Drug Detail:Leqembi (Lecanemab-irmb)
Drug Class: Miscellaneous central nervous system agents
Highlights of Prescribing Information
LEQEMBI® (lecanemab-irmb) injection, for intravenous use
Initial U.S. Approval: 2023
WARNING: AMYLOID RELATED IMAGING ABNORMALITIES
See full prescribing information for complete boxed warning.
Monoclonal antibodies directed against aggregated forms of beta amyloid, including LEQEMBI, can cause amyloid related imaging abnormalities (ARIA), characterized as ARIA with edema (ARIA-E) and ARIA with hemosiderin deposition (ARIA-H). ARIA is usually asymptomatic, although rarely serious and life-threatening events can occur. Serious intracerebral hemorrhage greater than 1 cm have occurred in patients treated with this class of medications. (5.1, 6.1)
ApoE ε4 Homozygotes
Patients treated with this class of medications, including LEQEMBI, who are ApoE ε4 homozygotes have a higher incidence of ARIA, including symptomatic and serious ARIA, compared to heterozygotes and noncarriers. Testing for ApoE ε4 status should be performed prior to initiation of treatment to inform the risk of developing ARIA. Prior to testing, prescribers should discuss with patients the risk of ARIA across genotypes and the implications of genetic testing results. (5.1)
Consider the benefit of LEQEMBI for the treatment of Alzheimer’s disease and potential risk of serious adverse events associated with ARIA when deciding to initiate treatment with LEQEMBI. (5.1, 14)
Recent Major Changes
| Boxed Warning | 7/2023 |
| Indications and Usage (1) | 7/2023 |
| Dosage and Administration (2.3) | 7/2023 |
| Contraindications (4) | 7/2023 |
| Warnings and Precautions (5.1, 5.2, 5.3) | 7/2023 |
Indications and Usage for Leqembi
LEQEMBI is indicated for the treatment of Alzheimer’s disease. Treatment with LEQEMBI should be initiated in patients with mild cognitive impairment or mild dementia stage of disease, the population in which treatment was initiated in clinical trials. (1)
Leqembi Dosage and Administration
- Confirm the presence of amyloid beta pathology prior to initiating treatment. (2.1)
- The recommended dosage is 10 mg/kg that must be diluted then administered as an intravenous infusion over approximately one hour, once every two weeks. (2.2)
- Obtain a recent baseline brain MRI prior to initiating treatment. (2.3, 5.1)
- Obtain an MRI prior to the 5th, 7th, and 14th infusions. If radiographically observed ARIA occurs, treatment recommendations are based on type, severity, and presence of symptoms. (2.3, 5.1)
- Dilution in 250 mL of 0.9% Sodium Chloride Injection, USP, is required prior to administration. (2.4)
- Administer as an intravenous infusion over approximately one hour via a terminal low-protein binding 0.2 micron in-line filter. (2.5)
Dosage Forms and Strengths
Injection:
- 500 mg/5 mL (100 mg/mL) solution in a single-dose vial (3)
- 200 mg/2 mL (100 mg/mL) solution in a single-dose vial (3)
Contraindications
LEQEMBI is contraindicated in patients with serious hypersensitivity to lecanemab-irmb or to any of the excipients of LEQEMBI. (4)
Warnings and Precautions
- Amyloid Related Imaging Abnormalities (ARIA): Enhanced clinical vigilance for ARIA is recommended during the first 14 weeks of treatment with LEQEMBI. Risk of ARIA, including symptomatic ARIA, was increased in apolipoprotein E ε4 homozygotes compared to heterozygotes and noncarriers. If a patient experiences symptoms suggestive of ARIA, clinical evaluation should be performed, including MRI scanning if indicated. (2.3, 5.1)
- Infusion-Related Reactions: The infusion rate may be reduced, or the infusion may be discontinued, and appropriate therapy administered as clinically indicated. Consider pre-medication at subsequent dosing with antihistamines, non-steroidal anti-inflammatory drugs, or corticosteroids. (5.3)
Adverse Reactions/Side Effects
Most common adverse reactions (at approximately 10% and higher incidence compared to placebo): infusion-related reactions, amyloid related imaging abnormality-microhemorrhages, amyloid related imaging abnormality-edema/effusion, and headache. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Eisai Inc. at 1-888-274-2378 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 7/2023
Related/similar drugs
donepezil, memantine, Aricept, Namenda, rivastigmine, ExelonFull Prescribing Information
1. Indications and Usage for Leqembi
LEQEMBI is indicated for the treatment of Alzheimer’s disease. Treatment with LEQEMBI should be initiated in patients with mild cognitive impairment or mild dementia stage of disease, the population in which treatment was initiated in clinical trials.
2. Leqembi Dosage and Administration
2.1 Patient Selection
Confirm the presence of amyloid beta pathology prior to initiating treatment [see Clinical Pharmacology (12.1)].
2.2 Dosing Instructions
The recommended dosage of LEQEMBI is 10 mg/kg that must be diluted then administered as an intravenous infusion over approximately one hour, once every two weeks.
If an infusion is missed, administer the next dose as soon as possible.
2.3 Monitoring and Dosing Interruption for Amyloid Related Imaging Abnormalities
LEQEMBI can cause amyloid related imaging abnormalities -edema (ARIA-E) and -hemosiderin deposition (ARIA-H) [see Warnings and Precautions (5.1)].
Monitoring for ARIA
Obtain a recent baseline brain magnetic resonance imaging (MRI) prior to initiating treatment with LEQEMBI. Obtain an MRI prior to the 5th, 7th, and 14th infusions. If a patient experiences symptoms suggestive of ARIA, clinical evaluation should be performed, including an MRI if indicated.
Recommendations for Dosing Interruptions in Patients with ARIA
ARIA-E
The recommendations for dosing interruptions for patients with ARIA-E are provided in Table 1.
| Clinical Symptom Severity1 | ARIA-E Severity on MRI2 | ||
| Mild | Moderate | Severe | |
| Asymptomatic | May continue dosing | Suspend dosing3 | Suspend dosing3 |
| Mild | May continue dosing based on clinical judgment | Suspend dosing3 | |
| Moderate or Severe | Suspend dosing3 | ||
1 Clinical Symptom Severity Categories:
Mild: discomfort noticed, but no disruption of normal daily activity.
Moderate: discomfort sufficient to reduce or affect normal daily activity.
Severe: incapacitating, with inability to work or to perform normal daily activity.
2 See Table 3 for MRI severity [Warnings and Precautions (5.1)].
3 Suspend until MRI demonstrates radiographic resolution and symptoms, if present, resolve; consider a follow-up MRI to assess for resolution 2 to 4 months after initial identification. Resumption of dosing should be guided by clinical judgment.
ARIA-H
The recommendations for dosing interruptions for patients with ARIA-H are provided in Table 2.
| Clinical Symptom Severity | ARIA-H Severity on MRI1 | ||
| Mild | Moderate | Severe | |
| Asymptomatic | May continue dosing | Suspend dosing2 | Suspend dosing3 |
| Symptomatic | Suspend dosing2 | Suspend dosing2 | |
1 See Table 3 for MRI severity [Warnings and Precautions (5.1)].
2 Suspend until MRI demonstrates radiographic stabilization and symptoms, if present, resolve; resumption of dosing should be guided by clinical judgment; consider a follow-up MRI to assess for stabilization 2 to 4 months after initial identification.
3 Suspend until MRI demonstrates radiographic stabilization and symptoms, if present, resolve; use clinical judgment in considering whether to continue treatment or permanently discontinue LEQEMBI.
In patients who develop intracerebral hemorrhage greater than 1 cm in diameter during treatment with LEQEMBI, suspend dosing until MRI demonstrates radiographic stabilization and symptoms, if present, resolve. Use clinical judgment in considering whether to continue treatment after radiographic stabilization and resolution of symptoms or permanently discontinue LEQEMBI.
2.4 Dilution Instructions
- Prior to administration, LEQEMBI must be diluted in 250 mL of 0.9% Sodium Chloride Injection, USP.
- Use aseptic technique when preparing the LEQEMBI diluted solution for intravenous infusion.
- Calculate the dose (mg), the total volume (mL) of LEQEMBI solution required, and the number of vials needed based on the patient’s actual body weight and the recommended dose of 10 mg/kg. Each vial contains a LEQEMBI concentration of 100 mg/mL.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Check that the LEQEMBI solution is clear to opalescent and colorless to pale yellow. Do not use if opaque particles, discoloration, or other foreign particles are present.
- Remove the flip-off cap from the vial. Insert the sterile syringe needle into the vial through the center of the rubber stopper.
- Withdraw the required volume of LEQEMBI from the vial(s) and add to an infusion bag containing 250 mL of 0.9% Sodium Chloride Injection, USP.
- Each vial is for one-time use only. Discard any unused portion.
- Gently invert the infusion bag containing the LEQEMBI diluted solution to mix completely. Do not shake.
- After dilution, immediate use is recommended [see Description (11)]. If not administered immediately, store LEQEMBI refrigerated at 2°C to 8°C (36°F to 46°F) for up to 4 hours, or at room temperature up to 30°C (86°F) for up to 4 hours. Do not freeze.
2.5 Administration Instructions
- Visually inspect the LEQEMBI diluted solution for particles or discoloration prior to administration. Do not use if it is discolored, or opaque or foreign particles are seen.
- Prior to infusion, allow the LEQEMBI diluted solution to warm to room temperature.
- Infuse the entire volume of the LEQEMBI diluted solution intravenously over approximately one hour through an intravenous line containing a terminal low-protein binding 0.2 micron in-line filter. Flush infusion line to ensure all LEQEMBI is administered.
- Monitor for any signs or symptoms of an infusion-related reaction. The infusion rate may be reduced, or the infusion may be discontinued, and appropriate therapy administered as clinically indicated. Consider pre-medication at subsequent dosing with antihistamines, non-steroidal anti-inflammatory drugs, or corticosteroids [see Warnings and Precautions (5.3)].
3. Dosage Forms and Strengths
LEQEMBI is a clear to opalescent and colorless to pale yellow solution, available as:
- Injection: 500 mg/5 mL (100 mg/mL) in a single-dose vial
- Injection: 200 mg/2 mL (100 mg/mL) in a single-dose vial
4. Contraindications
LEQEMBI is contraindicated in patients with serious hypersensitivity to lecanemab-irmb or to any of the excipients of LEQEMBI. Reactions have included angioedema and anaphylaxis [see Warnings and Precautions (5.2)].
5. Warnings and Precautions
5.1 Amyloid Related Imaging Abnormalities
Monoclonal antibodies directed against aggregated forms of beta amyloid, including LEQEMBI, can cause amyloid related imaging abnormalities (ARIA), characterized as ARIA with edema (ARIA-E), which can be observed on MRI as brain edema or sulcal effusions, and ARIA with hemosiderin deposition (ARIA-H), which includes microhemorrhage and superficial siderosis. ARIA can occur spontaneously in patients with Alzheimer’s disease. ARIA-H associated with monoclonal antibodies directed against aggregated forms of beta amyloid generally occurs in association with an occurrence of ARIA-E. ARIA-H of any cause and ARIA-E can occur together.
ARIA usually occurs early in treatment and is usually asymptomatic, although serious and life-threatening events, including seizure and status epilepticus, rarely can occur. When present, reported symptoms associated with ARIA may include headache, confusion, visual changes, dizziness, nausea, and gait difficulty. Focal neurologic deficits may also occur. Symptoms associated with ARIA usually resolve over time. The risk of ARIA, including symptomatic and serious ARIA, is increased in apolipoprotein E ε4 (ApoE ε4) homozygotes. In addition to ARIA, intracerebral hemorrhages greater than 1 cm in diameter have occurred in patients treated with LEQEMBI.
Consider the benefit of LEQEMBI for the treatment of Alzheimer’s disease and potential risk of serious adverse events associated with ARIA when deciding to initiate treatment with LEQEMBI.
Incidence of ARIA
Symptomatic ARIA occurred in 3% (29/898) of patients treated with LEQEMBI in Study 2 [see Clinical Studies (14)]. Serious symptoms associated with ARIA were reported in 0.7% (6/898) of patients treated with LEQEMBI. Clinical symptoms associated with ARIA resolved in 79% (23/29) of patients during the period of observation. Similar findings were observed in Study 1.
Including asymptomatic radiographic events, ARIA was observed in 21% (191/898) of patients treated with LEQEMBI, compared to 9% (84/897) of patients on placebo in Study 2.
ARIA-E was observed in 13% (113/898) of patients treated with LEQEMBI compared with 2% (15/897) of patients on placebo. ARIA-H was observed in 17% (152/898) of patients treated with LEQEMBI compared with 9% (80/897) of patients on placebo. There was no increase in isolated ARIA-H (i.e., ARIA-H in patients who did not also experience ARIA-E) for LEQEMBI compared to placebo.
ApoE ε4 Carrier Status and Risk of ARIA
Approximately 15% of Alzheimer’s disease patients are ApoE ε4 homozygotes. In Study 2, 16% (141/898) of patients in the LEQEMBI arm were ApoE ε4 homozygotes, 53% (479/898) were heterozygotes, and 31% (278/898) were noncarriers. The incidence of ARIA was higher in ApoE ε4 homozygotes (45% on LEQEMBI vs. 22% on placebo) than in heterozygotes (19% on LEQEMBI vs 9% on placebo) and noncarriers (13% on LEQEMBI vs 4% on placebo). Among patients treated with LEQEMBI, symptomatic ARIA-E occurred in 9% of ApoE ε4 homozygotes compared with 2% of heterozygotes and 1% noncarriers. Serious events of ARIA occurred in 3% of ApoE ε4 homozygotes, and approximately 1% of heterozygotes and noncarriers. The recommendations on management of ARIA do not differ between ApoE ε4 carriers and noncarriers [see Dosage and Administration (2.3)]. Testing for ApoE ε4 status should be performed prior to initiation of treatment to inform the risk of developing ARIA. Prior to testing, prescribers should discuss with patients the risk of ARIA across genotypes and the implications of genetic testing results. Prescribers should inform patients that if genotype testing is not performed they can still be treated with LEQEMBI; however, it cannot be determined if they are ApoE ε4 homozygotes and at higher risk for ARIA. An FDA-authorized test for the detection of ApoE ε4 alleles to identify patients at risk of ARIA if treated with LEQEMBI is not currently available. Currently available tests used to identify ApoE ε4 alleles may vary in accuracy and design.
Radiographic Findings
The radiographic severity of ARIA associated with LEQEMBI was classified by the criteria shown in Table 3.
| ARIA Type | Radiographic Severity | ||
| Mild | Moderate | Severe | |
| ARIA-E | FLAIR hyperintensity confined to sulcus and/or cortex/subcortex white matter in one location <5 cm | FLAIR hyperintensity 5 to 10 cm in single greatest dimension, or more than 1 site of involvement, each measuring <10 cm | FLAIR hyperintensity >10 cm with associated gyral swelling and sulcal effacement. One or more separate/ independent sites of involvement may be noted. |
| ARIA-H microhemorrhage | ≤ 4 new incident microhemorrhages | 5 to 9 new incident microhemorrhages | 10 or more new incident microhemorrhages |
| ARIA-H superficial siderosis | 1 focal area of superficial siderosis | 2 focal areas of superficial siderosis | > 2 areas of superficial siderosis |
The majority of ARIA-E radiographic events occurred early in treatment (within the first 7 doses), although ARIA can occur at any time and patients can have more than 1 episode. The maximum radiographic severity of ARIA-E in patients treated with LEQEMBI was mild in 4% (37/898) of patients, moderate in 7% (66/898) of patients, and severe in 1% (9/898) of patients. Resolution on MRI occurred in 52% of ARIA-E patients by 12 weeks, 81% by 17 weeks, and 100% overall after detection. The maximum radiographic severity of ARIA-H microhemorrhage in patients treated with LEQEMBI was mild in 9% (79/898), moderate in 2% (19/898), and severe in 3% (28/898) of patients; superficial siderosis was mild in 4% (38/898), moderate in 1% (8/898), and severe in 0.4% (4/898). Among patients treated with LEQEMBI, the rate of severe radiographic ARIA-E was highest in ApoE ε4 homozygotes 5% (7/141), compared to heterozygotes 0.4% (2/479) or noncarriers 0% (0/278). Among patients treated with LEQEMBI, the rate of severe radiographic ARIA-H was highest in ApoE ε4 homozygotes 13.5% (19/141), compared to heterozygotes 2.1% (10/479) or noncarriers 1.1% (3/278).
Intracerebral Hemorrhage
Intracerebral hemorrhage greater than 1 cm in diameter was reported in 0.7% (6/898) of patients in Study 2 after treatment with LEQEMBI compared to 0.1% (1/897) on placebo. Fatal events of intracerebral hemorrhage in patients taking LEQEMBI have been observed.
Concomitant Antithrombotic Medication
In Study 2, baseline use of antithrombotic medication (aspirin, other antiplatelets, or anticoagulants) was allowed if the patient was on a stable dose. The majority of exposures to antithrombotic medications were to aspirin. Antithrombotic medications did not increase the risk of ARIA with LEQEMBI. The incidence of intracerebral hemorrhage was 0.9% (3/328 patients) in patients taking LEQEMBI with a concomitant antithrombotic medication at the time of the event compared to 0.6% (3/545 patients) in those who did not receive an antithrombotic. Patients taking LEQEMBI with an anticoagulant alone or combined with an antiplatelet medication or aspirin had an incidence of intracerebral hemorrhage of 2.5% (2/79 patients) compared to none in patients who received placebo.
Because intracerebral hemorrhages greater than 1 cm in diameter have been observed in patients taking LEQEMBI, additional caution should be exercised when considering the administration of anticoagulants or a thrombolytic agent (e.g., tissue plasminogen activator) to a patient already being treated with LEQEMBI.
Other Risk Factors for Intracerebral Hemorrhage
Patients were excluded from enrollment in Study 2 for findings on neuroimaging that indicated an increased risk for intracerebral hemorrhage. These included findings suggestive of cerebral amyloid angiopathy (prior cerebral hemorrhage greater than 1 cm in greatest diameter, more than 4 microhemorrhages, superficial siderosis, vasogenic edema) or other lesions (aneurysm, vascular malformation) that could potentially increase the risk of intracerebral hemorrhage.
The presence of an ApoE ε4 allele is also associated with cerebral amyloid angiopathy, which has an increased risk for intracerebral hemorrhage.
Caution should be exercised when considering the use of LEQEMBI in patients with factors that indicate an increased risk for intracerebral hemorrhage and in particular for patients who need to be on anticoagulant therapy.
Monitoring and Dose Management Guidelines
Recommendations for dosing in patients with ARIA-E depend on clinical symptoms and radiographic severity [see Dosage and Administration (2.3)]. Recommendations for dosing in patients with ARIA-H depend on the type of ARIA-H and radiographic severity [see Dosage and Administration (2.3)]. Use clinical judgment in considering whether to continue dosing in patients with recurrent ARIA-E.
Baseline brain MRI and periodic monitoring with MRI are recommended [see Dosage and Administration (2.3)]. Enhanced clinical vigilance for ARIA is recommended during the first 14 weeks of treatment with LEQEMBI. If a patient experiences symptoms suggestive of ARIA, clinical evaluation should be performed, including MRI if indicated. If ARIA is observed on MRI, careful clinical evaluation should be performed prior to continuing treatment.
There is no experience in patients who continued dosing through symptomatic ARIA-E, or through asymptomatic but radiographically severe ARIA-E. There is limited experience in patients who continued dosing through asymptomatic but radiographically mild to moderate ARIA-E. There are limited data in dosing patients who experienced recurrent ARIA-E.
The Alzheimer’s Network for Treatment and Diagnostics (ALZ-NET) is a voluntary provider-enrolled patient registry that collects information on treatments for Alzheimer’s disease, including LEQEMBI. Providers may obtain information about the registry at www.alz-net.org or contact [email protected].
5.2 Hypersensitivity Reactions
Hypersensitivity reactions, including angioedema, bronchospasm, and anaphylaxis, have occurred in patients who were treated with LEQEMBI. Promptly discontinue the infusion upon the first observation of any signs or symptoms consistent with a hypersensitivity reaction, and initiate appropriate therapy. LEQEMBI is contraindicated in patients with a history of serious hypersensitivity to lecanemab-irmb or to any of the excipients of LEQEMBI.
5.3 Infusion-Related Reactions
In Study 2, infusion-related reactions were observed in 26% (237/898) of patients treated with LEQEMBI compared to 7% (66/897) of patients on placebo; and the majority (75%, 178/237) occurred with the first infusion. Infusion-related reactions were mostly mild (69%) or moderate (28%) in severity. Infusion-related reactions resulted in discontinuations in 1% (12/898) of patients treated with LEQEMBI. Symptoms of infusion-related reactions include fever and flu-like symptoms (chills, generalized aches, feeling shaky, and joint pain), nausea, vomiting, hypotension, hypertension, and oxygen desaturation.
After the first infusion in Study 1, 38% of patients treated with LEQEMBI had transient decreased lymphocyte counts to less than 0.9 x109/L compared to 2% in patients on placebo, and 22% of patients treated with LEQEMBI had transient increased neutrophil counts to greater 7.9 x109/L compared to 1% of patients on placebo. Lymphocyte and neutrophil counts were not obtained after the first infusion in Study 2.
In the event of an infusion-related reaction, the infusion rate may be reduced, or the infusion may be discontinued, and appropriate therapy initiated as clinically indicated. Prophylactic treatment with antihistamines, acetaminophen, nonsteroidal anti-inflammatory drugs, or corticosteroids prior to future infusions may be considered.
6. Adverse Reactions/Side Effects
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Amyloid Related Imaging Abnormalities [see Warnings and Precautions (5.1)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.2)]
- Infusion-Related Reactions [see Warnings and Precautions (5.3)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of LEQEMBI has been evaluated in 2090 patients who received at least one dose of LEQEMBI. In Studies 1 and 2 in patients with Alzheimer’s disease, 1059 patients received LEQEMBI 10 mg/kg every two weeks [see Clinical Studies (14)]. Of these 1059 patients, 50% were female, 79% were White, 15% were Asian, 12% were of Hispanic or Latino ethnicity, and 2% were Black. The mean age at study entry was 72 years (range from 50 to 90 years).
In the combined double-blind, placebo-controlled period and long-term extension period of Studies 1 and 2, 1604 patients received LEQEMBI for at least 6 months, 1261 patients for at least 12 months, and 965 patients for 18 months.
In the double-blind, placebo-controlled period in Study 1 patients stopped study treatment because of an adverse reaction in 15% of patients treated with LEQEMBI, compared to 6% patients on placebo; in Study 2 patients stopped study treatment because of an adverse reaction in 7% of patients treated with LEQEMBI, compared to 3% patients on placebo. In Study 1, the most common adverse reaction leading to discontinuation of LEQEMBI was infusion-related reactions that led to discontinuation in 2% (4/161) of patients treated with LEQEMBI compared to 1% (2/245) of patients on placebo. In Study 2, the most common adverse reaction leading to discontinuation of LEQEMBI was ARIA-H microhemorrhages that led to discontinuation in 2% (15/898) of patients treated with LEQEMBI compared to <1% (1/897) of patients on placebo.
Table 4 shows adverse reactions that were reported in at least 5% of patients treated with LEQEMBI and at least 2% more frequently than in patients on placebo in Study 1.
| Adverse Reaction | LEQEMBI
10 mg/kg Every Two Weeks N=161 % | Placebo
N=245 % |
| Infusion-related reactions | 20 | 3 |
| Headache | 14 | 10 |
| ARIA-E | 10 | 1 |
| Cough | 9 | 5 |
| Diarrhea | 8 | 5 |
Table 5 shows adverse reactions that were reported in at least 5% of patients treated with LEQEMBI and at least 2% more frequently than in patients on placebo in Study 2.
| Adverse Reaction | LEQEMBI
10 mg/kg Every Two Weeks N=898 % | Placebo
N=897 % |
| Infusion-related reactions | 26 | 7 |
| ARIA-H | 14 | 8 |
| ARIA-E | 13 | 2 |
| Headache | 11 | 8 |
| Superficial siderosis of central nervous system | 6 | 3 |
| Rash1 | 6 | 4 |
| Nausea/Vomiting | 6 | 4 |
1 Rash includes acne, erythema, infusion site rash, injection site rash, rash, rash erythematous, rash pruritic, skin reactions, and urticaria.
Less Common Adverse Reactions
Atrial fibrillation occurred in 3% of patients treated with LEQEMBI compared to 2% in patients on placebo. In Study 1, lymphopenia or decreased lymphocyte count were reported in 4% of patients treated with LEQEMBI after the first dose, compared to less than 1% of patients on placebo [see Warnings and Precautions (5.3)]; lymphocytes were not measured after the first dose in Study 2.
11. Leqembi Description
Lecanemab-irmb is a recombinant humanized immunoglobulin gamma 1 (IgG1) monoclonal antibody directed against aggregated soluble and insoluble forms of amyloid beta, and is expressed in a Chinese hamster ovary cell line. Lecanemab-irmb has an approximate molecular weight of 150 kDa.
LEQEMBI (lecanemab-irmb) injection is a preservative-free, sterile, clear to opalescent and colorless to pale yellow solution for intravenous use by infusion after dilution. LEQEMBI is supplied in single-dose vials available in concentrations of 500 mg/5 mL (100 mg/mL) or 200 mg/2 mL (100 mg/mL).
Each mL of solution contains 100 mg of lecanemab-irmb and arginine hydrochloride (42.13 mg), histidine (0.18 mg), histidine hydrochloride monohydrate (4.99 mg), polysorbate 80 (0.50 mg), and Water for Injection at an approximate pH of 5.0.
12. Leqembi - Clinical Pharmacology
12.1 Mechanism of Action
Lecanemab-irmb is a humanized immunoglobulin gamma 1 (IgG1) monoclonal antibody directed against aggregated soluble and insoluble forms of amyloid beta. The accumulation of amyloid beta plaques in the brain is a defining pathophysiological feature of Alzheimer’s disease. LEQEMBI reduces amyloid beta plaques, as evaluated in Study 1 and Study 2 [see Clinical Studies (14)].
14. Clinical Studies
The efficacy of LEQEMBI was evaluated in two double-blind, placebo-controlled, parallel-group, randomized studies (Study 1, NCT01767311; Study 2 NCT03887455) in patients with Alzheimer’s disease (patients with confirmed presence of amyloid pathology and mild cognitive impairment [64% of patients in Study 1; 62% of patients in Study 2] or mild dementia stage of disease [36% of patients in Study 1; 38% of patients in Study 2], consistent with Stage 3 and Stage 4 Alzheimer’s disease). In both studies, patients were enrolled with a Clinical Dementia Rating (CDR) global score of 0.5 or 1.0 and a Memory Box score of 0.5 or greater. All patients had a Mini-Mental State Examination (MMSE) score of ≥22 and ≤30, and had objective impairment in episodic memory as indicated by at least 1 standard deviation below age-adjusted mean in the Wechsler-Memory Scale-IV Logical Memory II (subscale) (WMS-IV LMII). Patients were enrolled with or without concomitant approved therapies (cholinesterase inhibitors and the N-methyl-D-aspartate antagonist memantine) for Alzheimer’s disease. Patients in each study could enroll in an optional, long-term extension.
Study 1
In Study 1, 856 patients were randomized to receive one of 5 doses (161 of which were randomized to the recommended dosing regimen of 10 mg/kg every two weeks) of LEQEMBI or placebo (n=247). Of the total number of patients randomized, 71.4% were ApoE ε4 carriers and 28.6% were ApoE ε4 non-carriers. During the study the protocol was amended to no longer randomize ApoE ε4 carriers to the 10 mg/kg every two weeks dose arm. ApoE ε4 carriers who had been receiving LEQEMBI 10 mg/kg every two weeks for 6 months or less were discontinued from study drug. As a result, in the LEQEMBI 10 mg/kg every two weeks arm, 30.3% of patients were ApoE ε4 carriers and 69.7% were ApoE ε4 non-carriers. At baseline, the mean age of randomized patients was 71 years, with a range of 50 to 90 years. Fifty percent of patients were male and 90% were White.
In Study 1, a subgroup of 315 patients were enrolled in the amyloid PET substudy; of these, 277 were evaluated at Week 79. Results from the amyloid beta PET substudy are described in Figure 1 and Table 7. Plasma biomarkers are described in Table 5.
Figure 1: Reduction in Brain Amyloid Beta Plaque (Adjusted Mean Change from Baseline in Amyloid Beta PET Composite, SUVR and Centiloids) in Study 1
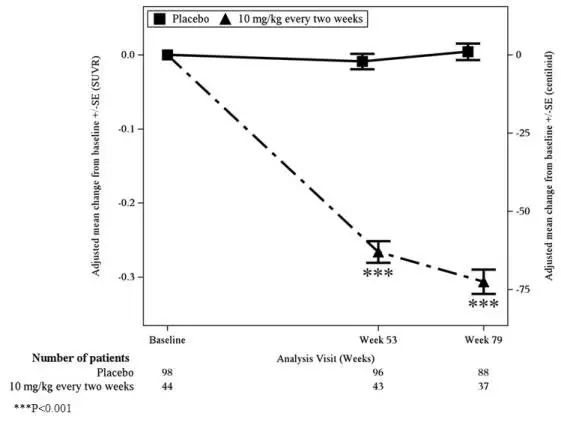
| Biomarker Endpoints | LEQEMBI
10 mg/kg Every Two Weeks | Placebo |
| Amyloid Beta PET Composite SUVR | N=44 | N=98 |
| Mean baseline | 1.373 | 1.402 |
| Adjusted mean change from baseline at Week 79 | -0.306 | 0.004 |
| Difference from placebo | -0.310 (p<0.001) 1 | |
| Amyloid Beta PET Centiloid | N=44 | N=98 |
| Mean baseline | 78.0 | 84.8 |
| Adjusted mean change from baseline at Week 79 | -72.5 | 1.0 |
| Difference from placebo | -73.5 (p<0.001) 1 | |
| N is the number of patients with baseline value. 1 P-values were not statistically controlled for multiple comparisons. |
||
The primary endpoint was change from baseline on a weighted composite score consisting of selected items from the Clinical Dementia Rating scale Sum of Boxes (CDR-SB), MMSE, and Alzheimer Disease Assessment Scale – Cognitive Subscale 14 (ADAS-Cog 14) at Week 53. LEQEMBI had a 64% likelihood of 25% or greater slowing of progression on the primary endpoint relative to placebo at Week 53, which did not meet the prespecified success criterion of 80%.
Key secondary efficacy endpoints included the change from baseline in amyloid PET SUVR composite at Week 79 and change from baseline in the CDR-SB and ADAS-Cog14 at Week 79. Results for clinical assessments showed less change from baseline in CDR-SB and ADAS-Cog 14 scores at Week 79 in the LEQEMBI group than in patients on placebo (CDR-SB: -0.40 [26%], 90% CI [-0.82, 0.03]; ADAS-Cog 14: -2.31 [47%], 90% CI [-3.91, -0.72].
After the 79-week double-blind, placebo-controlled period of Study 1, patients could enroll in an open-label extension period for up to 260 weeks, which was initiated after a gap period (range 9 to 59 months; mean 24 months) off treatment.
Study 2
In Study 2, 1795 patients were enrolled and randomized 1:1 to receive LEQEMBI 10 mg/kg or placebo once every 2 weeks. Of the total number of patients randomized, 69% were ApoE ε4 carriers and 31% were ApoE ε4 non-carriers. Overall median age of patients was 72 years, with a range of 50 to 90 years. Fifty-two percent were women, and 1381 (77%) were White, 303 (17%) were Asian, and 47 (3%) were Black.
The randomization was stratified according to clinical subgroup (mild cognitive impairment or mild dementia stage of the disease); the presence or absence of concomitant approved therapies for Alzheimer’s disease at baseline (cholinesterase inhibitors and the N-methyl-D-aspartate antagonist memantine); ApoE ε4 carrier status; and geographical region.
The primary efficacy outcome was change from baseline at 18 months in the CDR-SB. Key secondary endpoints included change from baseline at 18 months for the following measures: amyloid Positron Emission Tomography (PET) using Centiloids, ADAS-Cog14, and Alzheimer's Disease Cooperative Study-Activities of Daily Living Scale for Mild Cognitive Impairment (ADCS MCI-ADL).
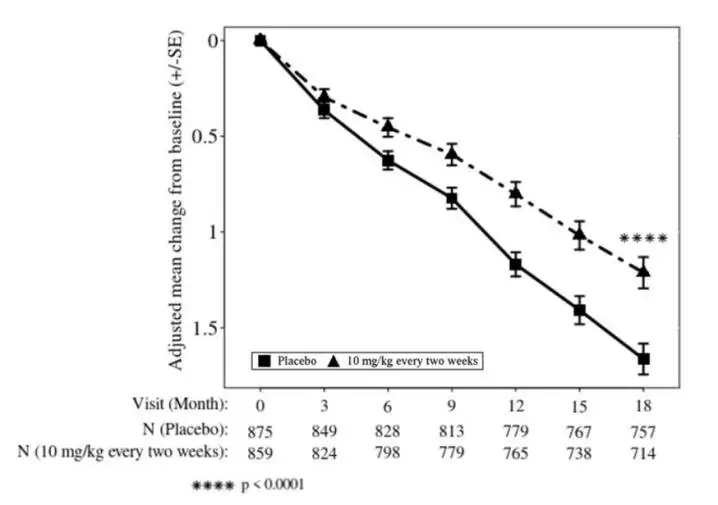
LEQEMBI treatment met the primary endpoint and reduced clinical decline on the global cognitive and functional scale, CDR-SB, compared with placebo at 18 months (-0.45 [-27%], p<0.0001).
Statistically significant differences (p<0.01) between treatment groups were also seen in the results for ADAS-Cog14 and ADCS MCI-ADL at 18 months as presented in Table 8.
Both ApoE ε4 carriers and ApoE ε4 noncarriers showed statistically significant treatment differences for the primary endpoint and all secondary endpoints. In an exploratory subgroup analysis of ApoE ε4 homozygotes, which represented 15% of the trial population, a treatment effect was not observed with LEQEMBI treatment on the primary endpoint, CDR-SB, compared to placebo, although treatment effects that favored LEQEMBI were observed for the secondary clinical endpoints, ADAS-Cog 14 and ADCS MCI-ADL. Treatment effects on disease-relevant biomarkers (amyloid beta PET, plasma Aβ42/40 ratio, plasma p-tau 181) also favored LEQEMBI in the ApoE ε4 homozygous subgroup.
Starting at six months, across all time points, LEQEMBI treatment showed statistically significant changes in the primary and all key secondary endpoints from baseline compared to placebo; see Figure 2.
| Clinical Endpoints | LEQEMBI
10 mg/kg Every Two Weeks | Placebo |
| CDR-SB | N=859 | N=875 |
| Mean baseline | 3.17 | 3.22 |
| Adjusted mean change from baseline at 18 months (%) Difference from placebo | 1.21 -0.45 (-27%) (p<0.0001) | 1.66 |
| ADAS-Cog14 | N=854 | N=872 |
| Mean baseline | 24.45 | 24.37 |
| Adjusted mean change from baseline at 18 months (%) Difference from placebo | 4.140 -1.442 (-26%) (p=0.00065) | 5.581 |
| ADCS MCI-ADL | N=783 | N=796 |
| Mean baseline | 41.2 | 40.9 |
| Adjusted mean change from baseline at 18 months Difference from placebo | -3.5 (-37%) 2.0 (p<0.0001) | -5.5 |
Figure 2: Adjusted Mean Change from Baseline in CDR-SB in Study 2
Medication Guide
| MEDICATION GUIDE
LEQEMBI® (leh-kem’-bee) (lecanemab-irmb) injection, for intravenous use |
|
| What is the most important information I should know about LEQEMBI?
LEQEMBI can cause serious side effects including:
|
|
|
|
|
|
|
|
| |
| Some people have a genetic risk factor (homozygous apolipoprotein E gene carriers) that may cause an increased risk for ARIA. Talk to your healthcare provider about testing to see if you have this risk factor. Some medicines can increase the risk for larger areas of bleeding in the brain in patients taking LEQEMBI. Talk to your healthcare provider to see if you are on any medicines that increase this risk. Your healthcare provider will do magnetic resonance imaging (MRI) scans before and during your treatment with LEQEMBI to check you for ARIA. Call your healthcare provider or go to the nearest hospital emergency room right away if you have any of the symptoms listed above. |
|
| What is LEQEMBI?
LEQEMBI is a prescription medicine used to treat people with Alzheimer’s disease. It is not known if LEQEMBI is safe and effective in children. |
|
Do not receive LEQEMBI if you:
|
|
Before receiving LEQEMBI, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take medicines to reduce blood clots from forming (antithrombotic medicines, including aspirin). Ask your healthcare provider for a list of these medicines if you are not sure. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|
How will I receive LEQEMBI?
|
|
| What are the possible side effects of LEQEMBI?
LEQEMBI can cause serious side effects, including:
|
|
|
|
|
|
|
|
| |
| If you have an infusion-related reaction, your healthcare provider may give you medicines before your LEQEMBI infusions to decrease your chance of having an infusion-related reaction. These medicines may include antihistamines, anti-inflammatory medicines, or steroids. The most common side effects of LEQEMBI include:
These are not all the possible side effects of LEQEMBI. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
| General information about the safe and effective use of LEQEMBI.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about LEQEMBI that is written for healthcare professionals. There is a registry that collects information on treatments for Alzheimer’s disease. The registry is named ALZ-NET (Alzheimer’s Network for Treatment and Diagnostics). Your healthcare provider can help you become enrolled in this registry. |
|
| What are the ingredients in LEQEMBI?
Active ingredient: lecanemab-irmb. Inactive ingredients: arginine hydrochloride, histidine, histidine hydrochloride monohydrate, polysorbate 80, and water for injection. |
|
| Manufactured by: Eisai Inc. Nutley, NJ 07110 U.S. License No. 1862 LEQEMBI® is a registered trademark of Eisai R&D Management Co., Ltd. © 2023 Eisai Inc. and Biogen For more information, go to www.LEQEMBI.com or call 1-888-274-2378. |
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. Revised: 7/2023
| LEQEMBI
lecanemab injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| LEQEMBI
lecanemab injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Eisai Inc. (189246791) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Biogen International GmbH | 480114999 | analysis(62856-215, 62856-212) , api manufacture(62856-215, 62856-212) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Biogen U.S. Corporation | 078734950 | analysis(62856-215, 62856-212) , manufacture(62856-215, 62856-212) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Bioreliance Corporation | 147227730 | analysis(62856-215, 62856-212) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Eurofins Biopharma Product Testing Munich GmbH | 313046917 | analysis(62856-215, 62856-212) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Packaging Coordinators, LLC | 078525133 | label(62856-215, 62856-212) , pack(62856-215, 62856-212) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Pharma Packaging Solutions, LLC dba Tjoapack, LLC | 928861723 | label(62856-215, 62856-212) , pack(62856-215, 62856-212) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| PPD Development, L.P. | 838082055 | analysis(62856-215, 62856-212) | |