Drug Detail:Vyvanse (Lisdexamfetamine [ lis-dex-am-fet-a-meen ])
Drug Class: CNS stimulants
Highlights of Prescribing Information
VYVANSE® (lisdexamfetamine dimesylate) capsules, for oral use, CII
VYVANSE® (lisdexamfetamine dimesylate) chewable tablets, for oral use, CII
Initial U.S. Approval: 2007
WARNING: ABUSE AND DEPENDENCE
See full prescribing information for complete boxed warning.
- CNS stimulants, including VYVANSE, other amphetamine-containing products, and methylphenidate have a high potential for abuse and dependence (5.1, 9.3)
- Assess the risk of abuse prior to prescribing and monitor for signs of abuse and dependence while on therapy (5.1, 9.2)
Recent Major Changes
| Indications and Usage (1) | 7/2021 |
| Warnings and Precautions (5.5) | 7/2021 |
Indications and Usage for Vyvanse
VYVANSE is a central nervous system (CNS) stimulant indicated for the treatment of (1):
- Attention Deficit Hyperactivity Disorder (ADHD) in adults and pediatric patients 6 years and older
- Moderate to severe binge eating disorder (BED) in adults
Limitations of Use:
- Pediatric patients with ADHD younger than 6 years of age experienced more long-term weight loss than patients 6 years and older (8.4)
- VYVANSE is not indicated for weight loss. Use of other sympathomimetic drugs for weight loss has been associated with serious cardiovascular adverse events. The safety and effectiveness of VYVANSE for the treatment of obesity have not been established (5.2)
Vyvanse Dosage and Administration
| Indicated Population | Initial Dose | Titration Schedule | Recommended Dose | Maximum Dose |
|---|---|---|---|---|
| ADHD (Adults and pediatric patients 6 years and older) (2.2) | 30 mg every morning | 10 mg or 20 mg weekly | 30 mg to 70 mg per day | 70 mg per day |
| BED (Adults) (2.3) | 30 mg every morning | 20 mg weekly | 50 mg to 70 mg per day | 70 mg per day |
- Prior to treatment, assess for presence of cardiac disease (2.4)
- Severe renal impairment: Maximum dose is 50 mg/day (2.5)
- End stage renal disease (ESRD): Maximum dose is 30 mg/day (2.5)
Dosage Forms and Strengths
- Capsules: 10 mg, 20 mg, 30 mg, 40 mg, 50 mg, 60 mg, 70 mg (3)
- Chewable tablets: 10 mg, 20 mg, 30 mg, 40 mg, 50 mg, 60 mg (3)
Contraindications
- Known hypersensitivity to amphetamine products or other ingredients in VYVANSE (4)
- Use with monoamine oxidase (MAO) inhibitor, or within 14 days of the last MAO inhibitor dose (4, 7.2)
Warnings and Precautions
- Serious Cardiovascular Reactions: Sudden death has been reported in association with CNS stimulant treatment at recommended doses in pediatric patients with structural cardiac abnormalities or other serious heart problems. In adults, sudden death, stroke, and myocardial infarction have been reported. Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart arrhythmia, or coronary artery disease (5.2)
- Blood Pressure and Heart Rate Increases: Monitor blood pressure and pulse. Consider benefits and risks before use in patients for whom blood pressure increases may be problematic (5.3)
- Psychiatric Adverse Reactions: May cause psychotic or manic symptoms in patients with no prior history, or exacerbation of symptoms in patients with pre-existing psychosis. Evaluate for bipolar disorder prior to stimulant use (5.4)
- Suppression of Growth: Monitor height and weight in pediatric patients during treatment (5.5)
- Peripheral Vasculopathy, including Raynaud's phenomenon: Stimulants are associated with peripheral vasculopathy, including Raynaud's phenomenon. Careful observation for digital changes is necessary during treatment with stimulants (5.6)
- Serotonin Syndrome: Increased risk when co-administered with serotonergic agents (e.g., SSRIs, SNRIs, triptans), but also during overdosage situations. If it occurs, discontinue VYVANSE and initiate supportive treatment (4, 5.7, 10)
Adverse Reactions/Side Effects
Most common adverse reactions (incidence ≥5% and at a rate at least twice placebo) in pediatric patients ages 6 to 17 years, and/or adults with ADHD were anorexia, anxiety, decreased appetite, decreased weight, diarrhea, dizziness, dry mouth, irritability, insomnia, nausea, upper abdominal pain, and vomiting (6.1)
Most common adverse reactions (incidence ≥ 5% and at a rate at least twice placebo) in adults with BED were dry mouth, insomnia, decreased appetite, increased heart rate, constipation, feeling jittery, and anxiety (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Takeda Pharmaceuticals at 1-800-828-2088 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Acidifying and Alkalinizing Agents: Agents that alter urinary pH can alter blood levels of amphetamine. Acidifying agents decrease amphetamine blood levels, while alkalinizing agents increase amphetamine blood levels. Adjust VYVANSE dosage accordingly (2.6, 7.1)
Use In Specific Populations
- Pregnancy: May cause fetal harm (8.1)
- Lactation: Breastfeeding not recommended (8.2)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 1/2022
Related/similar drugs
Adderall, methylphenidate, Strattera, Ritalin, Concerta, atomoxetine, lisdexamfetamineFull Prescribing Information
WARNING: ABUSE AND DEPENDENCE
CNS stimulants, including VYVANSE, other amphetamine-containing products, and methylphenidate, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing and monitor for signs of abuse and dependence while on therapy [see Warnings and Precautions (5.1), and Drug Abuse and Dependence (9.2, 9.3)].
1. Indications and Usage for Vyvanse
VYVANSE® is indicated for the treatment of:
- Attention Deficit Hyperactivity Disorder (ADHD) in adults and pediatric patients 6 years and older [see Clinical Studies (14.1)]
- Moderate to severe binge eating disorder (BED) in adults [see Clinical Studies (14.2)].
2. Vyvanse Dosage and Administration
2.1 Pre-treatment Screening
Prior to treating patients with CNS stimulants, including VYVANSE, assess for the presence of cardiac disease (e.g., a careful history, family history of sudden death or ventricular arrhythmia, and physical exam) [see Warnings and Precautions (5.2)].
To reduce the abuse of CNS stimulants including VYVANSE, assess the risk of abuse, prior to prescribing. After prescribing, keep careful prescription records, educate patients about abuse, monitor for signs of abuse and overdose, and re-evaluate the need for VYVANSE use [see Warnings and Precautions (5.1), Drug Abuse and Dependence (9)].
2.2 General Instructions for Use
Take VYVANSE by mouth in the morning with or without food; avoid afternoon doses because of the potential for insomnia. VYVANSE may be administered in one of the following ways:
2.3 Dosage for Treatment of ADHD
The recommended starting dosage in adults and pediatric patients 6 years and older is 30 mg once daily in the morning. Dosage may be adjusted in increments of 10 mg or 20 mg at approximately weekly intervals up to maximum recommended dosage of 70 mg once daily [see Clinical Studies (14.1)].
2.4 Dosage for Treatment of Moderate to Severe BED in Adults
The recommended starting dosage in adults is 30 mg once daily to be titrated in increments of 20 mg at approximately weekly intervals to achieve the recommended target dose of 50 mg to 70 mg once daily. The maximum recommended dosage is 70 mg once daily [see Clinical Studies (14.2)]. Discontinue VYVANSE if binge eating does not improve.
2.5 Dosage in Patients with Renal Impairment
In patients with severe renal impairment (GFR 15 to < 30 mL/min/1.73 m2), the maximum dosage should not exceed 50 mg once daily. In patients with end stage renal disease (ESRD, GFR < 15 mL/min/1.73 m2), the maximum recommended dosage is 30 mg once daily [see Use in Specific Populations (8.6)].
2.6 Dosage Modifications due to Drug Interactions
Agents that alter urinary pH can impact urinary excretion and alter blood levels of amphetamine. Acidifying agents (e.g., ascorbic acid) decrease blood levels, while alkalinizing agents (e.g., sodium bicarbonate) increase blood levels. Adjust VYVANSE dosage accordingly [see Drug Interactions (7.1)].
4. Contraindications
VYVANSE is contraindicated in patients with:
- Known hypersensitivity to amphetamine products or other ingredients of VYVANSE. Anaphylactic reactions, Stevens-Johnson Syndrome, angioedema, and urticaria have been observed in postmarketing reports [see Adverse Reactions (6.2)].
- Patients taking monoamine oxidase inhibitors (MAOIs), or within 14 days of stopping MAOIs (including MAOIs such as linezolid or intravenous methylene blue), because of an increased risk of hypertensive crisis [see Warnings and Precautions (5.7) and Drug Interactions (7.1)].
5. Warnings and Precautions
5.1 Potential for Abuse and Dependence
CNS stimulants, including VYVANSE, other amphetamine-containing products, and methylphenidate, have a high potential for abuse and dependence. Assess the risk of abuse prior to prescribing, and monitor for signs of abuse and dependence while on therapy [see Drug Abuse and Dependence (9.2, 9.3)].
5.2 Serious Cardiovascular Reactions
Sudden death, stroke, and myocardial infarction have been reported in adults with CNS stimulant treatment at recommended doses. Sudden death has been reported in pediatric patients with structural cardiac abnormalities and other serious heart problems taking CNS stimulants at recommended doses for ADHD. Avoid use in patients with known structural cardiac abnormalities, cardiomyopathy, serious heart arrhythmia, coronary artery disease, and other serious heart problems. Further evaluate patients who develop exertional chest pain, unexplained syncope, or arrhythmias during VYVANSE treatment.
5.3 Blood Pressure and Heart Rate Increases
CNS stimulants cause an increase in blood pressure (mean increase about 2 to 4 mm Hg) and heart rate (mean increase about 3 to 6 bpm). Monitor all patients for potential tachycardia and hypertension.
5.5 Suppression of Growth
CNS stimulants have been associated with weight loss and slowing of growth rate in pediatric patients. Closely monitor growth (weight and height) in pediatric patients treated with CNS stimulants, including VYVANSE. In a 4-week, placebo-controlled trial of VYVANSE in pediatric patients ages 6 to 12 years old with ADHD, there was a dose-related decrease in weight in the VYVANSE groups compared to weight gain in the placebo group. Additionally, in studies of another stimulant, there was slowing of the increase in height [see Adverse Reactions (6.1)].
Patients who are not growing or gaining height or weight as expected may need to have their treatment interrupted. VYVANSE is not approved for use in pediatric patients below 6 years of age [see Use in Specific Populations (8.4)].
5.6 Peripheral Vasculopathy, including Raynaud's Phenomenon
Stimulants, including VYVANSE, are associated with peripheral vasculopathy, including Raynaud's phenomenon. Signs and symptoms are usually intermittent and mild; however, very rare sequelae include digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud's phenomenon, were observed in post-marketing reports at different times and at therapeutic doses in all age groups throughout the course of treatment. Signs and symptoms generally improve after reduction in dose or discontinuation of drug. Careful observation for digital changes is necessary during treatment with stimulants. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for certain patients.
5.7 Serotonin Syndrome
Serotonin syndrome, a potentially life-threatening reaction, may occur when amphetamines are used in combination with other drugs that affect the serotonergic neurotransmitter systems such as monoamine oxidase inhibitors (MAOIs), selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, and St. John's Wort [see Drug Interactions (7.1)]. The co-administration with cytochrome P450 2D6 (CYP2D6) inhibitors may also increase the risk with increased exposure to the active metabolite of VYVANSE (dextroamphetamine). In these situations, consider an alternative non-serotonergic drug or an alternative drug that does not inhibit CYP2D6 [see Drug Interactions (7.1)].
Serotonin syndrome symptoms may include mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and/or gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea).
Concomitant use of VYVANSE with MAOI drugs is contraindicated [see Contraindications (4)].
Discontinue treatment with VYVANSE and any concomitant serotonergic agents immediately if symptoms of serotonin syndrome occur, and initiate supportive symptomatic treatment. If concomitant use of VYVANSE with other serotonergic drugs or CYP2D6 inhibitors is clinically warranted, initiate VYVANSE with lower doses, monitor patients for the emergence of serotonin syndrome during drug initiation or titration, and inform patients of the increased risk for serotonin syndrome.
6. Adverse Reactions/Side Effects
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- Known hypersensitivity to amphetamine products or other ingredients of VYVANSE [see Contraindications (4)]
- Hypertensive Crisis When Used Concomitantly with Monoamine Oxidase Inhibitors [see Contraindications (4) and Drug Interactions (7.1)]
- Drug Dependence [see Boxed Warning, Warnings and Precautions (5.1), and Drug Abuse and Dependence (9.2, 9.3)]
- Serious Cardiovascular Reactions [see Warnings and Precautions (5.2)]
- Blood Pressure and Heart Rate Increases [see Warnings and Precautions (5.3)]
- Psychiatric Adverse Reactions [see Warnings and Precautions (5.4)]
- Suppression of Growth [see Warnings and Precautions (5.5)]
- Peripheral Vasculopathy, including Raynaud's phenomenon [see Warnings and Precautions (5.6)]
- Serotonin Syndrome [see Warnings and Precautions (5.7)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of VYVANSE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These events are as follows: cardiomyopathy, mydriasis, diplopia, difficulties with visual accommodation, blurred vision, eosinophilic hepatitis, anaphylactic reaction, hypersensitivity, dyskinesia, dysgeusia, tics, bruxism, depression, dermatillomania, alopecia, aggression, Stevens-Johnson Syndrome, chest pain, angioedema, urticaria, seizures, libido changes, frequent or prolonged erections, constipation, rhabdomyolysis, and intestinal ischemia.
7. Drug Interactions
7.1 Drugs Having Clinically Important Interactions with Amphetamines
| MAO Inhibitors (MAOI) | |
| Clinical Impact | MAOI antidepressants slow amphetamine metabolism, increasing amphetamines effect on the release of norepinephrine and other monoamines from adrenergic nerve endings causing headaches and other signs of hypertensive crisis. Toxic neurological effects and malignant hyperpyrexia can occur, sometimes with fatal results. |
| Intervention | Do not administer VYVANSE during or within 14 days following the administration of MAOI [see Contraindications (4)]. |
| Serotonergic Drugs | |
| Clinical Impact | The concomitant use of VYVANSE and serotonergic drugs increases the risk of serotonin syndrome. |
| Intervention | Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome, particularly during VYVANSE initiation or dosage increase. If serotonin syndrome occurs, discontinue VYVANSE and the concomitant serotonergic drug(s) [see Warnings and Precautions (5.7)]. |
| CYP2D6 Inhibitors | |
| Clinical Impact | The concomitant use of VYVANSE and CYP2D6 inhibitors may increase the exposure of dextroamphetamine, the active metabolite of VYVANSE compared to the use of the drug alone and increase the risk of serotonin syndrome. |
| Intervention | Initiate with lower doses and monitor patients for signs and symptoms of serotonin syndrome particularly during VYVANSE initiation and after a dosage increase. If serotonin syndrome occurs, discontinue VYVANSE and the CYP2D6 inhibitor [see Warnings and Precautions (5.7) and Overdosage (10)]. |
| Alkalinizing Agents | |
| Clinical Impact | Urinary alkalinizing agents can increase blood levels and potentiate the action of amphetamine. |
| Intervention | Co-administration of VYVANSE and urinary alkalinizing agents should be avoided. |
| Acidifying Agents | |
| Clinical Impact | Urinary acidifying agents can lower blood levels and efficacy of amphetamines. |
| Intervention | Increase dose based on clinical response. |
| Tricyclic Antidepressants | |
| Clinical Impact | May enhance the activity of tricyclic or sympathomimetic agents causing striking and sustained increases in the concentration of d-amphetamine in the brain; cardiovascular effects can be potentiated. |
| Intervention | Monitor frequently and adjust or use alternative therapy based on clinical response. |
7.2 Drugs Having No Clinically Important Interactions with VYVANSE
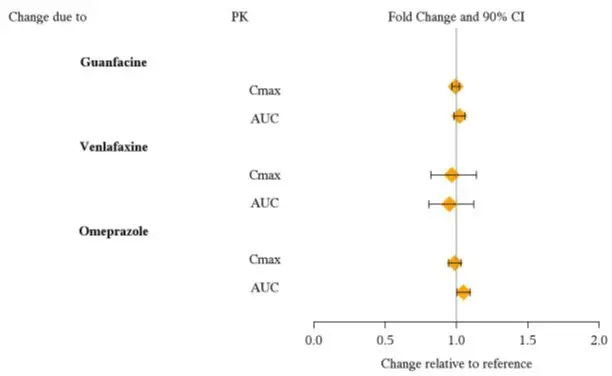
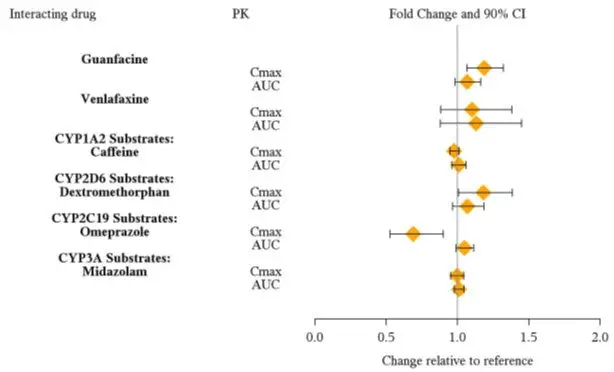
From a pharmacokinetic perspective, no dose adjustment of VYVANSE is necessary when VYVANSE is co-administered with guanfacine, venlafaxine, or omeprazole. In addition, no dose adjustment of guanfacine or venlafaxine is needed when VYVANSE is co-administered [see Clinical Pharmacology (12.3)].
From a pharmacokinetic perspective, no dose adjustment for drugs that are substrates of CYP1A2 (e.g., theophylline, duloxetine, melatonin), CYP2D6 (e.g., atomoxetine, desipramine, venlafaxine), CYP2C19 (e.g., omeprazole, lansoprazole, clobazam), and CYP3A4 (e.g., midazolam, pimozide, simvastatin) is necessary when VYVANSE is co-administered [see Clinical Pharmacology (12.3)].
8. Use In Specific Populations
8.5 Geriatric Use
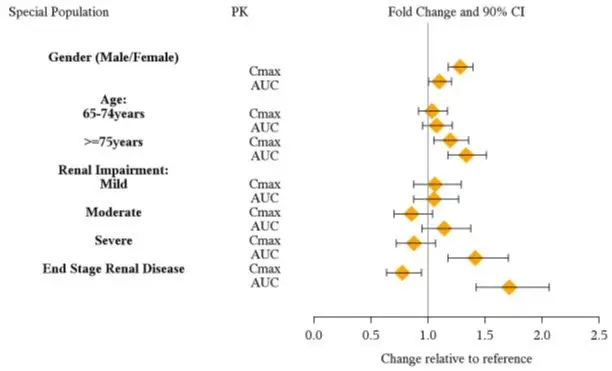
Clinical studies of VYVANSE did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience and pharmacokinetic data [see Clinical Pharmacology (12.3)] have not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should start at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
8.6 Renal Impairment
Due to reduced clearance in patients with severe renal impairment (GFR 15 to < 30 mL/min/1.73 m2), the maximum dose should not exceed 50 mg/day. The maximum recommended dose in ESRD (GFR < 15 mL/min/1.73 m2) patients is 30 mg/day [see Clinical Pharmacology (12.3)].
Lisdexamfetamine and d-amphetamine are not dialyzable.
9. Drug Abuse and Dependence
9.1 Controlled Substance
VYVANSE contains lisdexamfetamine, a prodrug of amphetamine, a Schedule II controlled substance.
9.2 Abuse
CNS stimulants, including VYVANSE, other amphetamine-containing products, and methylphenidate have a high potential for abuse. Abuse is the intentional non-therapeutic use of a drug, even once, to achieve a desired psychological or physiological effect. Abuse is characterized by impaired control over drug use, compulsive use, continued use despite harm, and craving. Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence. Both abuse and misuse may lead to addiction, and some individuals may develop addiction even when taking VYVANSE as prescribed.
Signs and symptoms of amphetamine abuse may include increased heart rate, respiratory rate, blood pressure, and/or sweating, dilated pupils, hyperactivity, restlessness, insomnia, decreased appetite, loss of coordination, tremors, flushed skin, vomiting, and/or abdominal pain. Anxiety, psychosis, hostility, aggression, suicidal or homicidal ideation have also been seen. Abusers of CNS stimulants may chew, snort, inject, or use other unapproved routes of administration which can result in overdose and death [see Overdosage (10)].
To reduce the abuse of CNS stimulants, including VYVANSE, assess the risk of abuse prior to prescribing. After prescribing, keep careful prescription records, educate patients and their families about abuse and on proper storage and disposal of CNS stimulants. Monitor for signs of abuse while on therapy, and re-evaluate the need for VYVANSE use.
10. Overdosage
Consult with a Certified Poison Control Center (1-800-222-1222) for up-to-date guidance and advice for treatment of overdosage. Individual patient response to amphetamines varies widely. Toxic symptoms may occur idiosyncratically at low doses.
Manifestations of amphetamine overdose include restlessness, tremor, hyperreflexia, rapid respiration, confusion, assaultiveness, hallucinations, panic states, hyperpyrexia and rhabdomyolysis. Fatigue and depression usually follow the central nervous system stimulation. Serotonin syndrome has been reported with amphetamine use, including VYVANSE. Cardiovascular effects include arrhythmias, hypertension or hypotension and circulatory collapse. Gastrointestinal symptoms include nausea, vomiting, diarrhea and abdominal cramps. Fatal poisoning is usually preceded by convulsions and coma.
Lisdexamfetamine and d-amphetamine are not dialyzable.
11. Vyvanse Description
VYVANSE (lisdexamfetamine dimesylate), a CNS stimulant, is for once-a-day oral administration. The chemical designation for lisdexamfetamine dimesylate is (2S)-2,6-diamino-N-[(1S)-1-methyl-2-phenylethyl] hexanamide dimethanesulfonate. The molecular formula is C15H25N3O∙(CH4O3S)2, which corresponds to a molecular weight of 455.60. The chemical structure is:
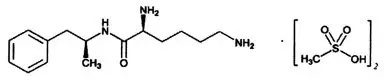
Lisdexamfetamine dimesylate is a white to off-white powder that is soluble in water (792 mg/mL).
12. Vyvanse - Clinical Pharmacology
12.1 Mechanism of Action
Lisdexamfetamine is a prodrug of dextroamphetamine. Amphetamines are non-catecholamine sympathomimetic amines with CNS stimulant activity. The exact mode of therapeutic action in ADHD and BED is not known.
12.2 Pharmacodynamics
Amphetamines block the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space. The parent drug, lisdexamfetamine, does not bind to the sites responsible for the reuptake of norepinephrine and dopamine in vitro.
12.3 Pharmacokinetics
Pharmacokinetic studies after oral administration of lisdexamfetamine dimesylate have been conducted in healthy adult (capsule and chewable tablet formulations) and pediatric (6 to 12 years) patients with ADHD (capsule formulation). After single dose administration of lisdexamfetamine dimesylate, pharmacokinetics of dextroamphetamine was found to be linear between 30 mg and 70 mg in a pediatric study (6 to 12 years), and between 50 mg and 250 mg in an adult study. Dextroamphetamine pharmacokinetic parameters following administration of lisdexamfetamine dimesylate in adults exhibited low inter-subject (<25%) and intra-subject (<8%) variability. There is no accumulation of lisdexamfetamine and dextroamphetamine at steady state in healthy adults.
14. Clinical Studies
14.2 Binge Eating Disorder (BED)
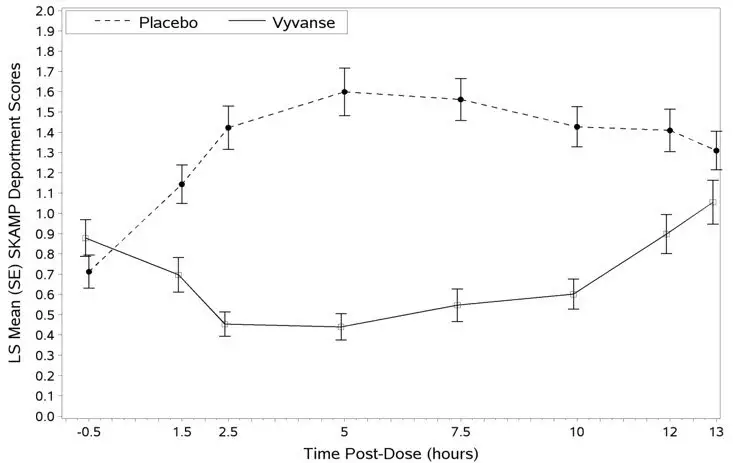
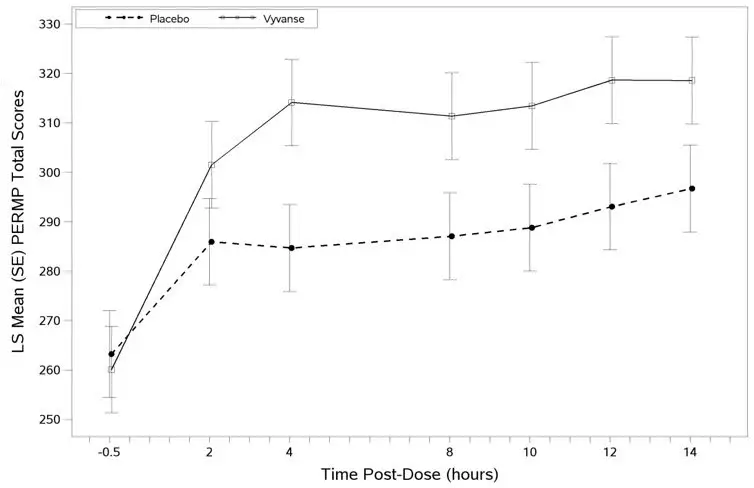
A phase 2 study evaluated the efficacy of VYVANSE 30, 50 and 70 mg/day compared to placebo in reducing the number of binge days/week in adults with at least moderate to severe BED. This randomized, double-blind, parallel-group, placebo-controlled, forced-dose titration study (Study 10) consisted of an 11-week double-blind treatment period (3 weeks of forced-dose titration followed by 8 weeks of dose maintenance). VYVANSE 30 mg/day was not statistically different from placebo on the primary endpoint. The 50 and 70 mg/day doses were statistically superior to placebo on the primary endpoint.
The efficacy of VYVANSE in the treatment of BED was demonstrated in two 12-week randomized, double-blind, multi-center, parallel-group, placebo-controlled, dose-optimization studies (Study 11 and Study 12) in adults aged 18-55 years (Study 11: N=374, Study 12: N=350) with moderate to severe BED. A diagnosis of BED was confirmed using DSM-IV criteria for BED. Severity of BED was determined based on having at least 3 binge days per week for 2 weeks prior to the baseline visit and on having a Clinical Global Impression Severity (CGI-S) score of ≥4 at the baseline visit. For both studies, a binge day was defined as a day with at least 1 binge episode, as determined from the subject's daily binge diary.
Both 12-week studies consisted of a 4-week dose-optimization period and an 8-week dose-maintenance period. During dose-optimization, subjects assigned to VYVANSE began treatment at the titration dose of 30 mg/day and, after 1 week of treatment, were subsequently titrated to 50 mg/day. Additional increases to 70 mg/day were made as tolerated and clinically indicated. Following the dose-optimization period, subjects continued on their optimized dose for the duration of the dose-maintenance period.
The primary efficacy outcome for the two studies was defined as the change from baseline at Week 12 in the number of binge days per week. Baseline is defined as the weekly average of the number of binge days per week for the 14 days prior to the baseline visit. Subjects from both studies on VYVANSE had a statistically significantly greater reduction from baseline in mean number of binge days per week at Week 12. In addition, subjects on VYVANSE showed greater improvement as compared to placebo across key secondary outcomes with higher proportion of subjects rated improved on the CGI-I rating scale, higher proportion of subjects with 4-week binge cessation, and greater reduction in the Yale-Brown Obsessive Compulsive Scale Modified for Binge Eating (Y-BOCS-BE) total score.
| Study Number | Treatment Group | Primary Efficacy Measure: Binge Days per Week at Week 12 | ||
|---|---|---|---|---|
| Mean Baseline Score (SD) | LS Mean Change from Baseline (SE) | Placebo-subtracted Difference* (95% CI) | ||
| SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: confidence interval. | ||||
|
||||
| Study 11 | VYVANSE (50 or 70 mg/day)† | 4.79 (1.27) | -3.87 (0.12) | -1.35 (-1.70, -1.01) |
| Placebo | 4.60 (1.21) | -2.51 (0.13) | -- | |
| Study 12 | VYVANSE (50 or 70 mg/day)† | 4.66 (1.27) | -3.92 (0.14) | -1.66 (-2.04, -1.28) |
| Placebo | 4.82 (1.42) | -2.26 (0.14) | -- | |
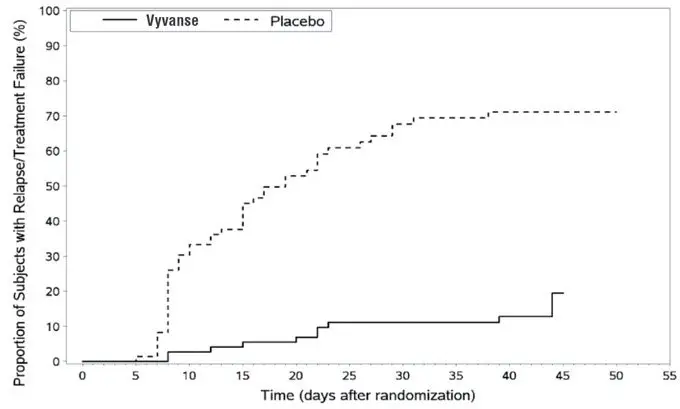
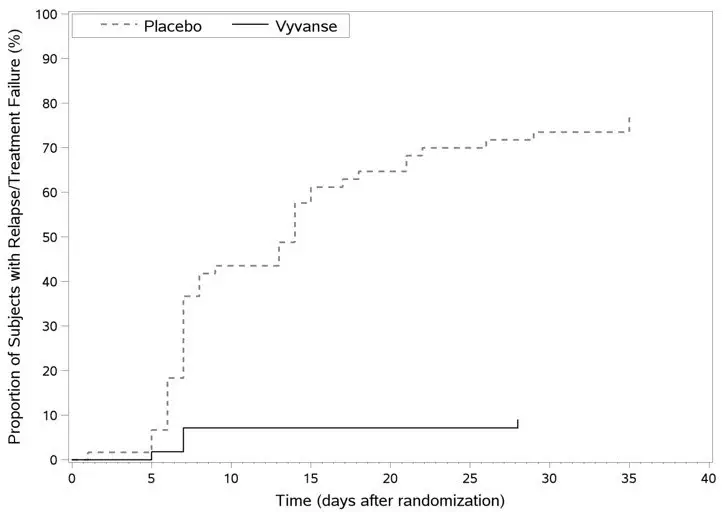
A double-blind, placebo controlled, randomized withdrawal design study (Study 13) was conducted to evaluate maintenance of efficacy based on time to relapse between VYVANSE and placebo in adults aged 18 to 55 (N=267) with moderate to severe BED. In this longer-term study patients who had responded to VYVANSE in the preceding 12-week open-label treatment phase were randomized to continuation of VYVANSE or placebo for up to 26 weeks of observation for relapse. Response in the open-label phase was defined as 1 or fewer binge days each week for four consecutive weeks prior to the last visit at the end of the 12-week open-label phase and a CGI-S score of 2 or less at the same visit. Relapse during the double-blind phase was defined as having 2 or more binge days each week for two consecutive weeks (14 days) prior to any visit and having an increase in CGI-S score of 2 or more points compared to the randomized-withdrawal baseline. Maintenance of efficacy for patients who had an initial response during the open-label period and then continued on VYVANSE during the 26-week double-blind randomized-withdrawal phase was demonstrated with VYVANSE being superior over placebo as measured by time to relapse.
Figure 8 Kaplan-Meier Estimated Proportions of Subjects with Relapse in Adults with BED (Study 13)
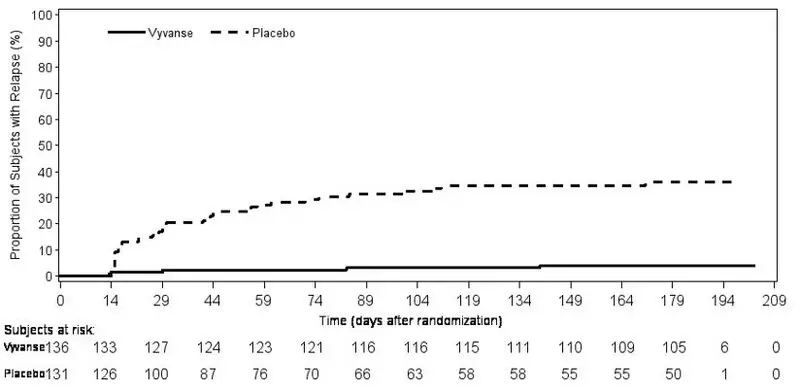
Examination of population subgroups based on age (there were no patients over 65), gender, and race did not reveal any clear evidence of differential responsiveness in the treatment of BED.
16. How is Vyvanse supplied
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
| VYVANSE
lisdexamfetamine dimesylate capsule |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| VYVANSE
lisdexamfetamine dimesylate capsule |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| VYVANSE
lisdexamfetamine dimesylate capsule |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| VYVANSE
lisdexamfetamine dimesylate capsule |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| VYVANSE
lisdexamfetamine dimesylate capsule |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| VYVANSE
lisdexamfetamine dimesylate capsule |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| VYVANSE
lisdexamfetamine dimesylate capsule |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| VYVANSE
lisdexamfetamine dimesylate tablet, chewable |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| VYVANSE
lisdexamfetamine dimesylate tablet, chewable |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| VYVANSE
lisdexamfetamine dimesylate tablet, chewable |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| VYVANSE
lisdexamfetamine dimesylate tablet, chewable |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| VYVANSE
lisdexamfetamine dimesylate tablet, chewable |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| VYVANSE
lisdexamfetamine dimesylate tablet, chewable |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Takeda Pharmaceuticals America, Inc. (039997266) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Curia New York, Inc. | 124193793 | ANALYSIS(59417-101, 59417-102, 59417-103, 59417-104, 59417-105, 59417-106, 59417-107, 59417-115, 59417-116, 59417-117, 59417-118, 59417-119, 59417-120) , API MANUFACTURE(59417-101, 59417-102, 59417-103, 59417-104, 59417-105, 59417-106, 59417-107, 59417-115, 59417-116, 59417-117, 59417-118, 59417-119, 59417-120) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Cambrex Charles City, Inc | 782974257 | API MANUFACTURE(59417-101, 59417-102, 59417-103, 59417-104, 59417-105, 59417-106, 59417-107, 59417-115, 59417-116, 59417-117, 59417-118, 59417-119, 59417-120) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Patheon Manufacturing Services LLC | 079415560 | ANALYSIS(59417-101, 59417-102, 59417-103, 59417-104, 59417-105, 59417-106, 59417-107) , MANUFACTURE(59417-101, 59417-102, 59417-103, 59417-104, 59417-105, 59417-106, 59417-107) , PACK(59417-101, 59417-102, 59417-103, 59417-104, 59417-105, 59417-106, 59417-107) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Patheon Pharmaceuticals Inc. | 005286822 | ANALYSIS(59417-101, 59417-102, 59417-103, 59417-104, 59417-105, 59417-106, 59417-107, 59417-115, 59417-116, 59417-117, 59417-118, 59417-119, 59417-120) , MANUFACTURE(59417-101, 59417-102, 59417-103, 59417-104, 59417-105, 59417-106, 59417-107, 59417-115, 59417-116, 59417-117, 59417-118, 59417-119, 59417-120) , PACK(59417-101, 59417-102, 59417-103, 59417-104, 59417-105, 59417-106, 59417-107, 59417-115, 59417-116, 59417-117, 59417-118, 59417-119, 59417-120) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Sharp Packaging Services, LLC | 143696495 | PACK(59417-101, 59417-102, 59417-103, 59417-104, 59417-105, 59417-106, 59417-107) | |