Drug Detail:Epiduo gel (Adapalene and benzoyl peroxide (topical) [ a-dap-a-leen-and-ben-zoyl-per-ox-ide ])
Drug Class: Topical acne agents
Highlights of Prescribing Information
EPIDUO® (adapalene and benzoyl peroxide) Gel 0.1% / 2.5% For topical use
Initial U.S. Approval: 2008
Indications and Usage for Epiduo
EPIDUO gel is a combination of adapalene, a retinoid, and benzoyl peroxide, and is indicated for the topical treatment of acne vulgaris in patients 9 years of age and older. (1)
Epiduo Dosage and Administration
EPIDUO gel is not for oral, ophthalmic, or intravaginal use. (2)
Apply a thin film of EPIDUO gel to affected areas of the face and/or trunk once daily after washing. Use a pea-sized amount for each area of the face (e.g., forehead, chin, each cheek). Avoid the eyes, lips and mucous membranes. (2)
Dosage Forms and Strengths
Each gram of EPIDUO gel contains 1 mg (0.1%) adapalene and 25 mg (2.5%) benzoyl peroxide. (3)
Contraindications
None. (4)
Warnings and Precautions
Ultraviolet Light and Environmental Exposure: Avoid exposure to sunlight and sunlamps. Wear sunscreen when sun exposure cannot be avoided. (5.1)
Erythema, scaling, dryness, stinging/burning, irritant and allergic contact dermatitis may occur with use of EPIDUO gel and may necessitate discontinuation. (5.2)
Adverse Reactions/Side Effects
Most commonly reported adverse events (≥1%) in patients treated with EPIDUO gel were dry skin, contact dermatitis, application site burning, application site irritation and skin irritation. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Galderma Laboratories, L.P. at 1-866-735-4137, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 2/2018
Related/similar drugs
doxycycline, clindamycin topical, erythromycin topical, tetracycline, TazoracFull Prescribing Information
1. Indications and Usage for Epiduo
EPIDUO gel is indicated for the topical treatment of acne vulgaris in patients 9 years of age and older.
2. Epiduo Dosage and Administration
For topical use only; EPIDUO gel is not for oral, ophthalmic, or intravaginal use.
Apply a thin film of EPIDUO gel to affected areas of the face and/or trunk once daily after washing. Use a pea-sized amount for each area of the face (e.g., forehead, chin, each cheek). Avoid the eyes, lips and mucous membranes.
3. Dosage Forms and Strengths
Each gram of EPIDUO gel contains 1 mg (0.1%) adapalene and 25 mg (2.5%) benzoyl peroxide in a white to very pale yellow, opaque, aqueous based gel.
PACKAGE LABEL 45 g PUMP
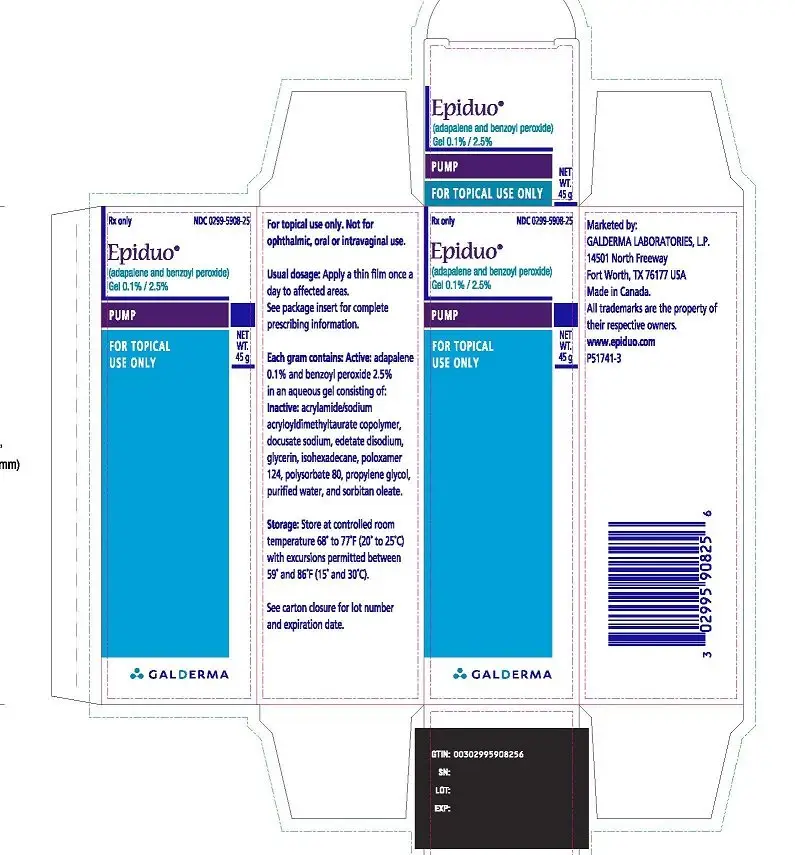
Rx Only
NDC 0299-5908-25
Epiduo®
(adapalene and benzoyl peroxide) Gel 0.1% / 2.5%
PUMP
FOR TOPICAL USE ONLY
NET WT. 45g
GALDERMA
For topical use only. Not for ophthalmic, oral or intravaginal use.
Usual dosage: Apply a thin film once a day to affected areas.
See package insert for complete prescribing information.
Each gram contains: Active: adapalene 0.1% and benzoyl peroxide 2.5% in an aqueous gel consisting of: Inactive: acrylamide/sodium acryloyldimethyltaurate copolymer, docusate sodium, edetate disodium, glycerin, isohexadecane, poloxamer 124, polysorbate 80, propylene glycol, purified water, and sorbitan oleate.
Storage: Store at controlled room temperature 68° to 77°F (20° to 25°C) with excursions permitted between 59° and 86°F (15° and 30°C).
See carton closure for lot number and expiration date.
Marketed by:
GALDERMA LABORATORIES, L.P.
14501 North Freeway, Fort Worth, TX 76177 USA
Made in Canada.
All trademarks are the property of their respective owners.
www.epiduo.com
P51741-3
| EPIDUO
adapalene and benzoyl peroxide gel |
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Galderma Laboratories, L.P. (047350186) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| G Production Inc. | 251676961 | manufacture(0299-5908) | |