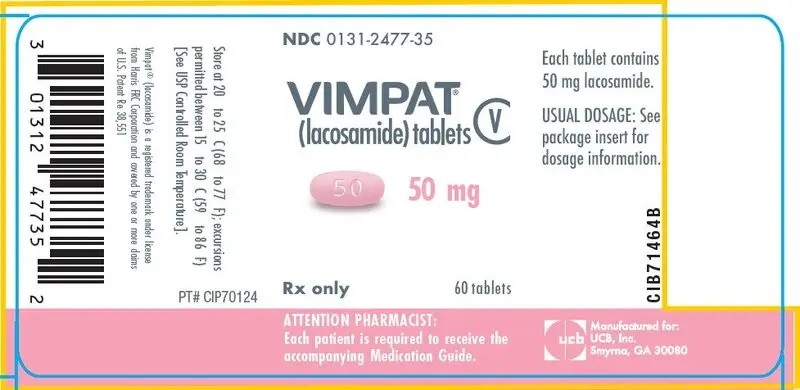
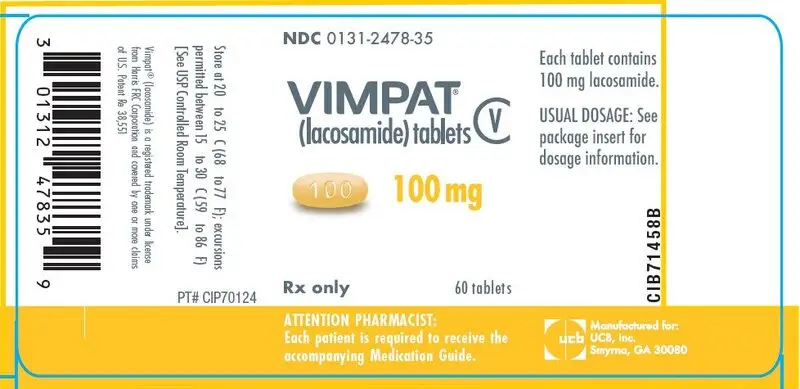
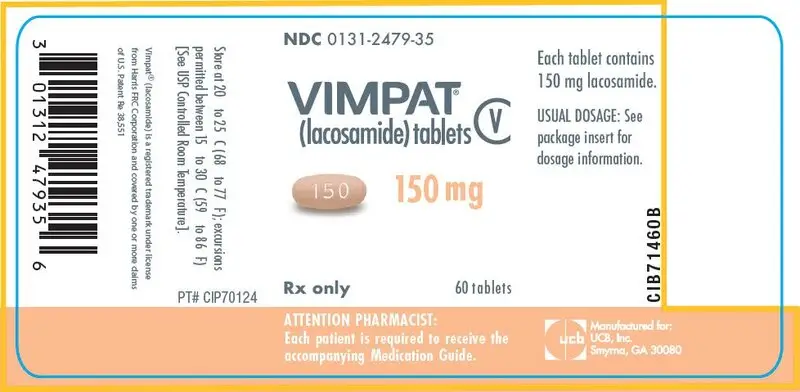
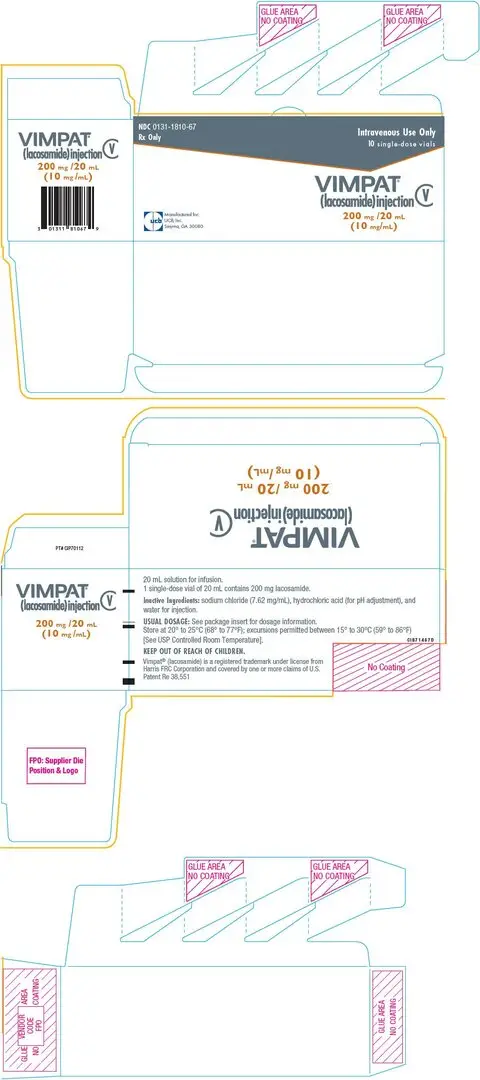
Drug Detail:Vimpat (Lacosamide (oral/injection) [ la-koe-sa-mide ])
Drug Class: Miscellaneous anticonvulsants
Highlights of Prescribing Information
VIMPAT® (lacosamide) film coated tablet, for oral use, CV
VIMPAT® (lacosamide) injection, for intravenous use, CV
VIMPAT® (lacosamide) oral solution, CV
Initial U.S. Approval: 2008
Recent Major Changes
| Dosage and Administration (2.1, 2.2) | 4/2023 |
Indications and Usage for Vimpat
VIMPAT is indicated for:
- Treatment of partial-onset seizures in patients 1 month of age and older (1.1)
- Adjunctive therapy in the treatment of primary generalized tonic-clonic seizures in patients 4 years of age and older (1.2)
Vimpat Dosage and Administration
-
Adults (17 years and older):
- Initial dosage for monotherapy for the treatment of partial-onset seizures is 100 mg twice daily (2.1)
- Initial dosage for adjunctive therapy for the treatment of partial-onset seizures or primary generalized tonic-clonic seizures is 50 mg twice daily (2.1)
- Maximum recommended dosage for monotherapy and adjunctive therapy is 200 mg twice daily (2.1)
- Pediatric Patients 1 month to less than 17 years: The recommended dosage is based on body weight and is administered orally twice daily (2.1)
- Increase dosage based on clinical response and tolerability, no more frequently than once per week (2.1)
- Injection: for intravenous use only when oral administration is temporarily not feasible; the recommended dosage is based on body weight and is administered two or three times daily over 15 to 60 minutes; obtaining ECG before initiation is recommended in certain patients (2.7, 5.3)
- Dose adjustment is recommended for severe renal impairment (2.4, 12.3)
- Dose adjustment is recommended for mild or moderate hepatic impairment; use in patients with severe hepatic impairment is not recommended (2.5, 12.3)
Dosage Forms and Strengths
- 50 mg, 100 mg, 150 mg, 200 mg tablets (3)
- 200 mg/20 mL single-dose vial for intravenous use (3)
- 10 mg/mL oral solution (3)
Contraindications
None (4)
Warnings and Precautions
- Monitor patients for suicidal behavior and ideation (5.1)
- VIMPAT may cause dizziness and ataxia (5.2)
- Cardiac Rhythm and Conduction Abnormalities: Obtaining ECG before beginning and after titration to steady-state maintenance is recommended in patients with underlying proarrhythmic conditions or on concomitant medications that affect cardiac conduction; closely monitor these patients (5.3, 7.2)
- VIMPAT may cause syncope (5.4)
- VIMPAT should be gradually withdrawn to minimize the potential of increased seizure frequency (5.5)
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/ Multi-Organ Hypersensitivity: Discontinue if no alternate etiology (5.6)
Adverse Reactions/Side Effects
- Adjunctive therapy: Most common adverse reactions in adults (≥10% and greater than placebo) are diplopia, headache, dizziness, nausea, and somnolence (6.1)
- Monotherapy: Most common adverse reactions are similar to those seen in adjunctive therapy studies (6.1)
- Pediatric patients: Adverse reactions are similar to those seen in adult patients (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact UCB, Inc. at 1-844-599-2273 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Use In Specific Populations
- Pregnancy: Based on animal data, may cause fetal harm (8.1)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 4/2023
Full Prescribing Information
1. Indications and Usage for Vimpat
2. Vimpat Dosage and Administration
2.1 Dosage Information
The recommended dosage for monotherapy and adjunctive therapy for partial-onset seizures in patients 1 month of age and older and for adjunctive therapy for primary generalized tonic-clonic seizures in patients 4 years of age and older is included in Table 1. In pediatric patients, the recommended dosing regimen is dependent upon body weight. Dosage should be increased based on clinical response and tolerability, no more frequently than once per week. Titration increments should not exceed those shown in Table 1.
| Age and Body Weight | Initial Dosage | Titration Regimen | Maintenance Dosage |
|---|---|---|---|
|
|||
| Adults (17 years and older) | Monotherapy†:
100 mg twice daily (200 mg per day) Adjunctive Therapy: 50 mg twice daily (100 mg per day) | Increase by 50 mg twice daily (100 mg per day) every week | Monotherapy†:
150 mg to 200 mg twice daily (300 mg to 400 mg per day) Adjunctive Therapy: 100 mg to 200 mg twice daily (200 mg to 400 mg per day) |
| Pediatric patients weighing at least 50 kg | 50 mg twice daily (100 mg per day) | Increase by 50 mg twice daily (100 mg per day) every week | Monotherapy†:
150 mg to 200 mg twice daily (300 mg to 400 mg per day) Adjunctive Therapy: 100 mg to 200 mg twice daily (200 mg to 400 mg per day) |
| Pediatric patients weighing 30 kg to less than 50 kg | 1 mg/kg twice daily (2 mg/kg/day) | Increase by 1 mg/kg twice daily (2 mg/kg/day) every week | 2 mg/kg to 4 mg/kg twice daily (4 mg/kg/day to 8 mg/kg/day) |
| Pediatric patients weighing 11 kg to less than 30 kg | 1 mg/kg twice daily (2 mg/kg/day) | Increase by 1 mg/kg twice daily (2 mg/kg/day) every week | 3 mg/kg to 6 mg/kg twice daily (6 mg/kg/day to 12 mg/kg/day) |
| Pediatric patients weighing 6 kg to less than 11 kg ‡ | |||
| Pediatric patients weighing less than 6 kg ‡ | Intravenous:
0.66 mg/kg three times daily (2 mg/kg/day) | Intravenous:
Increase by 0.66 mg/kg three times daily (2 mg/kg/day) every week | Intravenous:
2.5 mg/kg to 5 mg/kg three times daily (7.5 mg/kg/day to 15 mg/kg/day) |
| Oral:
1 mg/kg twice daily (2 mg/kg/day) | Oral:
Increase by 1 mg/kg twice daily (2 mg/kg/day) every week | Oral:
3.75 mg/kg to 7.5 mg/kg twice daily (7.5 mg/kg/day to 15 mg/kg/day) |
|
In adjunctive clinical trials in adult patients with partial-onset seizures, a dosage higher than 200 mg twice daily (400 mg per day) was not more effective and was associated with a substantially higher rate of adverse reactions [see Adverse Reactions (6.1) and Clinical Studies (14.2)].
2.2 Alternate Initial Dosage Information to Achieve the Maintenance Dosage in a Shorter Timeframe
For monotherapy and adjunctive therapy for partial-onset seizures in patients 1 month of age and older and for adjunctive therapy for primary generalized tonic-clonic seizures in patients 4 years of age and older, an alternate initial dosing regimen for week 1 (e.g., including a loading dose and/or a higher initial dosage) may be administered in patients for whom achieving the recommended maintenance dosage in a shorter timeframe is clinically indicated (see Table 2). The alternate initial dosage regimen should be continued for one week. VIMPAT may then be titrated based on clinical response and tolerability, no more frequently than once per week, if needed. The loading dose should be administered with medical supervision because of the possibility of increased incidence of adverse reactions, including central nervous system (CNS) and cardiovascular adverse reactions [see Warnings and Precautions (5.2, 5.3), Adverse Reactions (6.1), and Clinical Pharmacology (12.3)]. Titration increments should not exceed those shown in Table 2.
| Age and Body Weight | Alternate Initial Dosage | Titration Regimen | Maintenance Dosage |
|---|---|---|---|
|
|||
| Adults (17 years and older) | Single loading dose:
200 mg 12 hours later initiate: 100 mg twice daily (200 mg per day) | Increase by 50 mg twice daily (100 mg per day) at weekly intervals, if needed | Monotherapy†:
150 mg to 200 mg twice daily (300 mg to 400 mg per day) Adjunctive Therapy: 100 mg to 200 mg twice daily (200 mg to 400 mg per day) |
| Pediatric patients weighing at least 50 kg | Single loading dose:
200 mg 12 hours later initiate: 100 mg twice daily (200 mg per day) | Increase by 50 mg twice daily (100 mg per day) at weekly intervals, if needed | Monotherapy†:
150 mg to 200 mg twice daily (300 mg to 400 mg per day) Adjunctive Therapy: 100 mg to 200 mg twice daily (200 mg to 400 mg per day) |
| Pediatric patients weighing 30 kg to less than 50 kg | Single loading dose:
4 mg/kg 12 hours later initiate: 2 mg/kg twice daily (4 mg/kg/day) | Increase by 1 mg/kg twice daily (2 mg/kg/day) at weekly intervals, if needed | 2 mg/kg to 4 mg/kg twice daily (4 mg/kg/day to 8 mg/kg/day) |
| Pediatric patients weighing 11 kg to less than 30 kg | Single loading dose:
4.5 mg/kg 12 hours later initiate: 3 mg/kg twice daily (6 mg/kg/day) | Increase by 1 mg/kg twice daily (2 mg/kg/day) at weekly intervals, if needed | 3 mg/kg to 6 mg/kg twice daily (6 mg/kg/day to 12 mg/kg/day) |
| Pediatric patients weighing 6 kg to less than 11 kg ‡ | |||
| Pediatric patients weighing less than 6 kg ‡ | Intravenous:
No loading dose required 2.5 mg/kg three times daily (7.5 mg per day) | Intravenous:
Increase by 0.66 mg/kg three times daily (2 mg/kg/day) at weekly intervals, if needed | Intravenous:
2.5 mg/kg to 5 mg/kg three times daily (7.5 mg/kg/day to 15 mg/kg/day) |
| Oral:
No loading dose required 3.75 mg/kg twice daily (7.5 mg per day) | Oral:
Increase by 1 mg/kg twice daily (2 mg/kg/day) at weekly intervals, if needed | Oral:
3.75 mg/kg to 7.5 mg/kg twice daily (7.5 mg/kg/day to 15 mg/kg/day) |
|
2.3 Converting From a Single Antiepileptic (AED) to VIMPAT Monotherapy for the Treatment of Partial-Onset Seizures
For patients who are already on a single AED and will convert to VIMPAT monotherapy, withdrawal of the concomitant AED should not occur until the therapeutic dosage of VIMPAT is achieved and has been administered for at least 3 days. A gradual withdrawal of the concomitant AED over at least 6 weeks is recommended.
2.4 Dosage Information for Patients with Renal Impairment
For patients with mild to moderate renal impairment, no dosage adjustment is necessary. For patients with severe renal impairment [creatinine clearance (CLCR) less than 30 mL/min as estimated by the Cockcroft-Gault equation for adults; CLCR less than 30 mL/min/1.73m2 as estimated by the Schwartz equation for pediatric patients] or end-stage renal disease, a reduction of 25% of the maximum dosage is recommended.
In all patients with renal impairment, dose initiation and titration should be based on clinical response and tolerability.
2.5 Dosage Information for Patients with Hepatic Impairment
For patients with mild or moderate hepatic impairment, a reduction of 25% of the maximum dosage is recommended. The dose initiation and titration should be based on clinical response and tolerability in patients with hepatic impairment.
VIMPAT use is not recommended in patients with severe hepatic impairment.
2.6 Administration Instructions for VIMPAT Tablets and Oral Solution
VIMPAT tablets and oral solution may be taken with or without food.
5. Warnings and Precautions
5.1 Suicidal Behavior and Ideation
Antiepileptic drugs (AEDs), including VIMPAT, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted Relative Risk 1.8, 95% CI:1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated.There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number of events is too small to allow any conclusion about drug effect on suicide.
The increased risk of suicidal thoughts or behavior with AEDs was observed as early as one week after starting treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5-100 years) in the clinical trials analyzed.
Table 3 shows absolute and relative risk by indication for all evaluated AEDs.
| Indication | Placebo Patients with Events Per 1000 Patients | Drug Patients with Events Per 1000 Patients | Relative Risk: Incidence of Events in Drug Patients/Incidence in Placebo Patients | Risk Difference: Additional Drug Patients with Events Per 1000 Patients |
|---|---|---|---|---|
| Epilepsy | 1.0 | 3.4 | 3.5 | 2.4 |
| Psychiatric | 5.7 | 8.5 | 1.5 | 2.9 |
| Other | 1.0 | 1.8 | 1.9 | 0.9 |
| Total | 2.4 | 4.3 | 1.8 | 1.9 |
The relative risk for suicidal thoughts or behavior was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar.
Anyone considering prescribing VIMPAT or any other AED must balance this risk with the risk of untreated illness. Epilepsy and many other illnesses for which antiepileptics are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
5.2 Dizziness and Ataxia
VIMPAT may cause dizziness and ataxia in adult and pediatric patients. In adult patients with partial-onset seizures taking 1 to 3 concomitant AEDs, dizziness was experienced by 25% of patients randomized to the recommended doses (200 to 400 mg/day) of VIMPAT (compared with 8% of placebo patients) and was the adverse reaction most frequently leading to discontinuation (3%). Ataxia was experienced by 6% of patients randomized to the recommended doses (200 to 400 mg/day) of VIMPAT (compared to 2% of placebo patients). The onset of dizziness and ataxia was most commonly observed during titration. There was a substantial increase in these adverse reactions at doses higher than 400 mg/day [see Adverse Reactions (6.1)]. If a loading dose is clinically indicated, administer with medical supervision because of the possibility of increased incidence of adverse reactions, including CNS adverse reactions such as dizziness and ataxia.
5.4 Syncope
In the short-term controlled trials of VIMPAT in adult patients with partial-onset seizures with no significant system illnesses, there was no increase in syncope compared to placebo. In the short-term controlled trials in adult patients with diabetic neuropathy, for which VIMPAT is not indicated, 1.2% of patients who were treated with VIMPAT reported an adverse reaction of syncope or loss of consciousness, compared with 0% of placebo-treated patients with diabetic neuropathy. Most of the cases of syncope were observed in patients receiving doses above 400 mg/day. The cause of syncope was not determined in most cases. However, several were associated with either changes in orthostatic blood pressure, atrial flutter/fibrillation (and associated tachycardia), or bradycardia. Cases of syncope have also been observed in open-label clinical partial-onset seizure studies in adult and pediatric patients. These cases were associated with a history of risk factors for cardiac disease and the use of drugs that slow AV conduction.
5.5 Withdrawal of Antiepileptic Drugs (AEDs)
As with all AEDs, VIMPAT should be withdrawn gradually (over a minimum of 1 week) to minimize the potential of increased seizure frequency in patients with seizure disorders.
5.6 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multi-Organ Hypersensitivity
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), also known as multi-organ hypersensitivity, has been reported in patients taking antiepileptic drugs, including VIMPAT. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy and/or facial swelling, in association with other organ system involvement, such as hepatitis, nephritis, hematologic abnormalities, myocarditis, or myositis, sometimes resembling an acute viral infection. Eosinophilia is often present. This disorder is variable in its expression, and other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity (e.g., fever, lymphadenopathy) may be present even though rash is not evident. If such signs or symptoms are present, the patient should be evaluated immediately. VIMPAT should be discontinued if an alternative etiology for the signs or symptoms cannot be established.
5.7 Risks in Patients with Phenylketonuria
Phenylalanine can be harmful in patients with phenylketonuria (PKU). VIMPAT oral solution contains aspartame, a source of phenylalanine. A 200 mg dose of VIMPAT oral solution (equivalent to 20 mL) contains 0.32 mg of phenylalanine. Before prescribing VIMPAT oral solution to a patient with PKU, consider the combined daily amount of phenylalanine from all sources, including VIMPAT oral solution.
6. Adverse Reactions/Side Effects
The following serious adverse reactions are described below and elsewhere in the labeling:
- Suicidal Behavior and Ideation [see Warnings and Precautions (5.1)]
- Dizziness and Ataxia [see Warnings and Precautions (5.2)]
- Cardiac Rhythm and Conduction Abnormalities [see Warnings and Precautions (5.3)]
- Syncope [see Warnings and Precautions (5.4)]
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity Reactions [see Warnings and Precautions (5.6)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
VIMPAT Tablet and Oral Solution in Adults
In the premarketing development of adjunctive therapy for partial-onset seizures, 1327 adult patients received VIMPAT tablets in controlled and uncontrolled trials, of whom 1000 were treated for longer than 6 months, and 852 for longer than 12 months. The monotherapy development program for partial-onset seizures included 425 adult patients, 310 of whom were treated for longer than 6 months, and 254 for longer than 12 months.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of VIMPAT. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: Agranulocytosis
Psychiatric disorders: Aggression, agitation, hallucination, insomnia, psychotic disorder
Skin and subcutaneous tissue disorders: Angioedema, rash, urticaria, Stevens-Johnson syndrome, toxic epidermal necrolysis.
Neurologic disorders: Dyskinesia, new or worsening seizures
7. Drug Interactions
7.1 Strong CYP3A4 or CYP2C9 Inhibitors
Patients with renal or hepatic impairment who are taking strong inhibitors of CYP3A4 and CYP2C9 may have a significant increase in exposure to VIMPAT. Dose reduction may be necessary in these patients.
7.2 Concomitant Medications that Affect Cardiac Conduction
VIMPAT should be used with caution in patients on concomitant medications that affect cardiac conduction (sodium channel blockers, beta-blockers, calcium channel blockers, potassium channel blockers) including those that prolong PR interval (including sodium channel blocking AEDs), because of a risk of AV block, bradycardia, or ventricular tachyarrhythmia. In such patients, obtaining an ECG before beginning VIMPAT, and after VIMPAT is titrated to steady-state, is recommended. In addition, these patients should be closely monitored if they are administered VIMPAT through the intravenous route [see Warnings and Precautions (5.3)].
8. Use In Specific Populations
8.5 Geriatric Use
There were insufficient numbers of elderly patients enrolled in partial-onset seizure trials (n=18) to adequately determine whether they respond differently from younger patients.
No VIMPAT dose adjustment based on age is necessary. In elderly patients, dose titration should be performed with caution, usually starting at the lower end of the dosing range, reflecting the greater frequency of decreased hepatic function, decreased renal function, increased cardiac conduction abnormalities, and polypharmacy [see Dosage and Administration (2.1, 2.4, 2.5) and Clinical Pharmacology (12.3)].
8.6 Renal Impairment
No dose adjustment is necessary in patients with mild to moderate renal impairment (CLCR ≥30 mL/min). In patients with severe renal impairment (CLCR <30 mL/min as estimated by the Cockcroft-Gault equation for adults; CLCR <30 mL/min/1.73m2 as estimated by the Schwartz equation for pediatric patients) and in those with end-stage renal disease, a reduction of 25% of the maximum dosage is recommended [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)].
In all patients with renal impairment, dose initiation and titration should be based on clinical response and tolerability.
VIMPAT is effectively removed from plasma by hemodialysis. Dosage supplementation of up to 50% following hemodialysis should be considered.
8.7 Hepatic Impairment
For adult and pediatric patients with mild to moderate hepatic impairment, a reduction of 25% of the maximum dosage is recommended. Patients with mild to moderate hepatic impairment should be observed closely for adverse reactions, and dose initiation and titration should be based on clinical response and tolerability [see Dosage and Administration (2.5), Clinical Pharmacology (12.3)].
The pharmacokinetics of lacosamide has not been evaluated in severe hepatic impairment. VIMPAT use is not recommended in patients with severe hepatic impairment.
9. Drug Abuse and Dependence
9.2 Abuse
Abuse is the intentional, non-therapeutic use of a drug, even once, for its desirable psychological or physiological effects. In a human abuse potential study, single doses of 200 mg (equal to the maximum single dosage) and 800 mg lacosamide (equal to twice the recommended daily maintenance dosage) produced euphoria-type subjective responses that differentiated statistically from placebo; at 800 mg, these euphoria-type responses were statistically indistinguishable from those produced by alprazolam, a Schedule IV drug. The duration of the euphoria-type responses following lacosamide was less than that following alprazolam. A high rate of euphoria was also reported as an adverse event in the human abuse potential study following single doses of 800 mg lacosamide (15% [5/34]) compared to placebo (0%) and in two pharmacokinetic studies following single and multiple doses of 300-800 mg lacosamide (ranging from 6% [2/33] to 25% [3/12]) compared to placebo (0%). However, the rate of euphoria reported as an adverse event in the VIMPAT development program at therapeutic doses was less than 1%.
9.3 Dependence
Physical dependence is a state that develops as a result of physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug. Abrupt termination of lacosamide in clinical trials with diabetic neuropathic pain patients produced no signs or symptoms that are associated with a withdrawal syndrome indicative of physical dependence. However, psychological dependence cannot be excluded due to the ability of lacosamide to produce euphoria-type adverse events in humans.
10. Overdosage
Events reported after an intake of more than 800 mg (twice the maximum recommended daily dosage) of VIMPAT include dizziness, nausea, and seizures (generalized tonic-clonic seizures, status epilepticus). Cardiac conduction disorders, confusion, decreased level of consciousness, cardiogenic shock, cardiac arrest, and coma have also been observed. Fatalities have occurred following lacosamide overdoses of several grams.
There is no specific antidote for overdose with VIMPAT. Standard decontamination procedures should be followed. General supportive care of the patient is indicated including monitoring of vital signs and observation of the clinical status of patient. A Certified Poison Control Center should be contacted for up to date information on the management of overdose with VIMPAT.
Standard hemodialysis procedures result in significant clearance of VIMPAT (reduction of systemic exposure by 50% in 4 hours). Hemodialysis may be indicated based on the patient's clinical state or in patients with significant renal impairment.
11. Vimpat Description
The chemical name of lacosamide, the single (R)-enantiomer, is (R)-2-acetamido-N-benzyl-3-methoxypropionamide (IUPAC). Lacosamide is a functionalized amino acid. Its molecular formula is C13H18N2O3 and its molecular weight is 250.30. The chemical structure is:
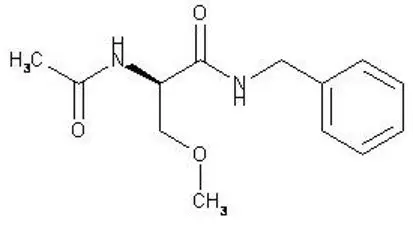
Lacosamide is a white to light yellow powder. It is sparingly soluble in water and slightly soluble in acetonitrile and ethanol.
11.1 VIMPAT Tablets
VIMPAT tablets for oral administration contain lacosamide and the following inactive ingredients: colloidal silicon dioxide, crospovidone, hydroxypropylcellulose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, talc, titanium dioxide, and dye pigments as specified below:
VIMPAT tablets are supplied as debossed tablets and contain the following coloring agents:
50 mg tablets: red iron oxide, black iron oxide, FD&C Blue #2/indigo carmine aluminum lake
100 mg tablets: yellow iron oxide
150 mg tablets: yellow iron oxide, red iron oxide, black iron oxide
200 mg tablets: FD&C Blue #2/indigo carmine aluminum lake
11.2 VIMPAT Injection
VIMPAT injection is a clear, colorless, sterile solution containing 10 mg lacosamide per mL for intravenous infusion. One 20-mL vial contains 200 mg of lacosamide drug substance. The inactive ingredients are sodium chloride (7.62 mg/mL) and water for injection. Hydrochloric acid is used for pH adjustment. VIMPAT injection has a pH of 3.8 to 5.0.
11.3 VIMPAT Oral Solution
VIMPAT oral solution contains 10 mg of lacosamide per mL. The inactive ingredients are purified water, sorbitol solution, glycerin, polyethylene glycol, carboxymethylcellulose sodium, acesulfame potassium, methylparaben, flavoring (including natural and artificial flavors, propylene glycol, aspartame, and maltol), anhydrous citric acid and sodium chloride.
12. Vimpat - Clinical Pharmacology
12.1 Mechanism of Action
The precise mechanism by which VIMPAT exerts its antiepileptic effects in humans remains to be fully elucidated. In vitro electrophysiological studies have shown that lacosamide selectively enhances slow inactivation of voltage-gated sodium channels, resulting in stabilization of hyperexcitable neuronal membranes and inhibition of repetitive neuronal firing.
12.2 Pharmacodynamics
A pharmacokinetic-pharmacodynamic (efficacy) analysis was performed based on the pooled data from the 3 efficacy trials for partial-onset seizures. Lacosamide exposure is correlated with the reduction in seizure frequency. However, doses above 400 mg/day do not appear to confer additional benefit in group analyses.
12.3 Pharmacokinetics
The pharmacokinetics of VIMPAT have been studied in healthy adult subjects (age range 18 to 87), adults with partial-onset seizures, adults with diabetic neuropathy, and subjects with renal and hepatic impairment.
The pharmacokinetics of VIMPAT are similar in healthy subjects, patients with partial-onset seizures, and patients with primary generalized tonic-clonic seizures.
VIMPAT is completely absorbed after oral administration with negligible first-pass effect with a high absolute bioavailability of approximately 100%. The maximum lacosamide plasma concentrations occur approximately 1-to-4-hour post-dose after oral dosing, and elimination half-life is approximately 13 hours. Steady state plasma concentrations are achieved after 3 days of twice daily repeated administration. Pharmacokinetics of VIMPAT are dose proportional (100-800 mg) and time invariant, with low inter- and intra-subject variability. Compared to lacosamide the major metabolite, O-desmethyl metabolite, has a longer Tmax (0.5 to 12 hours) and elimination half-life (15-23 hours).
14. Clinical Studies
14.1 Monotherapy in Patients with Partial-Onset Seizures
The efficacy of VIMPAT in monotherapy was established in a historical-control, multicenter, randomized trial that included 425 patients, age 16 to 70 years, with partial-onset seizures (Study 1). To be included in Study 1, patients were required to be taking stable doses of 1 or 2 marketed antiepileptic drugs. This treatment continued into the 8 week baseline period. To remain in the study, patients were required to have at least 2 partial-onset seizures per 28 days during the 8 week baseline period. The baseline period was followed by a 3 week titration period, during which VIMPAT was added to the ongoing antiepileptic regimen. This was followed by a 16-week maintenance period (i.e., a 6-week withdrawal period for background antiepileptic drugs, followed by a 10-week monotherapy period). Patients were randomized 3 to 1 to receive VIMPAT 400 mg/day or VIMPAT 300 mg/day. Treatment assignments were blinded. Response to treatment was based upon a comparison of the number of patients who met exit criteria during the maintenance phase, compared to historical controls. The historical control consisted of a pooled analysis of the control groups from 8 studies of similar design, which utilized a sub-therapeutic dose of an antiepileptic drug. Statistical superiority to the historical control was considered to be demonstrated if the upper limit from a 2-sided 95% confidence interval for the percentage of patients meeting exit criteria in patients receiving VIMPAT remained below the lower 95% prediction limit of 65% derived from the historical control data.
The exit criteria were one or more of the following: (1) doubling of average monthly seizure frequency during any 28 consecutive days, (2) doubling of highest consecutive 2-day seizure frequency, (3) occurrence of a single generalized tonic-clonic seizure, (4) clinically significant prolongation or worsening of overall seizure duration, frequency, type or pattern considered by the investigator to require trial discontinuation, (5) status epilepticus or new onset of serial/cluster seizures. The study population profile appeared comparable to that of the historical control population.
For the VIMPAT 400 mg/day group, the estimate of the percentage of patients meeting at least 1 exit criterion was 30% (95% CI: 25%, 36%). The upper limit of the 2-sided 95% CI (36%) was below the threshold of 65% derived from the historical control data, meeting the pre-specified criteria for efficacy. VIMPAT 300 mg/day also met the pre-specified criteria for efficacy.
14.2 Adjunctive Therapy in Patients with Partial-Onset Seizures
The efficacy of VIMPAT as adjunctive therapy in partial-onset seizures was established in three 12-week, randomized, double-blind, placebo-controlled, multicenter trials in adult patients (Study 2, Study 3, and Study 4). Enrolled patients had partial-onset seizures with or without secondary generalization, and were not adequately controlled with 1 to 3 concomitant AEDs. During an 8-week baseline period, patients were required to have an average of ≥4 partial-onset seizures per 28 days with no seizure-free period exceeding 21 days. In these 3 trials, patients had a mean duration of epilepsy of 24 years and a median baseline seizure frequency ranging from 10 to 17 per 28 days. 84% of patients were taking 2 to 3 concomitant AEDs with or without concurrent vagal nerve stimulation.
Study 2 compared doses of VIMPAT 200, 400, and 600 mg/day with placebo. Study 3 compared doses of VIMPAT 400 and 600 mg/day with placebo. Study 4 compared doses of VIMPAT 200 and 400 mg/day with placebo. In all three trials, following an 8-week baseline phase to establish baseline seizure frequency prior to randomization, patients were randomized and titrated to the randomized dose (a 1-step back-titration of VIMPAT 100 mg/day or placebo was allowed in the case of intolerable adverse reactions at the end of the titration phase). During the titration phase, in all 3 adjunctive therapy trials, treatment was initiated at 100 mg/day (50 mg twice daily), and increased in weekly increments of 100 mg/day to the target dose. The titration phase lasted 6 weeks in Study 2 and Study 3, and 4 weeks in Study 4. In all three trials, the titration phase was followed by a maintenance phase that lasted 12 weeks, during which patients were to remain on a stable dose of VIMPAT.
A reduction in 28-day seizure frequency (baseline to maintenance phase), as compared to the placebo group, was the primary variable in all three adjunctive therapy trials. A statistically significant effect was observed with VIMPAT treatment (Figure 1) at doses of 200 mg/day (Study 4), 400 mg/day (Studies 2, 3, and 4), and 600 mg/day (Studies 2 and 3).
Subset evaluations of VIMPAT demonstrate no important differences in seizure control as a function of gender or race, although data on race was limited (about 10% of patients were non-Caucasian).
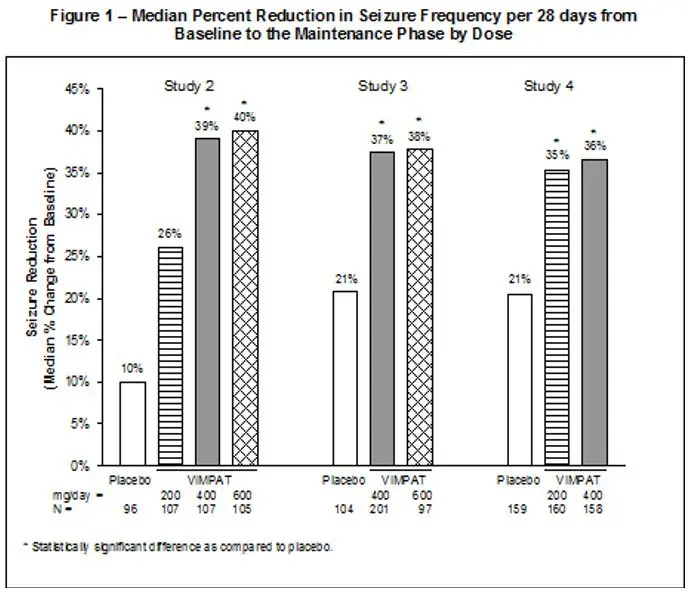
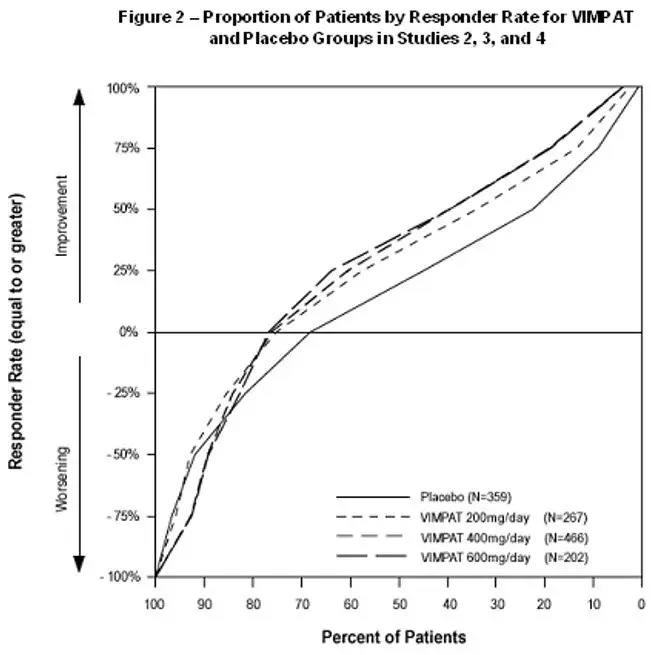
Figure 2 presents the percentage of patients (X-axis) with a percent reduction in partial seizure frequency (responder rate) from baseline to the maintenance phase at least as great as that represented on the Y-axis. A positive value on the Y-axis indicates an improvement from baseline (i.e., a decrease in seizure frequency), while a negative value indicates a worsening from baseline (i.e., an increase in seizure frequency). Thus, in a display of this type, a curve for an effective treatment is shifted to the left of the curve for placebo. The proportion of patients achieving any particular level of reduction in seizure frequency was consistently higher for the VIMPAT groups, compared to the placebo group. For example, 40% of patients randomized to VIMPAT (400 mg/day) experienced a 50% or greater reduction in seizure frequency, compared to 23% of patients randomized to placebo. Patients with an increase in seizure frequency >100% are represented on the Y-axis as equal to or greater than -100%.
14.3 Adjunctive Therapy in Patients with Primary Generalized Tonic-Clonic Seizures
The efficacy of VIMPAT as adjunctive therapy in patients 4 years of age and older with idiopathic generalized epilepsy experiencing primary generalized tonic-clonic (PGTC) seizures was established in a 24-week double-blind, randomized, placebo-controlled, parallel-group, multi-center study (Study 5). The study consisted of a 12-week historical baseline period, a 4-week prospective baseline period, and a 24-week treatment period (which included a 6-week titration period and an 18-week maintenance period). Eligible patients on a stable dose of 1 to 3 antiepileptic drugs experiencing at least 3 documented PGTC seizures during the 16-week combined baseline period were randomized 1:1 to receive VIMPAT (n=121) or placebo (n=121).
Patients were dosed on a fixed-dose regimen. Dosing was initiated at a dose of 2 mg/kg/day in patients weighing less than 50 kg or 100 mg/day in patients weighing 50 kg or more in 2 divided doses. During the titration period, VIMPAT doses were adjusted in 2 mg/kg/day increments in patients weighing less than 50 kg or 100 mg/day in patients weighing 50 kg or more at weekly intervals to achieve the target maintenance period dose of 12 mg/kg/day in patients weighing less than 30 kg, 8 mg/kg/day in patients weighing from 30 to less than 50 kg, or 400 mg/day in patients weighing 50 kg or more.
The primary efficacy endpoint (patients in the modified full analysis set: VIMPAT n=118, placebo n=121) was the time to second PGTC seizure during the 24-week treatment period (Figure 3). The risk of developing a second PGTC seizure was statistically significantly lower in VIMPAT group than in the placebo group during the 24-week treatment period (hazard ratio=0.548, 95% CI of hazard ratio: 0.381, 0.788, p-value = 0.001), with the corresponding risk reduction being 45.2%.
The key secondary efficacy endpoint was the percentage of patients not experiencing a PGTC seizure during the 24-week treatment period. The adjusted Kaplan-Meier estimates of 24-week freedom from PGTC seizures were 31.3% in VIMPAT group and 17.2% in placebo group. The adjusted difference between treatment groups was 14.1% (95% CI: 3.2, 25.1, p-value=0.011).
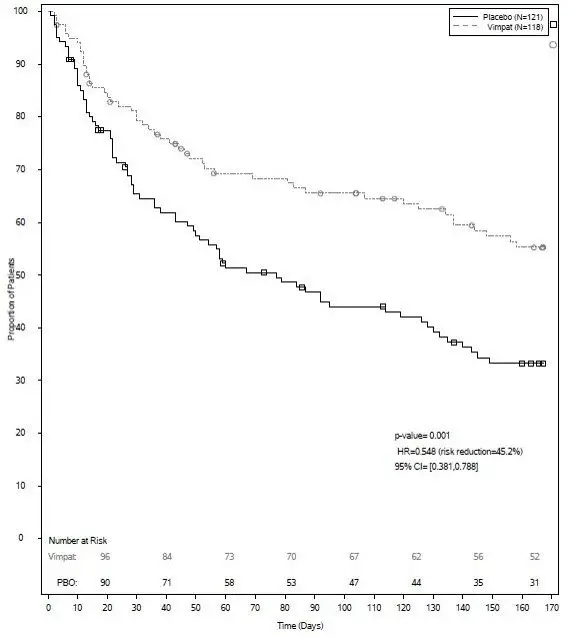 |
| The numbers at the bottom of the figure are for patients still at risk in the study at a given timepoint (i.e., the continuing patients in the study without an event or censoring prior to the timepoint). |
16. How is Vimpat supplied
16.2 Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F). [See USP Controlled Room Temperature]
Do not freeze VIMPAT injection or oral solution. Discard any unused VIMPAT oral solution remaining after six (6) months of first opening the bottle.
17. Patient Counseling Information
Advise the patient or caregiver to read the FDA-approved patient labeling (Medication Guide). The Medication Guide accompanies the product and can also be accessed on www.vimpat.com or by calling 1-844-599-2273.
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Revised: 9/2022 | ||
| MEDICATION GUIDE VIMPAT (VIM-păt) (lacosamide) film coated tablet, for oral use, CV VIMPAT (VIM-păt) (lacosamide) injection, for intravenous use, CV VIMPAT (VIM-păt) (lacosamide) oral solution, CV |
|||
| Read this Medication Guide before you start taking VIMPAT and each time you get a refill. There may be new information. This Medication Guide describes important safety information about VIMPAT. This information does not take the place of talking to your healthcare provider about your medical condition or treatment. | |||
| What is the most important information I should know about VIMPAT? Do not stop taking VIMPAT without first talking to your healthcare provider. Stopping VIMPAT suddenly can cause serious problems. Stopping seizure medicine suddenly in a patient who has epilepsy can cause seizures that will not stop (status epilepticus). VIMPAT can cause serious side effects, including:
|
|||
|
|
||
| How can I watch for early symptoms of suicidal thoughts and actions? | |||
|
|||
|
|||
|
|
||
| If you have fainted or feel like you are going to faint you should lay down with your legs raised. | |||
|
|||
| What is VIMPAT? VIMPAT is a prescription medicine used:
It is not known if VIMPAT is safe and effective for partial-onset seizures in children under 1 month of age or for primary generalized tonic-clonic seizures in children under 4 years of age. |
|||
| What should I tell my healthcare provider before taking VIMPAT? Before you take VIMPAT, tell your healthcare provider about all of your medical conditions, including if you:
Taking VIMPAT with certain other medicines may cause side effects or affect how well they work. Do not start or stop other medicines without talking to your healthcare provider. Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist each time you get a new medicine. |
|||
How should I take VIMPAT?
|
|||
| What should I avoid while taking VIMPAT?
Do not drive, operate heavy machinery, or do other dangerous activities until you know how VIMPAT affects you. VIMPAT may cause you to feel dizzy, have double vision, feel sleepy, or have problems with coordination and walking. |
|||
What are the possible side effects of VIMPAT?
|
|||
|
|
||
| The most common side effects of VIMPAT include: | |||
|
|
||
| These are not all of the possible side effects of VIMPAT. For more information ask your healthcare provider or pharmacist. Tell your healthcare provider about any side effect that bothers you or that does not go away. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | |||
How should I store VIMPAT?
|
|||
| General Information about the safe and effective use of VIMPAT. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use VIMPAT for a condition for which it was not prescribed. Do not give VIMPAT to other people, even if they have the same symptoms that you have. It may harm them. This Medication Guide summarizes the most important information about VIMPAT. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about VIMPAT that is written for health professionals. |
|||
| What are the ingredients in VIMPAT? Active ingredient: lacosamide Tablet inactive ingredients: colloidal silicon dioxide, crospovidone, hydroxypropylcellulose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, talc, titanium dioxide and additional ingredients listed below:
Oral solution inactive ingredients: purified water, sorbitol solution, glycerin, polyethylene glycol, carboxymethylcellulose sodium, acesulfame potassium, methylparaben, flavoring (including natural and artificial flavors, propylene glycol, aspartame, and maltol), anhydrous citric acid and sodium chloride. Manufactured for: UCB, Inc. Smyrna, GA 30080 VIMPAT® is a registered trademark under license from Harris FRC Corporation and covered by one or more claims of U.S. Patent 38,551. For more information, go to www.vimpat.com or call 1-844-599-2273. |
|||
| VIMPAT
lacosamide tablet, film coated |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| VIMPAT
lacosamide tablet, film coated |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| VIMPAT
lacosamide tablet, film coated |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| VIMPAT
lacosamide tablet, film coated |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| VIMPAT
lacosamide injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| VIMPAT
lacosamide solution |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Labeler - UCB, Inc. (028526403) |