Drug Detail:Pennsaid (Diclofenac topical [ dye-kloe-fen-ak-top-ik-al ])
Drug Class: Topical non-steroidal anti-inflammatories
Highlights of Prescribing Information
PENNSAID (diclofenac sodium) topical solution 2% w/w, for topical use
Initial U.S. Approval: 1988
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS
See full prescribing information for complete boxed warning.
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction, and stroke, which can be fatal This risk may occur early in treatment and may increase with duration of use (5.1)
- PENNSAID is contraindicated in the setting of coronary artery bypass graft (CABG) surgery (4, 5.1)
- NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events (5.2)
Recent Major Changes
|
|
|
|
Indications and Usage for Pennsaid
PENNSAID is a nonsteroidal anti-inflammatory drug indicated for the treatment of the pain of osteoarthritis of the knee(s). (1)
Pennsaid Dosage and Administration
Use the lowest effective dosage for the shortest duration consistent with individual patient treatment goals.
The recommended dose is 2 pump actuations on each painful knee, 2 times a day. (2)
- Apply PENNSAID, to clean, dry skin. (2.1)
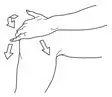
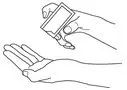
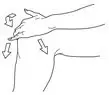
- Dispense 40 mg (2 pump actuations) directly onto the knee or first into the hand and then onto the knee. Spread evenly around front, back and sides of the knee. (2.1)
- Wash hands completely after administering the product. (2.2)
- Wait until the area is completely dry before covering with clothing or applying sunscreen, insect repellent, cosmetics, topical medications, or other substances. (2.2)
- Until the treated knee(s) is completely dry, avoid skin-to-skin contact between other people and the treated knee(s). (2.2)
- Do not get PENNSAID in your eyes, nose, or mouth (2.2).
Dosage Forms and Strengths
- PENNSAID (diclofenac sodium) topical solution 2% w/w (3)
Contraindications
- Known hypersensitivity to diclofenac or any components of the drug product. (4)
- History of asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. (4)
- In the setting of coronary artery bypass graft (CABG) surgery. (4)
Warnings and Precautions
- Hepatotoxicity: Inform patients of warning signs and symptoms of hepatotoxicity. Discontinue if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop (5.3)
- Hypertension: Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure (5.4, 7)
- Heart Failure and Edema: Avoid use of PENNSAID in patients with severe heart failure unless benefits are expected to outweigh risk of worsening heart failure (5.5)
- Renal Toxicity: Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of PENNSAID in patients with advanced renal disease unless benefits are expected to outweigh risk of worsening renal function (5.6)
- Anaphylactic Reactions: Seek emergency help if an anaphylactic reaction occurs (5.7)
- Exacerbation of Asthma Related to Aspirin Sensitivity: PENNSAID is contraindicated in patients with aspirin-sensitive asthma. Monitor patients with preexisting asthma (without aspirin sensitivity) (5.8)
- Serious Skin Reactions: Discontinue PENNSAID at first appearance of skin rash or other signs of hypersensitivity. (5.9, 5.15)
- Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): Discontinue and evaluate clinically (5.10)
- Fetal Toxicity: Limit use of NSAIDs, including PENNSAID, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal renal dysfunction and premature closure of the fetal ductus arteriosus (5.11, 8.1).
- Hematologic Toxicity: Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia (5.12, 7).
- Exposure to light: Avoid exposure of treated knee(s) to natural or artificial sunlight. (5.15)
- Eye Contact: Avoid contact of PENNSAID with eyes and mucosa. (5.16)
- Oral Nonsteroidal Anti-inflammatory Drugs: Avoid concurrent use with oral NSAIDs. (5.17)
Adverse Reactions/Side Effects
The most common adverse reactions with PENNSAID are application site reactions. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Horizon at 1-866-479-6742 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
- Drugs that Interfere with Hemostasis (e.g. warfarin, aspirin, SSRIs/SNRIs): Monitor patients for bleeding who are concomitantly using PENNSAID with drugs that interfere with hemostasis. Concomitant use of PENNSAID and analgesic doses of aspirin is not generally recommended (7)
- ACE Inhibitors, Angiotensin Receptor Blockers (ARB) or Beta-Blockers: Concomitant use with PENNSAID may diminish the antihypertensive effect of these drugs. Monitor blood pressure (7)
- ACE Inhibitors and ARBs: Concomitant use with PENNSAID in elderly, volume depleted, or those with renal impairment may result in deterioration of renal function. In such high risk patients, monitor for signs of worsening renal function (7)
- Diuretics: NSAIDs can reduce natriuretic effect of furosemide and thiazide diuretics. Monitor patients to assure diuretic efficacy including antihypertensive effects (7)
- Digoxin: Concomitant use with PENNSAID can increase serum concentration and prolong half-life of digoxin. Monitor serum digoxin levels (7)
Use In Specific Populations
- Infertility: NSAIDs are associated with reversible infertility. Consider withdrawal of PENNSAID in women who have difficulties conceiving. (8.3)
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 1/2022
Full Prescribing Information
1. Indications and Usage for Pennsaid
PENNSAID is indicated for the treatment of the pain of osteoarthritis of the knee(s).
2. Pennsaid Dosage and Administration
2.1 General Dosing Instructions
Use the lowest effective dosage for the shortest duration consistent with individual patient treatment goals [see Warnings and Precautions (5.2)].
For relief of the pain of osteoarthritis (OA) of the knee(s), the recommended dose is 40 mg of diclofenac sodium (2 pump actuations) on each painful knee, 2 times a day.
Apply PENNSAID to clean, dry skin.
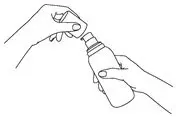
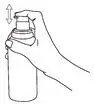
The pump must be primed before first use. Instruct patients to fully depress the pump mechanism (actuation) 4 times while holding the bottle in an upright position. This portion should be discarded to ensure proper priming of the pump. No further priming of the bottle should be required.
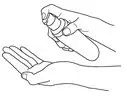
After the priming procedure, PENNSAID is properly dispensed by completely depressing the pump 2 times to achieve the prescribed dosage for one knee. Deliver the product directly into the palm of the hand and then apply evenly around front, back, and sides of the knee.
Application of PENNSAID in an amount exceeding or less than the recommended dose has not been studied and is therefore not recommended.
2.2 Special Precautions
- Avoid showering/bathing for at least 30 minutes after the application of PENNSAID to the treated knee.
- Wash and dry hands after use.
- Do not apply PENNSAID to open wounds.
- Avoid contact of PENNSAID with eyes and mucous membranes.
- Do not apply external heat and/or occlusive dressings to treated knees.
- Avoid wearing clothing over the PENNSAID-treated knee(s) until the treated knee is dry.
- Protect the treated knee(s) from natural and artificial sunlight.
- Wait until the treated area is dry before applying sunscreen, insect repellant, lotion, moisturizer, cosmetics, or other topical medication to the same knee you have just treated with PENNSAID.
- Until the treated knee(s) is completely dry, avoid skin-to-skin contact between other people and the treated knee(s).
- Do not use combination therapy with PENNSAID and an oral NSAID unless the benefit outweighs the risk and conduct periodic laboratory evaluations.
4. Contraindications
PENNSAID is contraindicated in the following patients:
- Known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to diclofenac or any components of the drug product [see Warnings and Precautions (5.7, 5.9)]
- History of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients [see Warnings and Precautions (5.7, 5.8)]
- In the setting of coronary artery bypass graft (CABG) surgery [see Warnings and Precautions (5.1)]
5. Warnings and Precautions
5.1 Cardiovascular Thrombotic Events
Clinical trials of several COX-2 selective and nonselective NSAIDs of up to three years duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, including myocardial infarction (MI), and stroke, which can be fatal. Based on available data, it is unclear that the risk for CV thrombotic events is similar for all NSAIDs. The relative increase in serious CV thrombotic events over baseline conferred by NSAID use appears to be similar in those with and without known CV disease or risk factors for CV disease. However, patients with known CV disease or risk factors had a higher absolute incidence of excess serious CV thrombotic events, due to their increased baseline rate. Some observational studies found that this increased risk of serious CV thrombotic events began as early as the first weeks of treatment. The increase in CV thrombotic risk has been observed most consistently at higher doses.
To minimize the potential risk for an adverse CV event in NSAID-treated patients, use the lowest effective dose for the shortest duration possible. Physicians and patients should remain alert for the development of such events, throughout the entire treatment course, even in the absence of previous CV symptoms. Patients should be informed about the symptoms of serious CV events and the steps to take if they occur.
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID, such as diclofenac, increases the risk of serious gastrointestinal (GI) events [see Warnings and Precautions (5.2)].
5.2 Gastrointestinal Bleeding, Ulceration, and Perforation
NSAIDs, including diclofenac, cause serious gastrointestinal (GI) adverse events including inflammation, bleeding, ulceration, and perforation of the esophagus, stomach, small intestine, or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with NSAIDs. Only one in five patients who develop a serious upper GI adverse event on NSAID therapy is symptomatic. Upper GI ulcers, gross bleeding, or perforation caused by NSAIDs occurred in approximately 1% of patients treated for 3-6 months, and in about 2%-4% of patients treated for one year. However, even short-term NSAID therapy is not without risk.
5.3 Hepatotoxicity
In clinical trials of oral diclofenac containing products, meaningful elevations (i.e., more than 3 times the ULN) of AST (SGOT) occurred in about 2% of approximately 5,700 patients at some time during diclofenac treatment (ALT was not measured in all studies).
In a large, open-label, controlled trial of 3,700 patients treated with oral diclofenac for 2 - 6 months, patients were monitored first at 8 weeks and 1,200 patients were monitored again at 24 weeks. Meaningful elevations of ALT and/or AST occurred in about 4% of 3,700 patients and included marked elevations (greater than 8 times the ULN) in about 1% of the 3,700 patients. In that open-label study, a higher incidence of borderline (less than 3 times the ULN), moderate (3 to 8 times the ULN), and marked (greater than 8 times the ULN) elevations of ALT or AST was observed in patients receiving diclofenac when compared to other NSAIDs. Elevations in transaminases were seen more frequently in patients with osteoarthritis than in those with rheumatoid arthritis.
Almost all meaningful elevations in transaminases were detected before patients became symptomatic. Abnormal tests occurred during the first 2 months of therapy with oral diclofenac in 42 of the 51 patients in all trials who developed marked transaminase elevations.
In postmarketing reports, cases of drug-induced hepatotoxicity have been reported in the first month, and in some cases, the first 2 months of NSAID therapy, but can occur at any time during treatment with diclofenac.
Postmarketing surveillance has reported cases of severe hepatic reactions, including liver necrosis, jaundice, fulminant hepatitis with and without jaundice, and liver failure. Some of these reported cases resulted in fatalities or liver transplantation.
In a European retrospective population-based, case-controlled study, 10 cases of oral diclofenac associated drug-induced liver injury with current use compared with non-use of diclofenac were associated with a statistically significant 4-fold adjusted odds ratio of liver injury. In this particular study, based on an overall number of 10 cases of liver injury associated with diclofenac, the adjusted odds ratio increased further with female gender, doses of 150 mg or more, and duration of use for more than 90 days.
Physicians should measure transaminases at baseline and periodically in patients receiving long-term therapy with diclofenac, because severe hepatotoxicity may develop without a prodrome of distinguishing symptoms. The optimum times for making the first and subsequent transaminase measurements are not known. Based on clinical trial data and postmarketing experiences, transaminases should be monitored within 4 to 8 weeks after initiating treatment with diclofenac. However, severe hepatic reactions can occur at any time during treatment with diclofenac.
If abnormal liver tests persist or worsen, if clinical signs and/or symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, abdominal pain, diarrhea, dark urine, etc.), PENNSAID should be discontinued immediately.
Inform patients of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, diarrhea, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, etc.), discontinue PENNSAID immediately, and perform a clinical evaluation of the patient.
To minimize the potential risk for an adverse liver-related event in patients treated with PENNSAID, use the lowest effective dose for the shortest duration possible. Exercise caution when prescribing PENNSAID with concomitant drugs that are known to be potentially hepatotoxic (e.g., acetaminophen, antibiotics, antiepileptics).
5.4 Hypertension
NSAIDs, including PENNSAID, can lead to new onset of hypertension or worsening of preexisting hypertension, either of which may contribute to the increased incidence of CV events. Patients taking angiotensin converting enzyme (ACE) inhibitors, thiazide diuretics, or loop diuretics may have impaired response to these therapies when taking NSAIDs [see Drug Interactions (7)].
Monitor blood pressure (BP) during the initiation of NSAID treatment and throughout the course of therapy.
5.5 Heart Failure and Edema
The Coxib and traditional NSAID Trialists' Collaboration meta-analysis of randomized controlled trials demonstrated an approximately two-fold increase in hospitalizations for heart failure in COX-2 selective treated patients and nonselective NSAID-treated patients compared to placebo-treated patients. In a Danish National Registry study of patients with heart failure, NSAID use increased the risk of MI, hospitalization for heart failure, and death.
Additionally, fluid retention and edema have been observed in some patients treated with NSAIDs. Use of diclofenac may blunt the CV effects of several therapeutic agents used to treat these medical conditions (e.g., diuretics, ACE inhibitors, or angiotensin receptor blockers [ARBs]) [see Drug Interactions (7)].
Avoid the use of PENNSAID in patients with severe heart failure unless the benefits are expected to outweigh the risk of worsening heart failure. If PENNSAID is used in patients with severe heart failure, monitor patients for signs of worsening heart failure.
5.7 Anaphylactic Reactions
Diclofenac has been associated with anaphylactic reactions in patients with and without known hypersensitivity to diclofenac and in patients with aspirin-sensitive asthma [see Contraindications (4) and Warnings and Precautions (5.8)].
Seek emergency help if an anaphylactic reaction occurs.
5.8 Exacerbation of Asthma Related to Aspirin Sensitivity
A subpopulation of patients with asthma may have aspirin-sensitive asthma which may include chronic rhinosinusitis complicated by nasal polyps; severe, potentially fatal bronchospasm; and/or intolerance to aspirin and other NSAIDs. Because cross-reactivity between aspirin and other NSAIDs has been reported in such aspirin-sensitive patients, PENNSAID is contraindicated in patients with this form of aspirin sensitivity [see Contraindications (4)]. When PENNSAID is used in patients with preexisting asthma (without known aspirin sensitivity), monitor patients for changes in the signs and symptoms of asthma.
5.9 Serious Skin Reactions
NSAIDs, including diclofenac, can cause serious skin adverse reactions such as exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. These serious events may occur without warning. Inform patients about the signs and symptoms of serious skin reactions, and to discontinue the use of PENNSAID at the first appearance of skin rash or any other sign of hypersensitivity. PENNSAID is contraindicated in patients with previous serious skin reactions to NSAIDs [see Contraindications (4)].
Do not apply PENNSAID to open skin wounds, infections, inflammations, or exfoliative dermatitis, as it may affect absorption and tolerability of the drug.
5.10 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) has been reported in patients taking NSAIDs such as PENNSAID. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling. Other clinical manifestations may include hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis. Sometimes symptoms of DRESS may resemble an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its presentation, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, discontinue PENNSAID and evaluate the patient immediately.
5.12 Hematologic Toxicity
Anemia has occurred in NSAID-treated patients. This may be due to occult or gross blood loss, fluid retention, or an incompletely described effect on erythropoiesis. If a patient treated with PENNSAID has any signs or symptoms of anemia, monitor hemoglobin or hematocrit.
NSAIDs, including PENNSAID, may increase the risk of bleeding events. Co-morbid conditions such as coagulation disorders or concomitant use of warfarin, other anticoagulants, antiplatelet agents (e.g., aspirin), serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs) may increase this risk. Monitor these patients for signs of bleeding [see Drug Interactions (7)].
5.13 Masking of Inflammation and Fever
The pharmacological activity of PENNSAID in reducing inflammation, and possibly fever, may diminish the utility of diagnostic signs in detecting infections.
5.14 Laboratory Monitoring
Because serious GI bleeding, hepatotoxicity, and renal injury can occur without warning symptoms or signs, consider monitoring patients on long-term NSAID treatment with a CBC and a chemistry profile periodically [see Warnings and Precautions (5.2, 5.3, 5.6)].
5.15 Sun Exposure
Instruct patients to avoid exposure to natural or artificial sunlight on treated knee(s) because studies in animals indicated topical diclofenac treatment resulted in an earlier onset of ultraviolet light-induced skin tumors. The potential effects of PENNSAID on skin response to ultraviolet damage in humans are not known.
5.16 Eye Exposure
Avoid contact of PENNSAID with eyes and mucosa. Advise patients that if eye contact occurs, immediately wash out the eye with water or saline and consult a physician if irritation persists for more than an hour.
5.17 Oral Nonsteroidal Anti-Inflammatory Drugs
Concomitant use of oral NSAIDs with PENNSAID 1.5% resulted in a higher rate of rectal hemorrhage, more frequent abnormal creatinine, urea and hemoglobin. Therefore, do not use combination therapy with PENNSAID and an oral NSAID unless the benefit outweighs the risk and conduct periodic laboratory evaluations.
6. Adverse Reactions/Side Effects
The following adverse reactions are discussed in greater detail in other sections of the labeling:
- Cardiovascular Thrombotic Events [see Warnings and Precautions (5.1)]
- GI Bleeding, Ulceration and Perforation [see Warnings and Precautions (5.2)]
- Hepatotoxicity [see Warnings and Precautions (5.3)]
- Hypertension [see Warnings and Precautions (5.4)]
- Heart Failure and Edema [see Warnings and Precautions (5.5)]
- Renal Toxicity and Hyperkalemia [see Warnings and Precautions (5.6)]
- Anaphylactic Reactions [see Warnings and Precautions (5.7)]
- Serious Skin Reactions [see Warnings and Precautions (5.9)]
- Hematologic Toxicity [see Warnings and Precautions (5.12)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
PENNSAID 1.5%
The safety of PENNSAID 2% is based in part, on prior experience with PENNSAID 1.5%. The data described below reflect exposure to PENNSAID 1.5% of 911 patients treated between 4 and 12 weeks (mean duration of 49 days) in seven Phase 3 controlled trials, as well as exposure of 793 patients treated in an open-label study, including 463 patients treated for at least 6 months, and 144 patients treated for at least 12 months. The population mean age was approximately 60 years, 89% of patients were Caucasian, 64% were females, and all patients had primary osteoarthritis. The most common adverse events with PENNSAID 1.5% were application site skin reactions. These events were the most common reason for withdrawing from the studies.
Other Common Adverse Reactions:
In controlled trials, subjects treated with PENNSAID 1.5% experienced some adverse events associated with the NSAID class more frequently than subjects using placebo (constipation, diarrhea, dyspepsia, nausea, flatulence, abdominal pain, edema; see Table 2). The combination of PENNSAID 1.5% and oral diclofenac, compared to oral diclofenac alone, resulted in a higher rate of rectal hemorrhage (3% vs. less than 1%), and more frequent abnormal creatinine (12% vs. 7%), urea (20% vs. 12%), and hemoglobin (13% vs. 9%), but no difference in elevation of liver transaminases.
Table 2 lists all adverse reactions occurring in ≥1% of patients receiving PENNSAID 1.5%, where the rate in the PENNSAID 1.5% group exceeded placebo, from seven controlled studies conducted in patients with osteoarthritis. Since these trials were of different durations, these percentages do not capture cumulative rates of occurrence.
| Treatment Group: | PENNSAID 1.5% N=911 | Topical Placebo N=332 |
|---|---|---|
| Adverse Reaction | N (%) | N (%) |
| Dry Skin (Application Site) | 292 (32) | 17 (5) |
| Contact Dermatitis (Application Site) | 83 (9) | 6 (2) |
| Dyspepsia | 72 (8) | 13 (4) |
| Abdominal Pain | 54 (6) | 10 (3) |
| Flatulence | 35 (4) | 1 (<1) |
| Pruritus (Application Site) | 34 (4) | 7 (2) |
| Diarrhea | 33 (4) | 7 (2) |
| Nausea | 33 (4) | 3 (1) |
| Pharyngitis | 40 (4) | 13 (4) |
| Constipation | 29 (3) | 1 (<1) |
| Edema | 26 (3) | 0 |
| Rash (Non-Application Site) | 25 (3) | 5 (2) |
| Infection | 25 (3) | 8 (2) |
| Ecchymosis | 19 (2) | 1 (<1) |
| Dry Skin (Non-Application Site) | 19 (2) | 1 (<1) |
| Contact Dermatitis, vesicles (Application Site) | 18 (2) | 0 |
| Paresthesia (Non-Application Site) | 14 (2) | 3 (<1) |
| Accidental Injury | 22 (2) | 7 (2) |
| Pruritus (Non-Application Site) | 15 (2) | 2 (<1) |
| Sinusitis | 10 (1) | 2 (<1) |
| Halitosis | 11 (1) | 1 (<1) |
| Application Site Reaction (not otherwise specified) | 11 (1) | 3 (<1) |
6.2 Postmarketing Experience
In postmarketing surveillance, the following adverse reactions have been reported during post- approval use of PENNSAID 1.5%. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body as a Whole: abdominal pain, accidental injury, allergic reactions, asthenia, back pain, body odor, chest pain, edema, face edema, halitosis, headache, neck rigidity, pain
Cardiovascular: palpitation, cardiovascular disorder
Gastrointestinal: diarrhea, dry mouth, dyspepsia, gastroenteritis, decreased appetite, lip swelling, mouth ulceration, nausea, rectal hemorrhage, ulcerative stomatitis, swollen tongue
Metabolic and Nutritional: creatinine increased
Musculoskeletal: leg cramps, myalgia
Nervous: depression, dizziness, drowsiness, lethargy, paresthesia at application site
Respiratory: asthma, dyspnea, laryngismus, laryngitis, pharyngitis, throat swelling
Skin and Appendages: At the Application Site: rash, skin burning sensation;
Other Skin and Appendages Adverse Reactions: eczema, skin discoloration, urticaria
Special Senses: abnormal vision, blurred vision, cataract, ear pain, eye disorder, eye pain, taste perversion
Vascular: blood pressure increased, hypertension
7. Drug Interactions
See Table 3 for clinically significant drug interactions with diclofenac.
| Drugs That Interfere with Hemostasis | |
|---|---|
| Clinical Impact: |
|
| Intervention: | Monitor patients with concomitant use of PENNSAID with anticoagulants (e.g., warfarin), antiplatelet agents (e.g., aspirin), selective serotonin reuptake inhibitors (SSRIs), and serotonin norepinephrine reuptake inhibitors (SNRIs) for signs of bleeding [see Warnings and Precautions (5.12)] |
| Aspirin | |
| Clinical Impact: | Controlled clinical studies showed that the concomitant use of NSAIDs and analgesic doses of aspirin does not produce any greater therapeutic effect than the use of NSAIDs alone. In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [see Warnings and Precautions (5.2)] |
| Intervention: | Concomitant use of PENNSAID and analgesic doses of aspirin is not generally recommended because of the increased risk of bleeding [see Warnings and Precautions (5.12)]. PENNSAID is not a substitute for low dose aspirin for cardiovascular protection. |
| ACE Inhibitors, Angiotensin Receptor Blockers, and Beta-Blockers | |
| Clinical Impact: |
|
| Intervention: |
|
| Diuretics | |
| Clinical Impact: | Clinical studies, as well as post-marketing observations, showed that NSAIDs reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. |
| Intervention: | During concomitant use of PENNSAID with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects [see Warnings and Precautions (5.6)]. |
| Digoxin | |
| Clinical Impact: | The concomitant use of diclofenac with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. |
| Intervention: | During concomitant use of PENNSAID and digoxin, monitor serum digoxin levels. |
| Lithium | |
| Clinical Impact: | NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance. The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis. |
| Intervention: | During concomitant use of PENNSAID and lithium, monitor patients for signs of lithium toxicity. |
| Methotrexate | |
| Clinical Impact: | Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction) |
| Intervention: | During concomitant use of PENNSAID and methotrexate, monitor patients for methotrexate toxicity. |
| Cyclosporine | |
| Clinical Impact: | Concomitant use of PENNSAID and cyclosporine may increase cyclosporine's nephrotoxicity. |
| Intervention: | During concomitant use of PENNSAID and cyclosporine, monitor patients for signs of worsening renal function. |
| NSAIDs and Salicylates | |
| Clinical Impact: | Concomitant use of diclofenac with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity, with little or no increase in efficacy [see Warnings and Precautions (5.2)] Concomitant use of oral NSAIDs with PENNSAID has been evaluated in one Phase 3 controlled trial and in combination with oral diclofenac, compared to oral diclofenac alone, resulted in a higher rate of rectal hemorrhage (3% vs. less than 1%), and more frequent abnormal creatinine (12% vs. 7%), urea (20% vs. 12%) and hemoglobin (13% vs. 9%). |
| Intervention: | The concomitant use of diclofenac with other NSAIDs or salicylates is not recommended. Do not use combination therapy with PENNSAID and an oral NSAID unless the benefit outweighs the risk and conduct periodic laboratory evaluations. |
| Pemetrexed | |
| Clinical Impact: | Concomitant use of PENNSAID and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). |
| Intervention: | During concomitant use of PENNSAID and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity. NSAIDs with short elimination half-lives (e.g., diclofenac, indomethacin) should be avoided for a period of two days before, the day of, and two days following administration of pemetrexed. In the absence of data regarding potential interaction between pemetrexed and NSAIDs with longer half-lives (e.g., meloxicam, nabumetone), patients taking these NSAIDs should interrupt dosing for at least five days before, the day of, and two days following pemetrexed administration. |
8. Use In Specific Populations
8.1 Pregnancy
8.5 Geriatric Use
Elderly patients, compared to younger patients, are at greater risk for NSAID-associated serious cardiovascular, gastrointestinal, and/or renal adverse reactions. If the anticipated benefit for the elderly patient outweighs these potential risks, start dosing at the low end of the dosing range, and monitor patients for adverse effects [see Warnings and Precautions (5.1, 5.2, 5.3, 5.6, 5.14)].
Of the 911 patients treated with PENNSAID 1.5% in seven controlled, Phase 3 clinical trials, 444 subjects were 65 years of age and over. There was no age-related difference in the incidence of adverse events. Of the 793 patients treated with PENNSAID 1.5% in one open-labeled safety trial, 334 subjects were 65 years of age and over including 107 subjects 75 and over. There was no difference in the incidence of adverse events with long-term exposure to PENNSAID 1.5% for this elderly population.
10. Overdosage
Symptoms following acute NSAID overdosages have been typically limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which have been generally reversible with supportive care. Gastrointestinal bleeding has occurred. Hypertension, acute renal failure, respiratory depression, and coma have occurred but were rare [see Warnings and Precautions (5.1, 5.2, 5.4, 5.6)].
Manage patients with symptomatic and supportive care following an NSAID overdosage. There are no specific antidotes. Emesis is not recommended due to a possibility of aspiration and subsequent respiratory irritation by DMSO contained in PENNSAID. Consider activated charcoal (60 to 100 grams in adults, 1 to 2 grams per kg of body weight in pediatric patients) and/or osmotic cathartic in symptomatic patients seen within four hours of ingestion or in patients with a large overdosage (5 to 10 times the recommended dosage). Forced diuresis, alkalinization of urine, hemodialysis, or hemoperfusion may not be useful due to high protein binding.
For additional information about overdosage treatment, contact a poison control center (1-800-222-1222).
11. Pennsaid Description
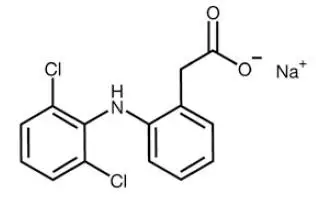
PENNSAID 2% topical solution, contains diclofenac sodium, a benzeneacetic acid derivative that is a nonsteroidal anti-inflammatory drug, and is available as a clear, colorless to faintly pink or orange solution for topical application. The chemical name is 2[(2,6-dichlorophenyl)amino]-benzeneacetic acid, monosodium salt. The molecular weight is 318.14. Its molecular formula is C14H10Cl2NNaO2, and it has the following chemical structure.
Each 1 gram of solution contains 20 mg of diclofenac sodium. The inactive ingredients: dimethyl sulfoxide USP (DMSO, 45.5% w/w), ethanol, purified water, propylene glycol, and hydroxypropyl cellulose.
12. Pennsaid - Clinical Pharmacology
12.1 Mechanism of Action
Diclofenac has analgesic, anti-inflammatory, and antipyretic properties.
The mechanism of action of PENNSAID, like that of other NSAIDs, is not completely understood but involves inhibition of cyclooxygenase (COX-1 and COX-2).
Diclofenac is a potent inhibitor of prostaglandin synthesis in vitro. Diclofenac concentrations reached during therapy have produced in vivo effects. Prostaglandins sensitize afferent nerves and potentiate the action of bradykinin in inducing pain in animal models. Prostaglandins are mediators of inflammation. Because diclofenac is an inhibitor of prostaglandin synthesis, its mode of action may be due to a decrease of prostaglandins in peripheral tissues.
13. Nonclinical Toxicology
16. How is Pennsaid supplied
PENNSAID (diclofenac sodium) topical solution 2% w/w, is supplied as a clear, colorless to faintly pink or orange solution containing 20 mg of diclofenac sodium per gram of solution, in a white polypropylene-dose pump bottle with a clear cap. Each pump actuation delivers 20 mg of diclofenac sodium in 1 gram of solution.
| NDC Number & Size | |
|---|---|
| 112 g bottle | NDC # 75987-040-05 |
17. Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide) that accompanies each prescription dispensed. Inform patients, families, or their caregivers of the following information before initiating therapy with PENNSAID and periodically during the course of ongoing therapy.
| PENNSAID
diclofenac sodium solution |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| Labeler - Horizon Therapeutics USA, Inc. (033470838) |