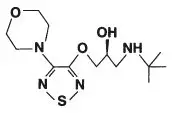
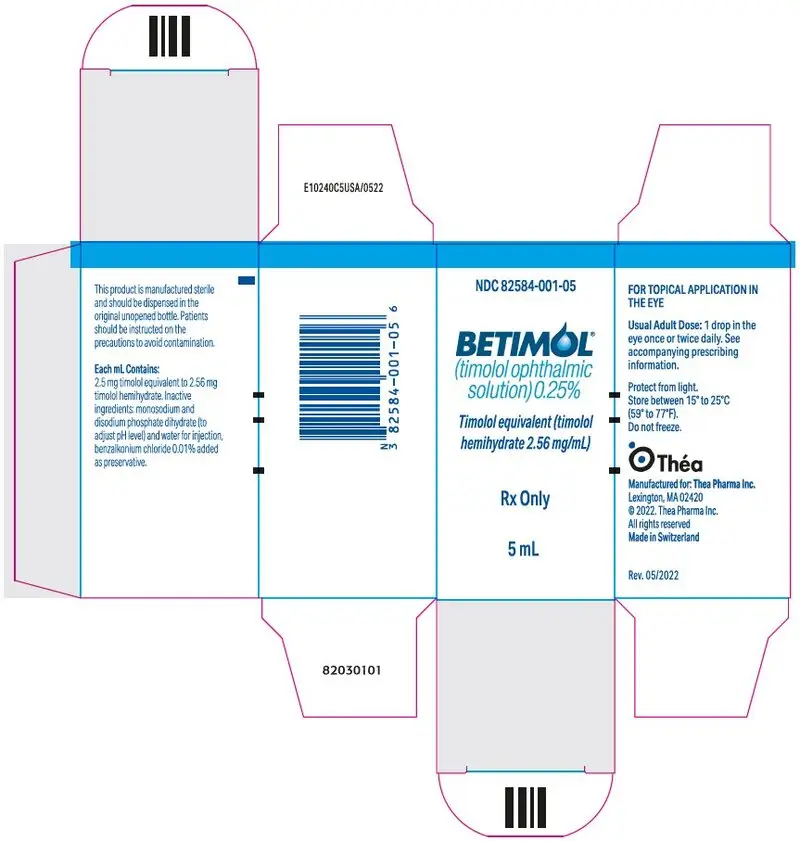
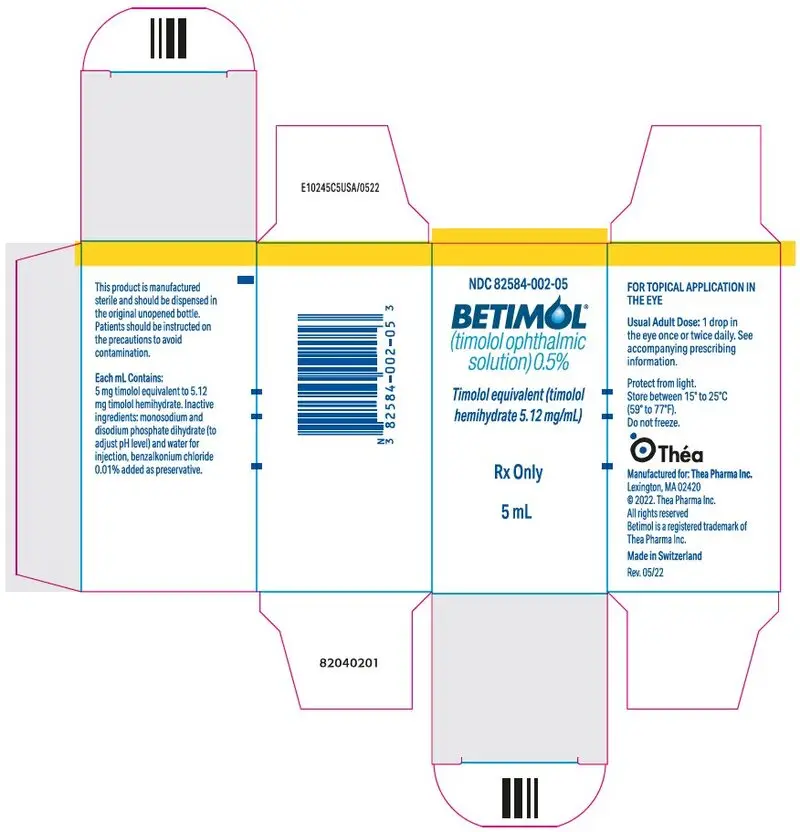
Drug Detail:Betimol (Timolol ophthalmic [ tim-oh-lol-off-thal-mik ])
Drug Class: Ophthalmic glaucoma agents
Contraindications
Betimol ® is contraindicated in patients with overt heart failure, cardiogenic shock, sinus bradycardia, second- or third-degree atrioventricular block, bronchial asthma or history of bronchial asthma, or severe chronic obstructive pulmonary disease, or hypersensitivity to any component of this product.
Warnings
As with other topically applied ophthalmic drugs, Betimol ® is absorbed systemically. The same adverse reactions found with systemic administration of beta-adrenergic blocking agents may occur with topical administration. For example, severe respiratory and cardiac reactions, including death due to bronchospasm in patients with asthma, and rarely, death in association with cardiac failure have been reported following systemic or topical administration of beta-adrenergic blocking agents.
Precautions
General
Because of the potential effects of beta-adrenergic blocking agents relative to blood pressure and pulse, these agents should be used with caution in patients with cerebrovascular insufficiency. If signs or symptoms suggesting reduced cerebral blood flow develop following initiation of therapy with Betimol ®, alternative therapy should be considered.
There have been reports of bacterial keratitis associated with the use of multiple dose containers of topical ophthalmic products. These containers had been inadvertently contaminated by patients who, in most cases, had a concurrent corneal disease or a disruption of the ocular epithelial surface. (See PRECAUTIONS, Information for Patients.)
Anaphylaxis
While taking beta-blockers, patients with a history of atopy or a history of severe anaphylactic reactions to a variety of allergens may be more reactive to repeated accidental, diagnostic, or therapeutic challenge with such allergens. Such patients may be unresponsive to the usual doses of epinephrine used to treat anaphylactic reactions.
The preservative benzalkonium chloride may be absorbed by soft contact lenses. Patients who wear soft contact lenses should wait 5 minutes after instilling Betimol ® before they insert their lenses.
Information for Patients
Patients should be instructed to avoid allowing the tip of the dispensing container to contact the eye or surrounding structures.
Patients should also be instructed that ocular solutions can become contaminated by common bacteria known to cause ocular infections. Serious damage to the eye and subsequent loss of vision may result from using contaminated solutions. (See PRECAUTIONS, General.)
Patients requiring concomitant topical ophthalmic medications should be instructed to administer these at least 5 minutes apart.
Patients with bronchial asthma, a history of bronchial asthma, severe chronic obstructive pulmonary disease, sinus bradycardia, second- or third-degree atrioventricular block, or cardiac failure should be advised not to take this product (See CONTRAINDICATIONS.)
| BETIMOL
timolol solution/ drops |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| BETIMOL
timolol solution/ drops |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Thea Pharma Inc. (117787029) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Akorn AG | 482198285 | manufacture(82584-001, 82584-002) , label(82584-001, 82584-002) , pack(82584-001, 82584-002) , analysis(82584-001, 82584-002) | |