Drug Detail:Motegrity (Prucalopride [ proo-kal-oh-pride ])
Drug Class: Serotoninergic neuroenteric modulators
Highlights of Prescribing Information
MOTEGRITY (prucalopride) tablets, for oral use
Initial U.S. Approval: 2018
Recent Major Changes
| Warnings and Precautions (5.1) | 11/2020 |
Indications and Usage for Motegrity
MOTEGRITY® is a serotonin-4 (5-HT4) receptor agonist indicated for the treatment of chronic idiopathic constipation (CIC) in adults. (1)
Motegrity Dosage and Administration
- Take with or without food. (2)
- Recommended dosage by patient population:
| Population with CIC | Recommended Oral Dose Regimen |
|---|---|
| Adults | 2 mg once daily. (2) |
| Patients with severe renal impairment (creatinine clearance (CrCL) less than 30 mL/min | 1 mg once daily. (2, 8.5, 8.6) |
Dosage Forms and Strengths
Tablets: 1 mg, 2 mg of prucalopride (3)
Contraindications
- Hypersensitivity to MOTEGRITY(4)
- Intestinal perforation or obstruction due to structural or functional disorder of the gut wall, obstructive ileus, severe inflammatory conditions of the intestinal tract such as Crohn's disease, ulcerative colitis, and toxic megacolon/megarectum. (4)
Warnings and Precautions
Suicidal Ideation and Behavior: Monitor patients for suicidal ideation and behavior as well as self-injurious ideation and new-onset or worsening of depression. Instruct patients to discontinue MOTEGRITY immediately and contact their healthcare provider if they experience any unusual changes in mood or behavior,or they experience emerging suicidal thoughts or behaviors. (5.1)
Adverse Reactions/Side Effects
Most common adverse reactions (≥2%) are headache, abdominal pain, nausea, diarrhea, abdominal distension, dizziness, vomiting, flatulence, and fatigue. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Takeda Pharmaceuticals at 1-800-828-2088 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 11/2020
Related/similar drugs
Linzess, Amitiza, lubiprostone, Trulance, linaclotide, prucaloprideFull Prescribing Information
1. Indications and Usage for Motegrity
MOTEGRITY® is indicated for the treatment of chronic idiopathic constipation (CIC) in adults.
2. Motegrity Dosage and Administration
MOTEGRITY can be taken with or without food. The recommended dosage by patient population is shown in Table 1.
| Population with CIC | Recommended Oral Dose Regimen |
|---|---|
| Adults | 2 mg once daily |
| Patients with severe renal impairment (creatinine clearance (CrCL) less than 30 mL/min) [see Use in Specific Populations (8.5 and 8.6)]. | 1 mg once daily |
3. Dosage Forms and Strengths
MOTEGRITY Tablets:
- 1 mg prucalopride: White to off-white, round, biconvex film-coated tablet debossed with "PRU 1" on one side and no debossing on the other side.
- 2 mg prucalopride: Pink, round, biconvex film-coated tablet debossed with "PRU 2" on one side and no debossing on the other side.
4. Contraindications
MOTEGRITY is contraindicated in patients with:
- A history of hypersensitivity to MOTEGRITY. Reactions including dyspnea, rash, pruritus, urticaria, and facial edema have been observed [(see Adverse Reactions (6.2)].
- Intestinal perforation or obstruction due to structural or functional disorder of the gut wall, obstructive ileus, severe inflammatory conditions of the intestinal tract such as Crohn's disease, ulcerative colitis, and toxic megacolon/megarectum.
5. Warnings and Precautions
5.1 Suicidal Ideation and Behavior
In clinical trials, suicides, suicide attempts, and suicidal ideation have been reported. Postmarketing cases of suicidal ideation and behavior as well as self-injurious ideation and new onset or worsening of depression have been reported within the first few weeks of starting MOTEGRITY [see Adverse Reactions (6.1, 6.2)].
A causal association between treatment with MOTEGRITY and an increased risk of suicidal ideation and behavior has not been established.
Monitor all patients treated with MOTEGRITY for new onset or worsening of depression or the emergence of suicidal thoughts and behaviors. Counsel patients, their caregivers, and family members of patients to be aware of any unusual changes in mood or behavior and alert the healthcare provider. Instruct patients to discontinue MOTEGRITY immediately and contact their healthcare provider if they experience any of these symptoms.
6. Adverse Reactions/Side Effects
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below represent 2530 patients (1251 received MOTEGRITY 2 mg once daily and 1279 received placebo) with CIC from 6 double-blind, placebo-controlled clinical trials of 12 weeks to 24 weeks in duration. In these trials overall, patients were primarily female (76%) and white (76%). The mean age was 47 years (range 17 to 95 years) [see Clinical Studies (14)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of prucalopride. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hypersensitivity reactions: dyspnea, rash, pruritus, urticaria, and facial edema [see Contraindications (4)].
Psychiatric disorders: Suicide, suicide attempts, suicidal ideation, self-injurious ideation, depression, anxiety, insomnia, nightmares, and visual hallucinations [see Warnings and Precautions (5.1)].
8. Use In Specific Populations
8.2 Lactation
Data
In an open-label study in 8 healthy lactating women in the weaning stage, plasma and milk samples were collected at predose (day 1 and 4), and then 2, 4, 8, 12, and 24 hours (day 4) after a 2-mg dose of prucalopride was administered once daily for 4 days. Prucalopride is excreted in breast milk with a milk to plasma AUC ratio of 2.65:1; the average amount passed to the infant was estimated to be 1.74 mcg/kg/day, which is about 6% of the maternal dose, adjusted for body weight. The prucalopride concentration detected in breast milk during weaning may not reflect the prucalopride concentration in breast milk during full milk production.
8.4 Pediatric Use
The safety and effectiveness of MOTEGRITY have not been established in pediatric patients.
8.5 Geriatric Use
Of the 2484 patients treated with MOTEGRITY 1 mg or 2 mg once daily in 6 controlled trials of at least 12-week duration in patients with CIC, 15% were 65 years of age and over, and 5% were 75 years of age and over [see Clinical Studies (14)]. No overall differences in safety and effectiveness were observed between elderly and younger patients.
In an additional 4-week double-blind, placebo-controlled dose escalation study in 89 elderly nursing home residents with CIC (PRU-USA-26, NCT00627692), no unanticipated safety issues were identified.
Elderly subjects had higher prucalopride exposure compared to younger subjects. However, the effect of age on the pharmacokinetics of prucalopride appeared to be related to decreased renal function [see Clinical Pharmacology (12.3)]. Adjust the dosage in elderly patients based on renal function [see Dosage and Administration (2), Use in Specific Populations (8.6)].
8.6 Renal Impairment
No dosage adjustment is required for patients with mild and moderate renal impairment (creatinine clearance at least 30 mL/min, as determined from a 24-hour urine collection in the clinical trial).
MOTEGRITY is known to be substantially excreted by the kidney, and the risk of adverse reactions may be greater in patients with impaired renal function. A decreased dosage is recommended in patients with severe renal impairment (creatinine clearance less than 30 mL/min, as determined from a 24-hour urine collection in the clinical trial) [see Dosage and Administration (2)].
Avoid MOTEGRITY in patients with end-stage renal disease requiring dialysis [see Clinical Pharmacology (12.3)].
10. Overdosage
An overdose may result in appearance of symptoms from an exaggeration of the known pharmacodynamic effects of prucalopride and includes headache, nausea, and diarrhea. Specific treatment is not available for MOTEGRITY overdose. Should an overdose occur, treat symptomatically and institute supportive measures, as required. Extensive fluid loss from diarrhea or vomiting may require correction of electrolyte disturbances.
11. Motegrity Description
MOTEGRITY (prucalopride) tablets for oral use contain prucalopride succinate, a dihydrobenzofurancarboxamide that is a serotonin type 4 (5-HT4) receptor agonist. The IUPAC name is: 4-amino-5-chloro-N-[1-(3-methoxypropyl)piperidin-4-yl]-2,3-dihydrobenzofuran-7-carboxamide succinate.
The molecular formula is C18H26ClN3O3.C4H6O4 and the molecular weight is 485.96. The structural formula is:
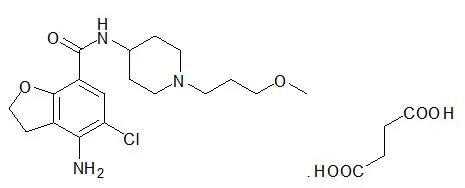
Prucalopride succinate is a white to almost white powder. It is highly soluble in acidic aqueous media and alkaline aqueous media up to a pH of approximately 9.
Each 1-mg film-coated tablet of MOTEGRITY contains 1 mg of prucalopride (equivalent to 1.32 mg prucalopride succinate), and the following inactive ingredients: colloidal silicon dioxide, lactose monohydrate, magnesium stearate, and microcrystalline cellulose. The coating for the 1-mg tablet contains hypromellose, lactose monohydrate, polyethylene glycol 3000, titanium dioxide, and triacetin.
Each 2-mg film-coated tablet of MOTEGRITY contains 2 mg of prucalopride (equivalent to 2.64 mg prucalopride succinate), and the following inactive ingredients: colloidal silicon dioxide, lactose monohydrate, magnesium stearate, and microcrystalline cellulose. The coating for the 2-mg tablet contains hypromellose, lactose monohydrate, polyethylene glycol 3000, titanium dioxide, triacetin, red iron oxide, yellow iron oxide, and FD&C Blue #2.
12. Motegrity - Clinical Pharmacology
12.1 Mechanism of Action
Prucalopride, a selective serotonin type 4 (5-HT4) receptor agonist, is a gastrointestinal (GI) prokinetic agent that stimulates colonic peristalsis (high-amplitude propagating contractions [HAPCs]), which increases bowel motility.
Prucalopride was devoid of effects mediated via 5-HT2A, 5-HT2B, 5-HT3, motilin or CCK-A receptors in vitro at concentrations exceeding 5-HT4 receptor affinity by 150-fold or greater. In isolated GI tissues from various animal species, prucalopride facilitated acetylcholine release to enhance the amplitude of contractions and stimulate peristalsis. In rats and dogs, prucalopride stimulated gastrointestinal motility with contractions starting from the proximal colon to the anal sphincter.
12.3 Pharmacokinetics
The pharmacokinetics of prucalopride has been evaluated in healthy subjects and is dose-proportional within and beyond the therapeutic range (tested up to 20 mg, 10 times the maximum approved recommended dose). Prucalopride administered once daily displays time-independent kinetics during prolonged treatment. With once daily administration of 2 mg prucalopride, pharmacokinetic steady-state is attained within 3 to 4 days, and steady-state plasma concentrations fluctuate between trough and peak values of 2.5 and 7 ng/mL, respectively, with mean plasma AUC0-24h of 109 ng∙h/mL. The accumulation ratio after once daily dosing ranged from 1.9 to 2.3. The terminal half-life is approximately 1 day.
Pharmacokinetic parameters in patients with CIC are similar to those seen in healthy subjects.
13. Nonclinical Toxicology
13.2 Animal Toxicology and/or Pharmacology
In safety pharmacology studies, no relevant effects were observed in any of the cardiovascular studies at concentrations at least 50 times the human therapeutic Cmax. Prucalopride had no effect on potassium current in hERG-transfected HEK cells at concentrations up to 1 micromolar (50 times the human therapeutic Cmax). At concentrations ≥3 micromolar, concentration-dependent inhibition of the current was observed (IC50=22 micromolar; 1100 times the human therapeutic Cmax). In studies in pigs, minor and transient increases in heart rate and blood pressure were noted upon first exposure to prucalopride, at plasma levels at least 10 times the human therapeutic Cmax.
In repeated-dose toxicology studies in male rats, increases in heart weight (up to 9%) were observed at doses of 20 mg/kg/day or higher (at least 75 times the human therapeutic AUC). Cardiac histology revealed an increase in focal infiltration of chronic inflammatory cells in the heart at a dose of 80 mg/kg/day (at least 785 times the human therapeutic AUC). In dogs, no changes in heart rate, blood pressure, electrocardiogram parameters, heart weight, or cardiac histology were observed at any dose tested (the highest dose of 30 mg/kg/day was 572 times the human therapeutic AUC).
In vitro studies demonstrated no effect of prucalopride on either contractile responses in human, canine, and porcine coronary arteries at concentrations up to 10 micromolar (500 times the human clinical Cmax) or on platelet aggregation at concentrations up to 200 nanomolar (10 times the human clinical Cmax).
14. Clinical Studies
The efficacy of MOTEGRITY for the treatment of CIC was evaluated in six double-blind, placebo-controlled, randomized, multicenter clinical trials in 2484 adult patients (Studies 1 to 6; see Table 3). Studies 1 through 5 were 12-week treatment duration and Study 6 included 24 weeks of treatment. Patients less than 65 years were dosed with MOTEGRITY 2 mg once daily. In Studies 2 and 6, the geriatric patients started on MOTEGRITY 1 mg once daily and, if necessary, the dose was increased to 2 mg after 2 or 4 weeks of treatment in the event of insufficient response at 1 mg; of these patients 81% increased to 2 mg. Overall, the majority of patients were female (76%) and white (76%), and also included Asian (19%) and black (3%). The mean adult age was 47±16 years (range 17 to 95 years) and the mean duration of constipation was 16±15 years with 28% of patients having chronic constipation for at least 20 years.
| Study Number | Duration |
|---|---|
| Study 1 (PRU-CRC-3001, NCT01116206) | 12 Weeks |
| Study 2 (SPD555-302, NCT01147926) | 12 Weeks |
| Study 3 (PRU-INT-6, NCT00488137) | 12 Weeks |
| Study 4 (PRU-USA-11, NCT00483886) | 12 Weeks |
| Study 5 (PRU-USA-13, NCT00485940) | 12 Weeks |
| Study 6 (SPD-555-401, NCT01424228) | 24 Weeks |
Eligible patients required a history of chronic constipation defined as having fewer than 3 spontaneous bowel movements (SBMs) per week that resulted in a feeling of complete evacuation (complete, spontaneous bowel movement [CSBM]) and 1 or more of the following symptoms for greater than 25% of bowel movements in the preceding 3 months, with symptoms onset more than 6 months prior to screening:
- Lumpy or hard stools
- Sensation of incomplete evacuation
- Straining at defecation
Patients who never had SBMs were eligible. In Study 1, eligibility also included sensation of ano-rectal obstruction or blockade or the need for digital manipulation in more than 25% of bowel movements. In all studies, patients were excluded if constipation was due to secondary causes or suspected to be drug-induced.
Efficacy was assessed using information provided by patients in a daily diary.
16. How is Motegrity supplied
MOTEGRITY tablets containing 1 mg prucalopride are white to off-white, round, biconvex film-coated tablets debossed with "PRU 1" on one side and no debossing on the other side. They are supplied as:
- NDC 54092-546-01: HDPE bottle of 30 tablets, with child-resistant closure.
MOTEGRITY tablets containing 2 mg prucalopride are pink, round, biconvex film-coated tablets debossed with "PRU 2" on one side and no debossing on the other side. They are supplied as:
- NDC 54092-547-01: HDPE bottle of 30 tablets, with child-resistant closure.
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information)
- Suicidal Ideation and Behavior: Inform patients, their caregivers, and family members that suicidal ideation and behavior, self-injurious ideation as well as new onset or worsening depression have been reported in patients treated with MOTEGRITY. Advise them to be aware of any unusual changes in mood or behavior, new onset or worsening of depression, or the emergence of suicidal thoughts or behavior. Instruct patients, caregivers, and family members to discontinue MOTEGRITY immediately and contact their healthcare provider if any of these symptoms occur [see Warnings and Precautions (5.1)].
- Pregnancy: Advise patients that there is a pregnancy registry that monitors pregnancy outcomes in women exposed to MOTEGRITY during pregnancy [see Use in Specific Populations (8.1)].
| MOTEGRITY
prucalopride tablet, film coated |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| MOTEGRITY
prucalopride tablet, film coated |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| Labeler - Takeda Pharmaceuticals America, Inc. (039997266) |






