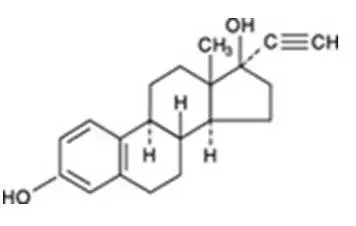
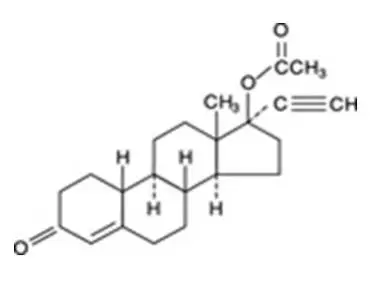
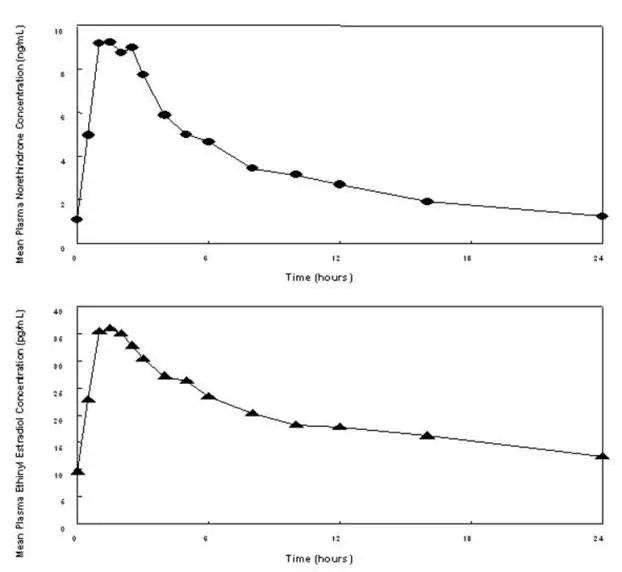
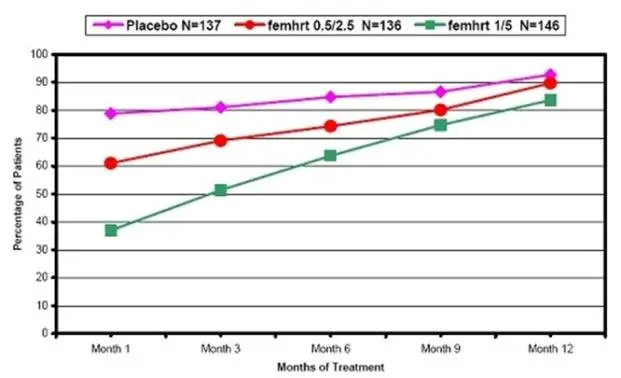
Drug Detail:Femhrt (Ethinyl estradiol and norethindrone (hrt) [ eth-in-ill-ess-tra-dye-ole-and-nor-eth-in-drone ])
Drug Class: Contraceptives Sex hormone combinations
Highlights of Prescribing Information
femhrt® (norethindrone acetate/ethinyl estradiol tablets)
Initial U.S. Approval: 1968
WARNING: CARDIOVASCULAR DISORDERS, BREAST CANCER, ENDOMETRIAL CANCER AND PROBABLE DEMENTIA
See full prescribing information for complete boxed warning.
Estrogen Plus Progestin Therapy
• Estrogen plus progestin therapy should not be used for the prevention of cardiovascular disease or dementia (5.1, 5.3)
• The Women’s Health Initiative (WHI) estrogen plus progestin substudy reported increased risks of stroke, deep vein thrombosis (DVT), pulmonary embolism (PE), and myocardial infarction (MI) (5.1)
• The WHI estrogen plus progestin substudy reported an increased risk of invasive breast cancer (5.2)
• The WHI Memory Study (WHIMS) estrogen plus progestin ancillary study of WHI reported an increased risk of probable dementia in postmenopausal women 65 years of age and older (5.3)
Estrogen-Alone Therapy
• There is an increased risk of endometrial cancer in a woman with a uterus who uses unopposed estrogens (5.2)
• Estrogen-alone therapy should not be used for the prevention of cardiovascular disease or dementia (5.1, 5.3)
• The WHI estrogen-alone substudy reported increased risks of stroke and DVT (5.1)
• The WHIMS estrogen-alone ancillary study of WHI reported an increased risk of probable dementia in postmenopausal women 65 years of age and older (5.3)
Recent Major Changes
Warnings and Precautions (5.2) 11/2017
Indications and Usage for Femhrt
femhrt is an estrogen plus progestin indicated in a woman with a uterus for:
- Treatment of Moderate to Severe Vasomotor Symptoms due to Menopause (1.1)
- Prevention of Postmenopausal Osteoporosis (1.2)
Femhrt Dosage and Administration
- One tablet taken orally once daily (2.1, 2.2)
Dosage Forms and Strengths
- Oval white tablet contains 0.5 mg norethindrone acetate and 2.5 mcg ethinyl estradiol (3)
- D-shaped white tablet contains 1 mg norethindrone acetate and 5 mcg ethinyl estradiol (3)
Contraindications
- Undiagnosed abnormal genital bleeding (4)
- Known, suspected, or history of breast cancer (4, 5.2)
- Known or suspected estrogen-dependent neoplasia (4, 5.2)
- Active DVT, PE, or history of these conditions (4, 5.1)
- Active arterial thromboembolic disease (for example, stroke and MI), or a history of these conditions (4, 5.1)
- Known anaphylactic reaction or angioedema to femhrt (4)
- Known liver impairment or disease (4, 5.10)
- Known protein C, protein S, or antithrombin deficiency, or other known thrombophilic disorders (4)
- Known or suspected pregnancy (4, 8.1)
Warnings and Precautions
- Estrogens increase the risk of gallbladder disease (5.4)
- Discontinue estrogen if severe hypercalcemia, loss of vision, severe hypertriglyceridemia or cholestatic jaundice occurs (5.5, 5.6, 5.9, 5.10)
- Monitor thyroid function in women on thyroid replacement therapy (5.11, 5.18)
Adverse Reactions/Side Effects
Most common adverse reactions (incidence greater than or equal to 5 percent) are headache, abdominal pain, breast pain, and edema (generalized). (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Allergan at 1-800-678-1605 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
To report SUSPECTED ADVERSE REACTIONS, contact Allergan at 1-800-678-1605 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Drug Interactions
- Inducers and/or inhibitors of CYP3A4 may affect estrogen drug metabolism (7.1)
Use In Specific Populations
- Nursing Mothers: Estrogen administration to nursing women has been shown to decrease the quantity and quality of breast milk (8.3)
- Geriatric Use: An increased risk of probable dementia in women over 65 years of age was reported in the Women’s Health Initiative Memory ancillary studies of the Women’s Health Initiative (5.3, 8.5)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 11/2017
Related/similar drugs
alendronate, estradiol, Fosamax, Premarin, Reclast, Estrace, raloxifeneFull Prescribing Information
2. Femhrt Dosage and Administration
Use of estrogen-alone, or in combination with a progestin, should be with the lowest effective dose and for the shortest duration consistent with treatment goals and risks for the individual woman. Postmenopausal women should be re-evaluated periodically as clinically appropriate to determine if treatment is still necessary.
3. Dosage Forms and Strengths
The following two strengths of femhrt are available:
femhrt (0.5 mg/2.5 mcg): Each oval white tablet contains 0.5 mg norethindrone acetate and 2.5 mcg ethinyl estradiol; imprinted with WC on one side and 145 on the other
4. Contraindications
femhrt is contraindicated in women with any of the following conditions:
- Undiagnosed abnormal genital bleeding
- Known, suspected, or history of breast cancer
- Known or suspected estrogen-dependent neoplasia
- Active DVT, PE or a history of these conditions
- Active arterial thromboembolic disease (for example, stroke and MI), or a history of these conditions
- Known anaphylactic reaction or angioedema to femhrt
- Known liver impairment or disease
- Known protein C, protein S, or antithrombin deficiency, or other known thrombophilic disorders
- Known or suspected pregnancy
5. Warnings and Precautions
5.1 Cardiovascular Disorders
An increased risk of PE, DVT, stroke, and MI has been reported with estrogen plus progestin therapy. An increased risk of stroke and DVT has been reported with estrogen-alone therapy. Should any of these occur or be suspected, estrogen with or without progestin therapy should be discontinued immediately.
Risk factors for arterial vascular disease (for example, hypertension, diabetes mellitus, tobacco use, hypercholesterolemia, and obesity) and/or venous thromboembolism (VTE) (for example, personal history or family history of VTE, obesity, and systemic lupus erythematosus) should be managed appropriately.
Stroke
In the WHI estrogen plus progestin substudy, a statistically significant increased risk of stroke was reported in women 50 to 79 years of age receiving daily CE (0.625 mg) plus MPA (2.5 mg) compared to women in the same age group receiving placebo (33 versus 25 per 10,000 women- years) [see Clinical Studies (14.5)]. The increase in risk was demonstrated after the first year and persisted.1 Should a stroke occur or be suspected, estrogen plus progestin therapy should be discontinued immediately.
In the WHI estrogen-alone substudy, a statistically significant increased risk of stroke was reported in women 50 to 79 years of age receiving daily conjugated estrogens CE (0.625 mg)-alone compared to women in the same age group receiving placebo (45 versus 33 per 10,000 women-years). The increase in risk was demonstrated in year 1 and persisted [see Clinical Studies (14.5)]. Should a stroke occur or be suspected, estrogen-alone therapy should be discontinued immediately.
Subgroup analyses of women 50 to 59 years of age suggest no increased risk of stroke for those women receiving CE (0.625 mg)-alone versus those receiving placebo (18 versus 21 per 10,000 women-years).1
Coronary Heart Disease
In the WHI estrogen plus progestin substudy, there was a statistically non-significant increased risk of coronary heart disease (CHD) events (defined as nonfatal MI, silent MI, or CHD death) reported in women receiving daily CE (0.625 mg) plus MPA (2.5 mg) compared to women receiving placebo (41 versus 34 per 10,000 women-years).1 An increase in relative risk was demonstrated in year 1, and a trend toward decreasing relative risk was reported in years 2 through 5 [see Clinical Studies (14.5)].
In the WHI estrogen-alone substudy, no overall effect on CHD events was reported in women receiving estrogen-alone compared to placebo2[see Clinical Studies (14.5)].
Subgroup analyses of women 50 to 59 years of age suggest a statistically non-significant reduction in CHD events (CE [0.625 mg]-alone compared to placebo) in women with less than 10 years since menopause (8 versus 16 per 10,000 woman-years).1
In postmenopausal women with documented heart disease (n = 2,763), average 66.7 years of age, in a controlled clinical trial of secondary prevention of cardiovascular disease (Heart and Estrogen/Progestin Replacement Study [HERS]), treatment with daily CE (0.625 mg) plus MPA (2.5 mg) demonstrated no cardiovascular benefit. During an average follow-up of 4.1 years, treatment with CE plus MPA did not reduce the overall rate of CHD events in postmenopausal women with established CHD. There were more CHD events in the CE plus MPA-treated group than in the placebo group in year 1, but not during the subsequent years. Two thousand, three hundred and twenty-one (2,321) women from the original HERS trial agreed to participate in an open label extension of HERS, HERS II. Average follow-up in HERS II was an additional 2.7 years, for a total of 6.8 years overall. Rates of CHD events were comparable among women in the CE plus MPA group and the placebo group in HERS, HERS II, and overall.
Venous Thromboembolism
In the WHI estrogen plus progestin substudy, a statistically significant 2-fold greater rate of VTE (DVT and PE) was reported in women receiving daily CE (0.625 mg) plus MPA (2.5 mg) compared to women receiving placebo (35 versus 17 per 10,000 women-years). Statistically significant increases in risk for both DVT (26 versus 13 per 10,000 women-years) and PE (18 versus 8 per 10,000 women-years) were also demonstrated. The increase in VTE risk was demonstrated during the first year and persisted3[see Clinical Studies (14.5)]. Should a VTE occur or be suspected, estrogen plus progestin therapy should be discontinued immediately.
In the WHI estrogen-alone substudy, the risk of VTE was increased for women receiving daily CE (0.625 mg)-alone compared to placebo (30 versus 22 per 10,000 women-years), although only the increased risk of DVT reached statistical significance (23 versus 15 per 10,000 women-years). The increase in VTE risk was demonstrated during the first 2 years4[see Clinical Studies (14.5)]. Should a VTE occur or be suspected, estrogen-alone therapy should be discontinued immediately.
If feasible, estrogens should be discontinued at least 4 to 6 weeks before surgery of the type associated with an increased risk of thromboembolism or during periods of prolonged immobilization.
5.2 Malignant Neoplasms
Breast Cancer
The most important randomized clinical trial providing information about breast cancer in estrogen plus progestin users is the WHI substudy of daily CE (0.625 mg) plus MPA (2.5 mg). After a mean follow-up of 5.6 years, the estrogen plus progestin substudy reported an increased risk of invasive breast cancer in women who took daily CE plus MPA. In this substudy, prior use of estrogen-alone or estrogen plus progestin therapy was reported by 26 percent of the women. The relative risk of invasive breast cancer was 1.24, and the absolute risk was 41 versus 33 cases per 10,000 women-years for CE plus MPA compared with placebo [see Clinical Studies (14.5)]. Among women who reported prior use of hormone therapy, the relative risk of invasive breast cancer was 1.86 and the absolute risk was 46 versus 25 cases per 10,000 women-years for CE plus MPA compared with placebo. Among women who reported no prior use of hormone therapy, the relative risk of invasive breast cancer was 1.09, and the absolute risk was 40 versus 36 cases per 10,000 women-years for CE plus MPA compared with placebo. In the same substudy, invasive breast cancers were larger, were more likely to be node positive, and were diagnosed at a more advanced stage in the CE (0.625 mg) plus MPA (2.5 mg) group compared with the placebo group. Metastatic disease was rare with no apparent difference between the two groups. Other prognostic factors, such as histologic subtype, grade and hormone receptor status did not differ between the groups5[see Clinical Studies (14.5)].
The most important randomized clinical trial providing information about breast cancer in estrogen-alone users is the WHI substudy of daily CE (0.625 mg)-alone. In the WHI estrogen-alone substudy, after an average follow up of 7.1 years, daily CE-alone was not associated with an increased risk of invasive breast cancer (relative risk [RR] 0.80 [see Clinical Studies (14.5)].
Consistent with the WHI clinical trials, observational studies have also reported an increased risk of breast cancer for estrogen plus progestin therapy, and a smaller increased risk for estrogen-alone therapy, after several years of use. The risk increased with duration of use, and appeared to return to baseline over about 5 years after stopping treatment (only the observational studies have substantial data on risk after stopping). Observational studies also suggest that the risk of breast cancer was greater, and became apparent earlier, with estrogen plus progestin therapy as compared to estrogen-alone therapy. However, these studies have not found significant variation in the risk of breast cancer among different estrogen plus progestin combinations, doses, or routes of administration.
The use of estrogen-alone and estrogen plus progestin has been reported to result in an increase in abnormal mammograms requiring further evaluation.
All women should receive yearly breast examinations by a healthcare provider and perform monthly breast self-examinations. In addition, mammography examinations should be scheduled based on patient age, risk factors and prior mammogram results.
Endometrial Cancer
Endometrial hyperplasia (a possible precursor of endometrial cancer) has been reported to occur at a rate of approximately 1 percent or less with femhrt.
An increased risk of endometrial cancer has been reported with the use of unopposed estrogen therapy in a woman with a uterus. The reported endometrial cancer risk among unopposed estrogen users is about 2 to 12 times greater than in non-users, and appears dependent on duration of treatment and on estrogen dose. Most studies show no significant increased risk associated with use of estrogens for less than 1 year. The greatest risk appears associated with prolonged use, with increased risks of 15- to 24-fold for 5 to 10 years or more. This risk has been shown to persist for at least 8 to 15 years after estrogen therapy is discontinued.
Clinical surveillance of all women using estrogen-alone or estrogen plus progestin therapy is important. Adequate diagnostic measures, including directed or random endometrial sampling when indicated, should be undertaken to rule out malignancy in postmenopausal women with undiagnosed persistent or recurring abnormal genital bleeding.
There is no evidence that the use of natural estrogens results in a different endometrial risk profile than synthetic estrogens of equivalent estrogen dose. Adding a progestin to estrogen therapy in postmenopausal women has been shown to reduce the risk of endometrial hyperplasia, which may be a precursor to endometrial cancer.
Ovarian Cancer
The WHI estrogen plus progestin substudy reported a statistically non-significant increased risk of ovarian cancer. After an average follow-up of 5.6 years, the relative risk for ovarian cancer for CE plus MPA versus placebo was 1.58 (95 percent CI, 0.77-3.24). The absolute risk for CE plus MPA versus placebo was 4 versus 3 cases per 10,000 women-years.7
A meta-analysis of 17 prospective and 35 retrospective epidemiology studies found that women who used hormonal therapy for menopausal symptoms had an increased risk for ovarian cancer. The primary analysis, using case-control comparisons, included 12,110 cancer cases from the 17 prospective studies. The relative risks associated with current use of hormonal therapy was 1.41 (95% confidence interval [CI] 1.32 to 1.50); there was no difference in the risk estimates by duration of the exposure (less than 5 years [median of 3 years] vs. greater than 5 years [median of 10 years] of use before the cancer diagnosis). The relative risk associated with combined current and recent use (discontinued use within 5 years before cancer diagnosis) was 1.37 (95% CI 1.27-1.48), and the elevated risk was significant for both estrogen-alone and estrogen plus progestin products. The exact duration of hormone therapy use associated with an increased risk of ovarian cancer, however, is unknown.
5.3 Probable Dementia
In the WHIMS estrogen plus progestin ancillary study of WHI, a population of 4,532 postmenopausal women 65 to 79 years of age was randomized to daily CE (0.625 mg) plus MPA (2.5 mg) or placebo.
After an average follow-up of 4 years, 40 women in the CE plus MPA group and 21 women in the placebo group were diagnosed with probable dementia. The relative risk of probable dementia for CE plus MPA versus placebo was 2.05 (95 percent CI, 1.21-3.48). The absolute risk of probable dementia for CE plus MPA versus placebo was 45 versus 22 cases per 10,000 women-years8[see Use in Specific Populations (8.5), and Clinical Studies (14.6)].
In the WHIMS estrogen-alone ancillary study of WHI, a population of 2,947 hysterectomized women 65 to 79 years of age was randomized to daily CE (0.625 mg)-alone or placebo.
After an average follow-up of 5.2 years, 28 women in the estrogen-alone group and 19 women in the placebo group were diagnosed with probable dementia. The relative risk of probable dementia for CE-alone versus placebo was 1.49 (95 percent CI, 0.83-2.66). The absolute risk of probable dementia for CE-alone versus placebo was 37 versus 25 cases per 10,000 women-years8 [see Use in Specific Populations (8.5), and Clinical Studies (14.6)].
When data from the two populations in the WHIMS estrogen-alone and estrogen plus progestin ancillary studies were pooled as planned in the WHIMS protocol, the reported overall relative risk for probable dementia was 1.76 (95 percent CI, 1.19-2.60). Since both ancillary substudies were conducted in women 65 to 79 years of age, it is unknown whether these findings apply to younger postmenopausal women8[see Use in Specific Populations (8.5), and Clinical Studies (14.6)].
5.4 Gallbladder Disease
A 2- to 4-fold increase in the risk of gallbladder disease requiring surgery in postmenopausal women receiving estrogens has been reported.
5.5 Hypercalcemia
Estrogen administration may lead to severe hypercalcemia in women with breast cancer and bone metastases. If hypercalcemia occurs, use of the drug should be stopped and appropriate measures taken to reduce the serum calcium level.
5.6 Visual Abnormalities
Retinal vascular thrombosis has been reported in women receiving estrogens. Discontinue medication pending examination if there is sudden partial or complete loss of vision, or a sudden onset of proptosis, diplopia, or migraine. If examination reveals papilledema or retinal vascular lesions, estrogens should be permanently discontinued.
5.9 Hypertriglyceridemia
In women with pre-existing hypertriglyceridemia, estrogen therapy may be associated with elevations of plasma triglycerides leading to pancreatitis. Consider discontinuation of treatment if pancreatitis occurs.
5.10 Hepatic Impairment and/or a Past History of Cholestatic Jaundice
Estrogens may be poorly metabolized in women with impaired liver function. For women with a history of cholestatic jaundice associated with past estrogen use or with pregnancy, caution should be exercised and in the case of recurrence, medication should be discontinued.
5.11 Hypothyroidism
Estrogen administration leads to increased thyroid-binding globulin (TBG) levels. Women with normal thyroid function can compensate for the increased TBG by making more thyroid hormone, thus maintaining free T4 and T3 serum concentrations in the normal range. Women dependent on thyroid hormone replacement therapy who are also receiving estrogen may require increased doses of their thyroid replacement therapy. These women should have their thyroid function monitored in order to maintain their free thyroid hormone levels in an acceptable range.
5.18 Drug-Laboratory Test Interactions
Accelerated prothrombin time, partial thromboplastin time, and platelet aggregation time; increased platelet count; increased factors II, VII antigen, VIII antigen, VIII coagulant activity, IX, X, XII, VII-X complex, II-VII-X complex, and beta-thromboglobulin; decreased levels of antifactor Xa and antithrombin III, decreased antithrombin III activity; increased levels of fibrinogen and fibrinogen activity; increased plasminogen antigen and activity.
Increased TBG levels leading to increased circulating total thyroid hormone levels as measured by protein-bound iodine (PBI), T4 levels (by column or by radioimmunoassay) or T3 levels by radioimmunoassay. T3 resin uptake is decreased, reflecting the elevated TBG. Free T4 and free T3 concentrations are unaltered. Women on thyroid replacement therapy may require higher doses of thyroid hormone.
Other binding proteins may be elevated in serum, for example, corticosteroid binding globulin (CBG), sex hormone-binding globulin (SHBG), leading to increased total circulating corticosteroids and sex steroids, respectively. femhrt 1/5 was associated with an SHBG increase of 22 percent. Free hormone concentrations, such as testosterone and estradiol, may be decreased. Other plasma proteins may be increased (angiotensinogen/renin substrate, alpha-1-antitrypsin, ceruloplasmin).
Increased plasma high-density lipoprotein (HDL) and HDL2 cholesterol subfraction concentrations, reduced low-density lipoprotein (LDL) cholesterol concentration, increased triglycerides levels.
Impaired glucose tolerance.
6. Adverse Reactions/Side Effects
The following serious adverse reactions are discussed elsewhere in the labeling:
- Cardiovascular Disorders [see Boxed Warning, Warnings and Precautions (5.1)].
- Malignant Neoplasms [see Boxed Warning, Warnings and Precautions (5.2)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse reactions reported by ≥5 percent of subjects in controlled clinical studies of femhrt are shown in Table 1.
| BODY SYSTEM/ Adverse Reaction | Number (Percent) of Subjects | ||
| Placebo | femhrt 0.5/2.5 | femhrt 1/5 | |
| N = 247 | N = 244 | N = 258 | |
| BODY AS A WHOLE | 23 (12.8) | 30 (16.9) | 30 (15.7) |
| Edema - Generalized | 10 (4.0) | 12 (4.9) | 11 (4.3) |
| Headache | 12 (4.9) | 14 (5.7) | 16 (6.2) |
| DIGESTIVE SYSTEM | 8 (4.4) | 17 (9.6) | 25 (13.1) |
| Abdominal Pain | 3 (1.2) | 13 (5.3) | 14 (6.8) |
| UROGENITAL SYSTEM | 20 (11.1) | 34 (19.2) | 45 (23.6) |
| Breast Pain | 9 (3.6) | 22 (9.0) | 20 (7.8) |
* The total number of subjects for each body system may be less than the number of subjects with AEs in that body system because a subject may have had more than one AE per body system
7. Drug Interactions
No drug-drug interaction studies have been conducted for femhrt.
7.1 Effect of Other Drugs on Combined Hormonal Products
Substances decreasing or increasing the plasma concentration of estrogen: In vitro and in vivo studies have shown that estrogens are metabolized partially by cytochrome P450 3A4 (CYP3A4). Therefore, inducers or inhibitors of CYP3A4 may affect estrogen drug metabolism. Inducers of CYP3A4 such as St. John’s wort (Hypericum perforatum) preparations, phenobarbital, carbamazepine and rifampin may decrease the plasma concentration of estrogens, possibly resulting in a decrease in therapeutic effects and/or changes in the uterine bleeding profile. Inhibitors of CYP3A4 such as erythromycin, clarithromycin, ketoconazole, itraconazole, ritonavir and grapefruit juice may increase the plasma concentration of estrogens and may result in side effects. Co-administration of atorvastatin and certain hormonal products containing ethinyl estradiol increase AUC values for ethinyl estradiol approximately 20 percent. Ascorbic acid and acetaminophen may increase the plasma ethinyl estradiol concentration, possibly by inhibition of conjugation.
8. Use In Specific Populations
8.1 Pregnancy
femhrt should not be used during pregnancy [see Contraindications (4)]. There appears to be little or no increased risk of birth defects in children born to women who have used estrogens and progestins as an oral contraceptive inadvertently during early pregnancy.
8.3 Nursing Mothers
femhrt should not be used during lactation. Estrogen administration to nursing women has been shown to decrease the quantity and quality of the breast milk. Detectable amounts of estrogen and progestin have been identified in the breast milk of women receiving estrogen plus progestin therapy. Caution should be exercised when femhrt is administered to a nursing woman.
8.5 Geriatric Use
There have not been sufficient numbers of geriatric women involved in clinical studies utilizing femhrt to determine whether those over 65 years of age differ from younger subjects in their response to femhrt.
The Women’s Health Initiative Studies
In the WHI estrogen plus progestin substudy (daily CE [0.625 mg] plus MPA [2.5 mg] versus placebo), there was a higher relative risk of nonfatal stroke and invasive breast cancer in women greater than 65 years of age [see Clinical Studies (14.5)].
In the WHI estrogen-alone substudy (daily CE [0.625 mg]-alone versus placebo), there was a higher relative risk of stroke in women greater than 65 years of age [see Clinical Studies (14.5)].
The Women’s Health Initiative Memory Study
In the WHIMS ancillary studies of postmenopausal women 65 to 79 years of age, there was an increased risk of developing probable dementia in women receiving estrogen plus progestin or estrogen-alone when compared to placebo [see Warnings and Precautions (5.3), and Clinical Studies (14.6)].
Since both ancillary studies were conducted in women 65 to 79 years of age, it is unknown whether these findings apply to younger postmenopausal women8[see Warnings and Precautions (5.3), and Clinical Studies (14.6)].
14. Clinical Studies
14.5 Women's Health Initiative Studies
The WHI enrolled approximately 27,000 predominantly healthy postmenopausal women in two substudies to assess the risks and benefits of daily oral CE (0.625 mg)-alone or in combination with MPA (2.5 mg) compared to placebo in the prevention of certain chronic diseases. The primary endpoint was the incidence of CHD (defined as nonfatal MI, silent MI and CHD death), with invasive breast cancer as the primary adverse outcome. A “global index” included the earliest occurrence of CHD, invasive breast cancer, stroke, PE, endometrial cancer (only in the CE plus MPA substudy), colorectal cancer, hip fracture, or death due to other cause. The study did not evaluate the effects of CE plus MPA or CE-alone on menopausal symptoms.
WHI Estrogen Plus Progestin Substudy
The WHI estrogen plus progestin substudy was stopped early. According to the predefined stopping rule, after an average follow-up of 5.6 years of treatment, the increased risk of invasive breast cancer and cardiovascular events exceeded the specified benefits included in the “global index”. The absolute excess risk of events included in the “global index” was 19 per 10,000 women-years.
For those outcomes included in the WHI “global index” that reached statistical significance after 5.6 years of follow-up, the absolute excess risks per 10,000 women-years in the group treated with CE plus MPA were 7 more CHD events, 8 more strokes, 10 more PEs, and 8 more invasive breast cancers, while the absolute risk reductions per 10,000 women-years were 6 fewer colorectal cancers and 5 fewer hip fractures.
Results of the CE plus MPA substudy, which included 16,608 women (average 63 years of age, range 50 to 79; 83.9 percent White, 6.8 percent Black, 5.4 percent Hispanic, 3.9 percent Other) are presented in Table 6. These results reflect centrally adjudicated data after an average follow-up of 5.6 years.
| Event | Relative Risk CE/MPA vs. Placebo (95 percent nCI ß ) | CE/MPA n = 8,506 | Placebo n = 8,102 |
| Absolute Risk per 10,000 Women-Years | |||
| CHD events | 1.23 (0.99 - 1.53) | 41 | 34 |
| Non-fatal MI | 1.28 (1.00 - 1.63) | 31 | 25 |
| CHD death | 1.10 (0.70 - 1.75) | 8 | 8 |
| All strokes | 1.31 (1.03 - 1.68) | 33 | 25 |
| Ischemic stroke | 1.44 (1.09 – 1.90) | 26 | 18 |
| Deep vein thrombosis à | 1.95 (1.43 - 2.67) | 26 | 13 |
| Pulmonary embolism | 2.13 (1.45 - 3.11) | 18 | 8 |
| Invasive breast cancerè | 1.24 (1.01 - 1.54) | 41 | 33 |
| Colorectal cancer | 0.61 (0.42 - 0.87) | 10 | 16 |
| Endometrial cancer à | 0.81 (0.48 - 1.36) | 6 | 7 |
| Cervical cancerà | 1.44 (0.47 - 4.42) | 2 | 1 |
| Hip fracture | 0.67 (0.47 - 0.96) | 11 | 16 |
| Vertebral fractures à | 0.65 (0.46 - 0.92) | 11 | 17 |
| Lower arm/wrist fractures à | 0.71 (0.59 - 0.85) | 44 | 62 |
| Total fractures à | 0.76 (0.69 - 0.83) | 152 | 199 |
| Overall Mortality ß ð | 1.00 (0.83 - 1.19) | 52 | 52 |
| Global Index ø | 1.13 (1.02 - 1.25) | 184 | 165 |
¶ Adapted from numerous WHI publications. WHI publications can be viewed at www.nhlbi.nih.gov/whi.
#Þ Results are based on centrally adjudicated data.
ß Nominal confidence intervals unadjusted for multiple looks and multiple comparisons.
à Not included in "global index".
è Includes metastic and non-metastic breast cancer with the exception of in situ cancer.
ð All deaths, except from breast or colorectal cancer, definite or probable CHD, PE or cerebrovascular disease.
ø A subset of the events was combined in a "global index" defined as the earliest occurrence of CHD events, invasive breast cancer, stroke, pulmonary embolism, colorectal cancer, hip fracture, or death due to other causes.
Timing of the initiation of estrogen plus progestin therapy relative to the start of menopause may affect the overall risk benefit profile. The WHI estrogen plus progestin substudy stratified by age showed in women 50 to 59 years of age, a non-significant trend toward reduced risk for overall mortality (hazard ratio (HR) 0.69 (95 percent CI, 0.44-1.07)].
WHI Estrogen-Alone Substudy
The WHI estrogen-alone substudy was also stopped early because an increased risk of stroke was observed, and it was deemed that no further information would be obtained regarding the risks and benefits of estrogen-alone in predetermined primary endpoints.
Results of the estrogen-alone substudy, which included 10,739 women (average 63 years of age, range 50 to 79; 75.3 percent White, 15.1 percent Black, 6.1 percent Hispanic, 3.6 percent Other), after an average follow-up of 7.1 years, are presented in Table 7.
| Event | Relative Risk CE vs. Placebo (95 percent nCIΠ| CE N = 5,310 | Placebo n = 5,429 |
| Absolute Risk per 10,000 Women-Years | |||
| CHD eventsœ | 0.95 (0.78 - 1.16) | 54 | 57 |
| Non-fatal MIœ | 0.91 (0.73-1.14) | 40 | 42 |
| CHD deathœ | 1.01 (0.71-1.43) | 16 | 16 |
| All strokesœ | 1.33 (1.05-1.68) | 45 | 33 |
| Ischemic strokeœ | 1.55 (1.19-2.01) | 38 | 25 |
| Deep vein thrombosisœ Ɖ | 1.47 (1.06-2.06) | 23 | 15 |
| Pulmonary embolismœ | 1.37 (0.90-2.07) | 14 | 10 |
| Invasive breast cancerœ | 0.80 (0.62-1.04) | 28 | 34 |
| Colorectal cancer* | 1.08 (0.75-1.55) | 17 | 16 |
| Hip fractureœ | 0.65 (0.45-0.94) | 12 | 19 |
| Vertebral fracturesœ Ɖ | 0.64 (0.44-0.93) | 11 | 18 |
| Lower arm/wrist fracturesœ Ɖ | 0.58 (0.47-0.72) | 35 | 59 |
| Total fracturesœ Ɖ | 0.71 (0.64-0.80) | 144 | 197 |
| Deaths due to other causes* † | 1.08 (0.88-1.32) | 53 | 50 |
| Overall Mortalityœ Ɖ | 1.04 (0.88-1.22) | 79 | 75 |
| Global Index‡ | 1.02(0.92-1.13) | 206 | 201 |
¥ Adapted from numerous WHI publications. WHI publications can be viewed at www.nblbi.nih.gov/whi.
ΠNominal confidence intervals unadjusted for multiple looks and multiple comparisons.
œ Results are based on centrally adjudicated data for an average follow-up of 7.1 years.
Ɖ Not included in "global index".
* Results are based on an average follow-up of 6.8 years.
† All deaths, except from breast or colorectal cancer, definite or probable CHD, PE or cerebrovascular disease.
‡ A subset of the events was combined in a "global index" defined as the earliest occurrence of CHD events, invasive breast cancer, stroke, pulmonary embolism, colorectal cancer, hip fracture, or death due to other causes.
For those outcomes included in the WHI "global index" that reached statistical significance, the absolute excess risk per 10,000 women-years in the group treated with CE-alone were 12 more strokes, while the absolute risk reduction per 10,000 women-years was 7 fewer hip fractures.9 The absolute excess risk of events included in the "global index" was a non-significant 5 events per 10,000 women-years. There was no difference between the groups in terms of all-cause mortality.
No overall difference for primary CHD events (nonfatal MI, silent MI and CHD death) and invasive breast cancer incidence in women receiving CE-alone compared with placebo was reported in final centrally adjudicated results from the estrogen-alone substudy, after an average follow-up of 7.1 years (see Table 7).
Centrally adjudicated results for stroke events from the estrogen-alone substudy, after an average follow-up of 7.1 years, reported no significant difference in distribution of stroke subtype or severity, including fatal strokes, in women receiving CE-alone compared to placebo. Estrogen-alone therapy increased the risk of ischemic stroke, and this excess was present in all subgroups of women examined10 (see Table 7).
Timing of the initiation of estrogen-alone therapy relative to the start of menopause may affect the overall risk benefit profile. The WHI estrogen-alone substudy stratified by age, showed in women 50-59 years of age a non-significant trend toward reduced risk for CHD [HR 0.63 (95 percent CI, 0.36-1.09)] and overall mortality [HR 0.71 (95 percent CI, 0.46-1.11)].
14.6 Women's Health Initiative Memory Study
The WHIMS estrogen plus progestin ancillary study of WHI enrolled 4,532 predominantly healthy postmenopausal women 65 years of age and older (47 percent were 65 to 69 years of age, 35 percent were 70 to 74 years of age, and 18 percent were 75 years of age and older) to evaluate the effects of CE (0.625 mg) plus MPA (2.5 mg) on the incidence of probable dementia (primary outcome) compared to placebo.
After an average follow-up of 4 years, the relative risk of probable dementia for CE plus MPA versus placebo was 2.05 (95 percent CI, 1.21-3.48). The absolute risk of probable dementia for CE plus MPA versus placebo was 45 versus 22 per 10,000 women-years. Probable dementia as defined in this study included Alzheimer’s disease (AD), vascular dementia (VaD) and mixed types (having features of both AD and VaD). The most common classification of probable dementia in the treatment group and the placebo group was AD. Since the ancillary study was conducted in women 65 to 79 years of age, it is unknown whether these findings apply to younger postmenopausal women [see Warnings and Precautions (5.3), and Use in Specific Populations (8.5)].
The WHIMS estrogen-alone ancillary study of WHI enrolled 2,947 predominantly healthy hysterectomized postmenopausal women 65 to 79 years of age (45 percent were 65 to 69 years of age; 36 percent were 70 to 74 years of age; 19 percent were 75 years of age and older) to evaluate the effects of daily CE (0.625 mg)-alone on the incidence of probable dementia (primary outcome) compared to placebo.
After an average follow-up of 5.2 years, the relative risk of probable dementia for CE-alone versus placebo was 1.49 (95 percent CI, 0.83-2.66). The absolute risk of probable dementia for CE-alone versus placebo was 37 versus 25 cases per 10,000 women-years. Probable dementia as defined in this study included AD, VaD and mixed types (having features of both AD and VaD). The most common classification of probable dementia in the treatment group and the placebo group was AD. Since the ancillary study was conducted in women 65 to 79 years of age, it is unknown whether these findings apply to younger postmenopausal women [see Warnings and Precautions (5.3), and Use in Specific Populations (8.5)].
When data from the two populations were pooled as planned in the WHIMS protocol, the reported overall relative risk for probable dementia was 1.76 (95 percent CI, 1.19-2.60). Differences between groups became apparent in the first year of treatment. It is unknown whether these findings apply to younger postmenopausal women [see Warnings and Precautions (5.3), and Use in Specific Populations (8.5)].
Patient Information
femhrt (fĕ’mĕrt)
(norethindrone acetate/ethinyl estradiol)
Tablets
Read this Patient Information before you start taking femhrt and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your menopausal symptoms or your treatment.
What is the most important information I should know about femhrt (a combination of estrogen and progestin)?
|
What is femhrt?
femhrt is a prescription medicine that contains two kinds of hormones, an estrogen and a progestin.
What is femhrt used for?
femhrt is used after menopause to:
- Reduce moderate to severe hot flushes
Estrogens are hormones made by a woman’s ovaries. The ovaries normally stop making estrogens when a woman is between 45 and 55 years old. This drop in body estrogen levels causes the “change of life” or menopause, the end of monthly menstrual periods. Sometimes both ovaries are removed during an operation before natural menopause takes place. The sudden drop in estrogen levels causes “surgical menopause”.
When estrogen levels begin dropping, some women get very uncomfortable symptoms, such as feelings of warmth in the face, neck, and chest, or sudden intense episodes of heat and sweating (“hot flashes” or “hot flushes”). In some women the symptoms are mild, and they will not need to take estrogens. In other women, symptoms can be more severe.
- Help reduce your chances of getting osteoporosis (thin weak bones)
If you use femhrt only to prevent osteoporosis from menopause, talk with your healthcare provider about whether a different treatment or medicine without estrogens might be better for you. You and your healthcare provider should talk regularly about whether you still need treatment with femhrt.
Who should not take femhrt?
Do not take femhrt if you have had your uterus (womb) removed (hysterectomy).
femhrt contains a progestin to decrease the chance of getting cancer of the uterus. If you do not have a uterus, you do not need a progestin and you should not take femhrt.
Do not take femhrt if you:
-
have unusual vaginal bleeding
Vaginal bleeding after menopause may be a warning sign of cancer of the uterus (womb). Your healthcare provider should check any unusual vaginal bleeding to find out the cause.
-
currently have or have had certain cancers.
Estrogens may increase the chance of getting certain types of cancers, including cancer of the breast or uterus. If you have or have had cancer, talk with your healthcare provider about whether you should take femhrt.
-
had a stroke or heart attack
-
currently have or have had blood clots
-
currently have or have had liver problems
-
have been diagnosed with a bleeding disorder
-
are allergic to femhrt or any of its ingredients.
See the list of ingredients in femhrt at the end of this leaflet.
-
think you may be pregnant
femhrt is not for pregnant women. If you think you may be pregnant, you should have a pregnancy test and know the results. Do not take femhrt if the test is positive and talk to your healthcare provider.
What should I tell my healthcare provider before I take femhrt?
Before you take femhrt, tell your healthcare provider if you:
-
have any unusual vaginal bleeding
Vaginal bleeding after menopause may be a warning sign of cancer of the uterus (womb). Your healthcare provider should check any unusual vaginal bleeding to find out the cause.
-
have any other medical conditions
Your healthcare provider may need to check you more carefully if you have certain conditions, such as asthma (wheezing), epilepsy (seizures), diabetes, migraine, endometriosis, lupus, angioedema (swelling of face and tongue), or problems with your heart, liver, thyroid, kidneys, or have high calcium levels in your blood.
-
are going to have surgery or will be on bed rest
Your healthcare provider will let you know if you need to stop taking femhrt.
-
are breastfeeding
The hormones in femhrt can pass into your breast milk.
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Some medicines may affect how femhrt works. femhrt may also affect how your other medicines work. Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine.
How should I take femhrt?
- Take femhrt exactly as your healthcare provider tells you to take it.
- Take 1 femhrt tablet at the same time each day.
- You and your healthcare provider should talk regularly (every 3 to 6 months) about the dose you are taking and whether or not you still need treatment with femhrt.
What are the possible side effects of femhrt?
Side effects are grouped by how serious they are and how often they happen when you are treated.
Serious, but less common side effects include:
- heart attack
- stroke
- blood clots
- dementia
- breast cancer
- cancer of the lining of the uterus (womb)
- cancer of the ovary
- high blood pressure
- high blood sugar
- gallbladder disease
- liver problems
- changes in your thyroid hormone levels
- enlargement of benign tumors of the uterus (“fibroids”)
Call your healthcare provider right away if you get any of the following warning signs or any other unusual symptoms that concern you:
- new breast lumps
- unusual vaginal bleeding
- changes in vision or speech
- sudden new severe headaches
- severe pains in your chest or legs with or without shortness of breath, weakness and fatigue
Less serious, but common side effects include:
- headache
- breast pain
- irregular vaginal bleeding or spotting
- stomach or abdominal cramps, bloating
- hair loss
- fluid retention
- vaginal yeast infection
These are not all the possible side effects of femhrt. For more information, ask your healthcare provider or pharmacist. Tell your healthcare provider if you have any side effects that bother you or does not go away.
You may report side effects to Allergan at 1-800-678-1605 or FDA at 1-800-FDA-1088.
What can I do to lower my chances of a serious side effect with femhrt?
- Talk with your healthcare provider regularly about whether you should continue taking femhrt.
- If you have a uterus, talk with your healthcare provider about whether the addition of a progestin is right for you.
- The addition of a progestin is generally recommended for a woman with a uterus to reduce the chance of getting cancer of the uterus (womb).
- See your healthcare provider right away if you develop vaginal bleeding while taking femhrt.
- Have a pelvic exam, breast exam and mammogram (breast x-ray) every year unless your healthcare provider tells you something else.
- If members of your family have had breast cancer or if you have ever had breast lumps or an abnormal mammogram (breast x-ray), you may need to have breast exams more often.
- If you have high blood pressure, high cholesterol (fat in the blood), diabetes, are overweight, or use tobacco, you may have a higher chance for getting heart disease.
Ask your healthcare provider for ways to lower your chances of getting heart disease.
How should I store femhrt?
- Store femhrt at room temperature between 68° F to 77° F (20° C to 25° C).
Keep femhrt out of the reach of children.
General information about the safe and effective use of femhrt.
Medicines are sometimes prescribed for conditions that are not mentioned in patient information leaflets. Do not take femhrt for conditions for which it was not prescribed. Do not give femhrt to other people, even if they have the same symptoms you have. It may harm them.
This leaflet summarizes the most important information about femhrt. If you would like more information, talk with your healthcare provider or pharmacist. You can ask your pharmacist or healthcare provider for information about femhrt that is written for health professionals.
For more information go to www.femhrt.com or call 1-800-678-1605.
What are the ingredients in femhrt?
Active Ingredients: norethindrone acetate and ethinyl estradiol
Inactive Ingredients: calcium stearate, lactose monohydrate, microcrystalline cellulose and corn starch
This Patient Information has been approved by the U.S Food and Drug Administration.
Distributed by:
Allergan USA, Inc.
Irvine, CA 92612
© 2017 Allergan. All rights reserved.
FEMHRT® is a registered trademark of Allergan Pharmaceuticals International Limited
Revised 11/2017
| FEMHRT
norethindrone acetate/ethinyl estradiol tablet |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Allergan, Inc. (144796497) |