE) screw cap. The bottle is foil-wrapped and stored in a carton. NDC 54482-020-02
Principal Display Panel - Carton
NDC 54482-020-01
cystaran®
(cysteamine ophthalmic
solution) 0.44%
For Ophthalmic Use Only
Sterile
15 mL
Rx only
Active Ingredient: Each mL
contains 6.5mg cysteamine
hydrochloride, equivalent to 4.4mg
cysteamine; Inactive Ingredients:
Benzalkonium chloride
(preservative); Sodium chloride;
hydrochloric acid and/or sodium
hydroxide (to adjust pH) and
purified water.
Dosage: See package insert for full
prescribing information.
Storage: Store in freezer at -25°C
to -15°C (-13°F to 5°F). Thaw for
approximately 24 hours before
use. Store thawed bottle at 2°C to
25°C (36°F to 77°F) for up to 1
week. Do not refreeze. Discard
after 1 week of use, even if there
is remaining drug product.
Avoid touching dropper tip to any
surface.
For Ophthalmic Use Only.
cysc-11-ht 0717
Leadiant
Biosciences
Manufactured by Hi-Tech
Pharmacal Co. Inc.,
Amityville, NY 11701 for
Leadiant Biosciences, Inc.,
Gaithersburg, MD 20878
Rev.915:04 07/17
| CYSTARAN
cysteamine hydrochloride solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Leadiant Biosciences, Inc. (068301431) |







