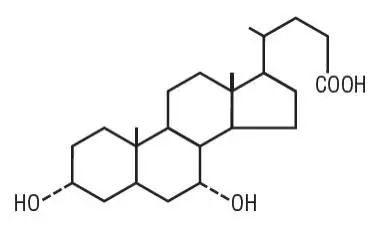
Drug Detail:Chenodal (Chenodiol (chenodeoxycholic acid) [ kee-noe-dye-ol ])
Drug Class: Gallstone solubilizing agents
Contraindications
Chenodal (chenodiol tablets) is contraindicated in the presence of known hepatocyte dysfunction or bile ductal abnormalities such as intrahepatic cholestasis, primary biliary cirrhosis or sclerosing cholangitis (see WARNINGS); a gallbladder confirmed as nonvisualizing after two consecutive single doses of dye; radiopaque stones; or gallstone complications or compelling reasons for gallbladder surgery including unremitting acute cholecystitis, cholangitis, biliary obstruction, gallstone pancreatitis, or biliary gastrointestinal fistula.
Pregnancy Category X:
Chenodal (chenodiol tablets) may cause fetal harm when administered to a pregnant woman. Serious hepatic, renal and adrenal lesions occurred in fetuses of female Rhesus monkeys given 60 to 90 mg/kg/day (4 to 6 times the maximum recommended human dose, MRHD) from day 21 to day 45 of pregnancy. Hepatic lesions also occurred in neonatal baboons whose mothers had received 18 to 38 mg/kg (1 to 2 times the MRHD), all during pregnancy. Fetal malformations were not observed. Neither fatal liver damage nor fetal abnormalities occurred in reproduction studies in rats and hamsters. No human data are available at this time. Chenodal (chenodiol tablets) is contraindicated in women who are or may become pregnant. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
Warnings
Safe use of chenodiol depends upon selection of patients without pre-existing liver disease and upon faithful monitoring of serum aminotransferase levels to detect drug-induced liver toxicity. Aminotransferase elevations over three times the upper limit of normal have required discontinuation of chenodiol in 2% to 3% of patients. Although clinical and biopsy studies have not shown fulminant lesions, the possibility remains that an occasional patient may develop serious hepatic disease. Three patients with biochemical and histologic pictures of chronic active hepatitis while on chenodiol, 375 mg/day or 750 mg/day, have been reported. The biochemical abnormalities returned spontaneously to normal in two of the patients within 13 and 17 months; and after 17 months’ treatment with prednisone in the third. Follow-up biopsies were not done; and the causal relationship of the drug could not be determined. Another biopsied patient was terminated from therapy because of elevated aminotransferase levels and a liver biopsy was interpreted as showing active drug hepatitis.
One patient with sclerosing cholangitis, biliary cirrhosis and history of jaundice died during chenodiol treatment for hepatic duct stones. Before treatment, serum aminotransferase and alkaline phosphate levels were over twice the upper limit of normal; within one month they rose to over 10 times. Chenodiol was discontinued at seven weeks, when the patient was hospitalized with advanced hepatic failure and E. coli peritonitis; death ensured at the eighth week. A contribution of chenodiol to the fatal outcome could not be ruled out.
Epidemiologic studies suggest that bile acids might contribute to human colon cancer, but direct evidence is lacking. Bile acids, including chenodiol and lithocholic acid, have no carcinogenic potential in animal models, but have been shown to increase the number of tumors when administered with certain known carcinogens. The possibility that chenodiol therapy might contribute to colon cancer in otherwise susceptible individuals cannot be ruled out.
Adverse Reactions/Side Effects
Hepatobiliary: Dose-related serum aminotransferase (mainly SGPT) elevations, usually not accompanied by rises in alkaline phosphatase or bilirubin, occurred in 30% or more of patients treated with the recommended dose of chenodiol. In most cases, these elevations were minor (1½ to 3 times the upper limit of laboratory normal) and transient, returning to within the normal range within six months despite continued administration of the drug. In 2% to 3% of patients, SGPT levels rose to over three times the upper limit of laboratory normal, recurred on rechallenge with the drug, and required discontinuation of chenodiol treatment. Enzyme levels have returned to normal following withdrawal of chenodiol (see WARNINGS).
Morphologic studies of liver biopsies taken before and after 9 and 24 months of treatment with chenodiol have shown that 63% of the patients prior to chenodiol treatment had evidence of intrahepatic cholestasis. Almost all pretreatment patients had electron microscopic abnormalities. By the ninth month of treatment, reexamination of two-thirds of the patients showed on 89% incidence of the signs of intrahepatic cholestasis. Two of 89 patients at the ninth month had lithocholate-like lesions in the canalicular membrane, although there were not clinical enzyme abnormalities in the face of continued treatment and no change in Type 2 light microscopic parameters.
Increased Cholecystectomy Rate: NCGS patients with a history of biliary pain prior to treatment had higher cholecystectomy rates during the study if assigned to low dosage chenodiol (375 mg/day) than if assigned to either placebo or high dosage chenodiol (750 mg/day). The association with low dosage chenodiol though not clearly a causal one, suggests that patients unable to take higher doses of chenodiol may be at greater risk of cholecystectomy.
Gastrointestinal: Dose-related diarrhea has been encountered in 30% to 40% of chenodiol-treated patients and may occur at any time during treatment, but is most commonly encountered when treatment is initiated. Usually, the diarrhea is mild, translucent, well-tolerated and dose not interfere with therapy. Dose reduction has been required in 10% to 15% of patients, and in a controlled trial about half of these required a permanent reduction in dose. Anti-diarrhea agents have proven useful in some patients.
Discontinuation of Chenodal (chenodiol tablets) because of failure to control diarrhea is to be expected in approximately 3% of patients treated. Steady epigastric pain with nausea typical of lithiasis (biliary colic) usually is easily distinguishable from the crampy abdominal pain of drug-induced diarrhea.
Other less frequent, gastrointestinal side effects reported include urgency, cramps, heartburn, constipation, nausea and vomiting, anorexia, epigastric distress, dyspepsia, flatulence and nonspecific abdominal pain.
Serum Lipids: Serum total cholesterol and low-density lipoprotein (LDL) cholesterol may rise 10% or more during administration of chenodiol; no change has been seen in the high-density lipoprotein (HDL) fraction; small decreases in serum triglyceride levels for females have been reported.
Hematologic: Decreases in white call count, never below 3000, have been noted in a few patients treated with chenodiol; the drug was continued in all patients without incident.
| CHENODAL
chenodiol tablet, film coated |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Travere Therapeutics (965454502) |




![PRINCIPAL DISPLAY PANEL
– 250 mg Tablet Bottle Label
NDC- 68974-876-40
Travere Therapeutics
Chenodal®
(chenodiol tablets)
250mg
Each Film-Coated Tablet Contains:
Chenodiol………………………… 250 mg
Rx only
100 Tablets
Usual Adult Dosage: Please see package insert for detailed prescribing information.
Store and Dispense: Store at 20° to 25°C (68° to 77°F)
[see USP Controlled Room Temperature].
Dispense in a tight container as defined in the USP.
KEEP THIS AND ALL MEDICATION OUT OF THE REACH OF CHILDREN.
Manufactured for:
Travere Therapeutics
San Diego, CA 92130
866-758-7068
7121
Rev 05/2021](https://cdn.themeditary.com/images/2023/09/06/chenodal-02.webp)
