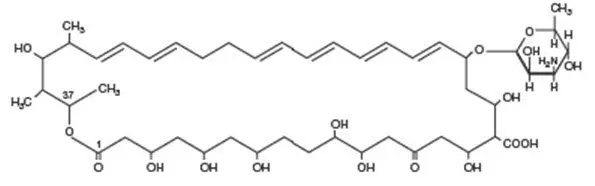
Nystatin Oral Suspension Description
Nystatin is an antimycotic polyene antibiotic obtained from
Streptomyces noursei. Structural formula:
Nystatin Oral Suspension USP, for oral administration, contains 100,000 USP Nystatin Units per mL. Inactive ingredients: alcohol (≤ 1% v/v), artificial peppermint flavor, cherry flavor, citric acid, D&C Yellow No. 10, FD&C Red No. 40, glycerin, magnesium aluminum silicate, methylparaben, potassium phosphate dibasic, propylene glycol, propylparaben, purified water and sucrose.