Drug Detail:Spiriva respimat (Tiotropium inhalation [ tye-oh-troe-pee-um ])
Drug Class: Anticholinergic bronchodilators
Highlights of Prescribing Information
SPIRIVA® RESPIMAT® (tiotropium bromide inhalation spray), for oral inhalation use
Initial U.S. Approval: 2004
Indications and Usage for Spiriva Respimat
SPIRIVA RESPIMAT is an anticholinergic indicated for:
- The long-term, once-daily, maintenance treatment of bronchospasm associated with chronic obstructive pulmonary disease (COPD), and for reducing COPD exacerbations (1.1)
- The long-term, once-daily, maintenance treatment of asthma in patients 6 years of age and older (1.2)
Limitation of Use:
- Not indicated for relief of acute bronchospasm (1.1, 1.2, 5.1)
Spiriva Respimat Dosage and Administration
For oral inhalation only
To receive the full dose of medication, SPIRIVA RESPIMAT must be administered as two inhalations once-daily.
- Treatment of COPD: 2 inhalations of SPIRIVA RESPIMAT 2.5 mcg once-daily (2)
- Treatment of asthma patients 6 years and older: 2 inhalations of SPIRIVA RESPIMAT 1.25 mcg once-daily (2)
Dosage Forms and Strengths
- Inhalation spray: 1.25 mcg or 2.5 mcg tiotropium per actuation with the SPIRIVA RESPIMAT inhaler. Two actuations equal one dose (2.5 mcg or 5 mcg). (3)
Contraindications
Hypersensitivity to tiotropium, ipratropium, or any component of this product (4)
Warnings and Precautions
- Not for acute use, i.e., not a rescue medication (5.1)
- Immediate hypersensitivity reactions: Discontinue SPIRIVA RESPIMAT at once and consider alternatives if immediate hypersensitivity reactions, including angioedema, urticaria, rash, bronchospasm, or anaphylaxis, occur. (5.2)
- Paradoxical bronchospasm: Discontinue SPIRIVA RESPIMAT and consider other treatments if paradoxical bronchospasm occurs. (5.3)
- Worsening of narrow-angle glaucoma may occur. Use with caution in patients with narrow-angle glaucoma and instruct patients to consult a physician immediately if this occurs. (5.4)
- Worsening of urinary retention may occur. Use with caution in patients with prostatic hyperplasia or bladder-neck obstruction and instruct patient to consult a physician immediately if this occurs. (5.5)
Adverse Reactions/Side Effects
The most common adverse reactions in:
- COPD: (>3% incidence in the placebo-controlled trials with treatment durations of between 4 and 48 weeks) were pharyngitis, cough, dry mouth, and sinusitis (6.1).
- Asthma: (>2% incidence in the placebo-controlled trials with treatment durations of between 12 and 52 weeks) were pharyngitis, headache, bronchitis, and sinusitis in adults (6.2).
To report SUSPECTED ADVERSE REACTIONS, contact Boehringer Ingelheim Pharmaceuticals, Inc. at (800) 542-6257 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Anticholinergics: May interact additively with concomitantly used anticholinergic medications. Avoid administration of SPIRIVA RESPIMAT with other anticholinergic-containing drugs. (7.2)
Use In Specific Populations
Patients with moderate to severe renal impairment should be monitored closely for potential anticholinergic side effects. (2, 8.6)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 11/2021
Related/similar drugs
Dupixent, Xolair, Symbicort, Breo Ellipta, Ventolin, Spiriva, Ventolin HFAFull Prescribing Information
1. Indications and Usage for Spiriva Respimat
1.1 Maintenance Treatment of Chronic Obstructive Pulmonary Disease
SPIRIVA RESPIMAT (tiotropium bromide) is indicated for the long-term, once-daily, maintenance treatment of bronchospasm associated with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and emphysema. SPIRIVA RESPIMAT is indicated to reduce exacerbations in COPD patients.
2. Spiriva Respimat Dosage and Administration
To receive the full dose of medication, SPIRIVA RESPIMAT must be administered as two inhalations once-daily. Do not take more than one dose (2 inhalations) in 24 hours.
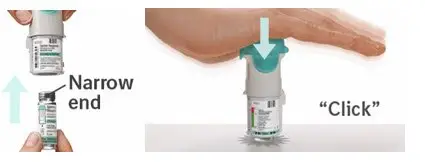
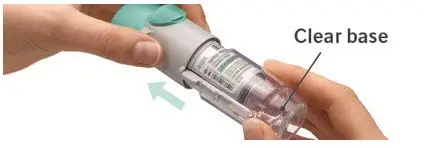
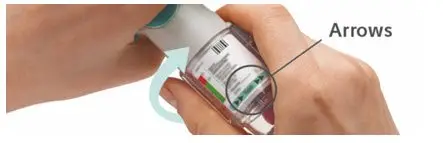
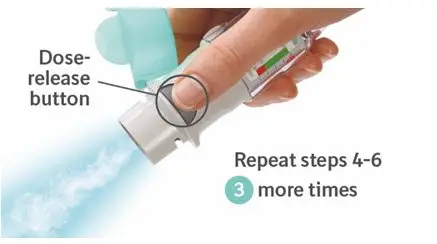
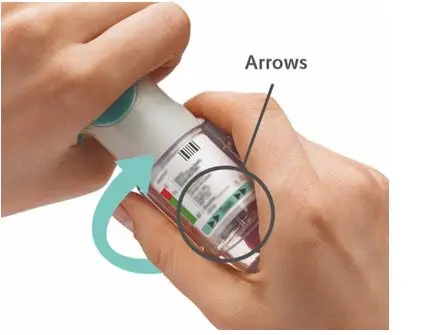
Prior to first use, the SPIRIVA RESPIMAT cartridge is inserted into the SPIRIVA RESPIMAT inhaler and the unit is primed. When using the unit for the first time, patients are to actuate the inhaler toward the ground until an aerosol cloud is visible and then repeat the process three more times. The unit is then considered primed and ready for use. If not used for more than 3 days, patients are to actuate the inhaler once to prepare the inhaler for use. If not used for more than 21 days, patients are to actuate the inhaler until an aerosol cloud is visible and then repeat the process three more times to prepare the inhaler for use [see Patient Counseling Information (17)].
2.1 Chronic Obstructive Pulmonary Disease
The recommended dosage for patients with COPD is 2 inhalations of SPIRIVA RESPIMAT 2.5 mcg per actuation once-daily; total dose equals 5 mcg of SPIRIVA RESPIMAT.
2.2 Asthma
The recommended dosage for patients with asthma is 2 inhalations of SPIRIVA RESPIMAT 1.25 mcg per actuation once-daily; total dose equals 2.5 mcg of SPIRIVA RESPIMAT. In the treatment of asthma, the maximum benefits in lung function may take up to 4 to 8 weeks of dosing [see Patient Counseling Information (17)].
2.3 Special Populations
No dosage adjustment is required for geriatric, hepatically-impaired, or renally-impaired patients. However, patients with moderate to severe renal impairment given SPIRIVA RESPIMAT should be monitored closely for anticholinergic effects [see Warnings and Precautions (5.6), Use in Specific Populations (8.5, 8.6, 8.7), and Clinical Pharmacology (12.3)].
3. Dosage Forms and Strengths
SPIRIVA RESPIMAT consists of a SPIRIVA RESPIMAT inhaler and an aluminum cylinder (SPIRIVA RESPIMAT cartridge) containing tiotropium bromide (as the monohydrate). The SPIRIVA RESPIMAT cartridge is only intended for use with the SPIRIVA RESPIMAT inhaler.
SPIRIVA RESPIMAT is available in two dosage strengths. Each actuation from the SPIRIVA RESPIMAT inhaler delivers 1.25 mcg or 2.5 mcg of tiotropium (equivalent to 1.562 mcg or 3.124 mcg, respectively, of tiotropium bromide monohydrate) from the mouthpiece. Two actuations equal one dose (2.5 mcg or 5 mcg).
4. Contraindications
SPIRIVA RESPIMAT is contraindicated in patients with a hypersensitivity to tiotropium, ipratropium, or any component of this product [see Warnings and Precautions (5.2)]. In clinical trials with SPIRIVA RESPIMAT, immediate hypersensitivity reactions, including angioedema (including swelling of the lips, tongue, or throat), itching, or rash have been reported [see Warnings and Precautions (5.2)].
5. Warnings and Precautions
5.1 Not for Acute Use
SPIRIVA RESPIMAT is intended as a once-daily maintenance treatment for COPD and asthma and should not be used for the relief of acute symptoms, i.e., as rescue therapy for the treatment of acute episodes of bronchospasm. In the event of an acute attack, a rapid-acting beta2-agonist should be used.
5.2 Immediate Hypersensitivity Reactions
Immediate hypersensitivity reactions, including urticaria, angioedema (including swelling of the lips, tongue or throat), rash, bronchospasm, anaphylaxis, or itching may occur after administration of SPIRIVA RESPIMAT. If such a reaction occurs, therapy with SPIRIVA RESPIMAT should be stopped at once and alternative treatments should be considered. Given the similar structural formula of atropine to tiotropium, patients with a history of hypersensitivity reactions to atropine or its derivatives should be closely monitored for similar hypersensitivity reactions to SPIRIVA RESPIMAT.
5.3 Paradoxical Bronchospasm
Inhaled medicines, including SPIRIVA RESPIMAT, may cause paradoxical bronchospasm. If this occurs, it should be treated immediately with an inhaled short-acting beta2-agonist such as albuterol. Treatment with SPIRIVA RESPIMAT should be stopped and other treatments considered.
5.4 Worsening of Narrow-Angle Glaucoma
SPIRIVA RESPIMAT should be used with caution in patients with narrow-angle glaucoma. Prescribers and patients should be alert for signs and symptoms of acute narrow-angle glaucoma (e.g., eye pain or discomfort, blurred vision, visual halos or colored images in association with red eyes from conjunctival congestion and corneal edema). Instruct patients to consult a physician immediately should any of these signs or symptoms develop.
5.5 Worsening of Urinary Retention
SPIRIVA RESPIMAT should be used with caution in patients with urinary retention. Prescribers and patients should be alert for signs and symptoms of urinary retention (e.g., difficulty passing urine, painful urination), especially in patients with prostatic hyperplasia or bladder-neck obstruction. Instruct patients to consult a physician immediately should any of these signs or symptoms develop.
6. Adverse Reactions/Side Effects
The following adverse reactions are described, or described in greater detail, in other sections:
- Immediate hypersensitivity reactions [see Warnings and Precautions (5.2)]
- Paradoxical bronchospasm [see Warnings and Precautions (5.3)]
- Worsening of narrow-angle glaucoma [see Warnings and Precautions (5.4)]
- Worsening of urinary retention [see Warnings and Precautions (5.5)]
Because clinical trials are conducted under widely varying conditions, the incidence of adverse reactions observed in the clinical trials of a drug cannot be directly compared to the incidences in the clinical trials of another drug and may not reflect the incidences observed in practice.
Since the same active ingredient (tiotropium bromide) is administered to COPD and asthma patients, prescribers and patients should take into account that the observed adverse reactions could be relevant for both patient populations independent of dosage strength.
6.1 Clinical Trials Experience in Chronic Obstructive Pulmonary Disease
The SPIRIVA RESPIMAT clinical development program included ten placebo controlled clinical trials in COPD. Two trials were four-week cross-over trials and eight were parallel group trials. The parallel group trials included a three week dose-ranging trial, two 12-week trials, three 48-week trials, and two trials of 4-week and 24-week duration conducted for a different program that contained tiotropium bromide 5 mcg treatment arms. The primary safety database consists of pooled data from the 7 randomized, parallel-group, double-blind, placebo-controlled studies of 4-48 weeks in treatment duration. These trials included 6565 adult COPD patients (75% males and 25% females) 40 years of age and older. Of these patients, 3282 patients were treated with SPIRIVA RESPIMAT 5 mcg and 3283 received placebo. The SPIRIVA RESPIMAT 5 mcg group was composed mostly of Caucasians (78%) with a mean age of 65 years and a mean baseline percent predicted post-bronchodilator FEV1 of 46%.
In these 7 clinical trials, 68.3% of patients exposed to SPIRIVA RESPIMAT 5 mcg reported an adverse event compared to 68.7% of patients in the placebo group. There were 68 deaths in the SPIRIVA RESPIMAT 5 mcg treatment group (2.1%) and 52 deaths (1.6%) in patients who received placebo [see Clinical Studies (14) Long-term Active-Controlled Mortality Trial: Survival]. The percentage of SPIRIVA RESPIMAT patients who discontinued due to an adverse event were 7.3% compared to 10% with placebo patients. The percentage of SPIRIVA RESPIMAT 5 mcg patients who experienced a serious adverse event were 15.0% compared to 15.1% with placebo patients. In both groups, the adverse event most commonly leading to discontinuation was COPD exacerbation (SPIRIVA RESPIMAT 2.0%, placebo 4.0%) which was also the most frequent serious adverse event. The most commonly reported adverse reactions were pharyngitis, cough, dry mouth, and sinusitis (Table 1). Other adverse reactions reported in individual patients and consistent with possible anticholinergic effects included constipation, dysuria, and urinary retention.
Table 1 shows all adverse reactions that occurred with an incidence of >3% in the SPIRIVA RESPIMAT 5 mcg treatment group, and a higher incidence rate on SPIRIVA RESPIMAT 5 mcg than on placebo.
| Body System (Reaction)* | SPIRIVA RESPIMAT 5 mcg [n=3282] | Placebo [n=3283] |
|---|---|---|
| *Adverse reactions include a grouping of similar terms | ||
| Gastrointestinal Disorders | ||
| Dry mouth | 134 (4.1) | 52 (1.6) |
| Infections and Infestations | ||
| Pharyngitis | 378 (11.5) | 333 (10.1) |
| Respiratory, Thoracic, and Mediastinal Disorders | ||
| Cough | 190 (5.8) | 182 (5.5) |
| Sinusitis | 103 (3.1) | 88 (2.7) |
Other reactions that occurred in the SPIRIVA RESPIMAT 5 mcg group at an incidence of 1% to 3% and at a higher incidence rate on SPIRIVA RESPIMAT 5 mcg than on placebo included: Cardiac disorders: palpitations; Gastrointestinal disorders: constipation, gastroesophageal reflux disease, oropharyngeal candidiasis; Nervous system disorders: dizziness; Respiratory, thoracic, and mediastinal disorders: dysphonia; Skin and subcutaneous tissue disorders: pruritus, rash; Renal and urinary disorders: urinary tract infection.
6.3 Postmarketing Experience
In addition to the adverse reactions observed during the SPIRIVA RESPIMAT clinical trials in COPD, the following adverse reactions have been observed during post-approval use of SPIRIVA RESPIMAT 5 mcg and another tiotropium formulation, SPIRIVA® HandiHaler® (tiotropium bromide inhalation powder). Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Glaucoma, intraocular pressure increased, vision blurred,
- Atrial fibrillation, tachycardia, supraventricular tachycardia,
- Bronchospasm,
- Glossitis, stomatitis,
- Dehydration,
- Insomnia,
- Hypersensitivity (including immediate reactions), and urticaria.
7. Drug Interactions
7.1 Concomitant Respiratory Medications
SPIRIVA RESPIMAT has been used concomitantly with short-acting and long-acting sympathomimetic (beta-agonists) bronchodilators, methylxanthines, oral and inhaled steroids, antihistamines, mucolytics, leukotriene modifiers, cromones, and anti-IgE treatment without increases in adverse reactions.
7.2 Anticholinergics
There is potential for an additive interaction with concomitantly used anticholinergic medications. Therefore, avoid coadministration of SPIRIVA RESPIMAT with other anticholinergic-containing drugs as this may lead to an increase in anticholinergic adverse effects [see Warnings and Precautions (5.4, 5.5) and Adverse Reactions (6)].
8. Use In Specific Populations
8.4 Pediatric Use
The safety and efficacy of SPIRIVA RESPIMAT 2.5 mcg have been established in pediatric patients aged 6 to 17 years with asthma in 6 clinical trials up to 1 year in duration. In three clinical trials, 327 patients aged 12 to 17 years with asthma were treated with SPIRIVA RESPIMAT 2.5 mcg; in three additional clinical trials, 345 patients aged 6 to 11 years with asthma were treated with SPIRIVA RESPIMAT 2.5 mcg. Patients in these age groups demonstrated efficacy results similar to those observed in patients aged 18 years and older with asthma [see Clinical Studies (14.2)].
The safety and efficacy of SPIRIVA RESPIMAT have not been established in pediatric patients less than 6 years of age. The safety of SPIRIVA RESPIMAT 2.5 mcg has been studied in pediatric patients with asthma aged 1 to 5 years who were on background treatment of at least ICS in one placebo-controlled clinical trial of 12 weeks duration (36 treated with SPIRIVA RESPIMAT 2.5 mcg and 34 with placebo RESPIMAT). In this study, SPIRIVA RESPIMAT or placebo RESPIMAT was delivered with the AeroChamber Plus Flow-Vu® valved holding chamber with facemask once daily. The majority of the patients in the trial were male (60.4%) and Caucasian (76.2%) with a mean age of 3.1 years. The adverse reaction profile was similar to that observed in adults and older pediatric patients [see Adverse Reactions (6.2)].
8.5 Geriatric Use
Based on available data, no adjustment of SPIRIVA RESPIMAT dosage in geriatric patients is warranted [see Clinical Pharmacology (12.3)].
Thirty nine percent of SPIRIVA RESPIMAT clinical trial patients with COPD were between 65 and 75 years of age and 14% were greater than or equal to 75 years of age. Approximately seven percent of SPIRIVA RESPIMAT clinical trial patients with asthma were greater than or equal to 65 years of age. The adverse drug reaction profiles were similar in the older population compared to the patient population overall.
8.6 Renal Impairment
Patients with moderate to severe renal impairment (creatinine clearance of <60 mL/min) treated with SPIRIVA RESPIMAT should be monitored closely for anticholinergic side effects [see Dosage and Administration (2), Warnings and Precautions (5.6), and Clinical Pharmacology (12.3)].
10. Overdosage
High doses of tiotropium may lead to anticholinergic signs and symptoms. However, there were no systemic anticholinergic adverse effects following a single inhaled dose of up to 282 mcg tiotropium dry powder in 6 healthy volunteers. Dry mouth/throat and dry nasal mucosa occurred in a dose-dependent [10-40 mcg daily] manner, following 14-day dosing of up to 40 mcg tiotropium bromide inhalation solution in healthy subjects.
Treatment of overdosage consists of discontinuation of SPIRIVA RESPIMAT together with institution of appropriate symptomatic and/or supportive therapy.
11. Spiriva Respimat Description
The active component of SPIRIVA RESPIMAT is tiotropium. The drug substance, tiotropium bromide monohydrate, is an anticholinergic with specificity for muscarinic receptors. It is chemically described as (1α, 2β, 4β, 5α, 7β)-7-[(Hydroxydi-2-thienylacetyl)oxy]-9,9-dimethyl-3-oxa-9-azoniatricyclo[3.3.1.02,4] nonane bromide monohydrate. It is a synthetic, non-chiral, quaternary ammonium compound. Tiotropium bromide is a white or yellowish white powder. It is sparingly soluble in water and soluble in methanol.
The structural formula is:
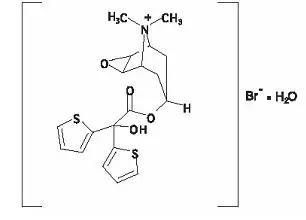
Tiotropium bromide (monohydrate) has a molecular mass of 490.4 and a molecular formula of C19H22NO4S2Br ∙ H2O.
The drug product, SPIRIVA RESPIMAT, is composed of a sterile, aqueous solution of tiotropium bromide filled into a 4.5 mL plastic container crimped into an aluminum cylinder (SPIRIVA RESPIMAT cartridge) for use with the SPIRIVA RESPIMAT inhaler. Excipients include water for injection, edetate disodium, benzalkonium chloride and hydrochloric acid. The SPIRIVA RESPIMAT cartridge is only intended for use with the SPIRIVA RESPIMAT inhaler. The RESPIMAT inhaler is a hand held, pocket sized oral inhalation device that uses mechanical energy to generate a slow moving aerosol cloud of medication from a metered volume of the drug solution.
When used with the SPIRIVA RESPIMAT inhaler, each cartridge containing 4 grams of sterile aqueous solution delivers the labeled number of metered actuations after preparation for use. Each dose (one dose equals two actuations) from the SPIRIVA RESPIMAT inhaler delivers 2.5 mcg or 5 mcg of tiotropium in 22.1 mcL from the mouthpiece. As with all inhaled drugs, the actual amount of drug delivered to the lung may depend on patient factors, such as the coordination between the actuation of the inhaler and inspiration through the delivery system. The duration of inspiration should be at least as long as the spray duration (1.5 seconds).
Prior to first use, the SPIRIVA RESPIMAT cartridge is inserted into the SPIRIVA RESPIMAT inhaler and the unit is primed. When using the unit for the first time, patients are to actuate the inhaler toward the ground until an aerosol cloud is visible and then repeat the process three more times. The unit is then considered primed and ready for use. If not used for more than 3 days, patients are to actuate the inhaler once to prepare the inhaler for use. If not used for more than 21 days, patients are to actuate the inhaler until an aerosol cloud is visible and then repeat the process three more times to prepare the inhaler for use [see Patient Counseling Information (17)].
12. Spiriva Respimat - Clinical Pharmacology
12.1 Mechanism of Action
Tiotropium is a long-acting, antimuscarinic agent, which is often referred to as an anticholinergic. It has similar affinity to the subtypes of muscarinic receptors, M1 to M5. In the airways, it exhibits pharmacological effects through inhibition of M3-receptors at the smooth muscle leading to bronchodilation. The competitive and reversible nature of antagonism was shown with human and animal origin receptors and isolated organ preparations. In preclinical in vitro as well as in vivo studies, prevention of methacholine-induced bronchoconstriction effects was dose-dependent and lasted longer than 24 hours. The bronchodilation following inhalation of tiotropium is predominantly a site-specific effect.
12.3 Pharmacokinetics
Tiotropium is administered as an inhalation spray. Some of the pharmacokinetic data described below were obtained with higher doses than recommended for therapy. A dedicated pharmacokinetic study in patients with COPD evaluating once-daily tiotropium delivered from the RESPIMAT inhaler (5 mcg) and as inhalation powder (18 mcg) from the HandiHaler resulted in a similar systemic exposure between the two products.
13. Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No evidence of tumorigenicity was observed in a 104-week inhalation study in rats at tiotropium doses up to 59 mcg/kg/day, in an 83-week inhalation study in female mice at doses up to 145 mcg/kg/day, and in a 101-week inhalation study in male mice at doses up to 2 mcg/kg/day. These doses correspond to approximately 30, 40, and 0.5, times the maximum recommended human daily inhalation dose (MRHDID) on a mcg/m2 basis, respectively.
Tiotropium bromide demonstrated no evidence of mutagenicity or clastogenicity in the following assays: the bacterial gene mutation assay, the V79 Chinese hamster cell mutagenesis assay, the chromosomal aberration assays in human lymphocytes in vitro and mouse micronucleus formation in vivo, and the unscheduled DNA synthesis in primary rat hepatocytes in vitro assay.
In rats, decreases in the number of corpora lutea and the percentage of implants were noted at inhalation tiotropium doses of 78 mcg/kg/day or greater (approximately 40 times the MRHDID on a mcg/m2 basis). No such effects were observed at 9 mcg/kg/day (approximately 5 times the MRHDID on a mcg/m2 basis). The fertility index, however, was not affected at inhalation doses up to 1689 mcg/kg/day (approximately 910 times the MRHDID on a mcg/m2 basis).
14. Clinical Studies
14.1 Chronic Obstructive Pulmonary Disease
The efficacy of SPIRIVA RESPIMAT compared to placebo was evaluated in 6 clinical trials: one dose-ranging trial and 5 confirmatory trials (Trials 1-5). In addition, SPIRIVA RESPIMAT was compared to SPIRIVA HandiHaler in a long-term active-controlled trial in COPD (Trial 6).
Long-term Active-Controlled Mortality Trial
Survival
In a pooled analysis of SPIRIVA RESPIMAT placebo-controlled clinical trials with complete vital status (mortality) follow-up, including the three 48-week trials (Trial 3, 4, and 5) and one 24-week placebo-controlled trial, 68 deaths (Incidence Rate 2.64 deaths per 100 patient years) were observed in the SPIRIVA RESPIMAT 5 mcg treatment group compared to 51 deaths (Incidence Rate 1.98 deaths per 100 patient years) in those treated with placebo. In a 4-year, randomized, double-blind, placebo-controlled, multicenter clinical trial of tiotropium bromide inhalation powder (SPIRIVA HandiHaler) in 5992 COPD patients a similar incidence rate of death had been observed between SPIRIVA HandiHaler and placebo treated groups.
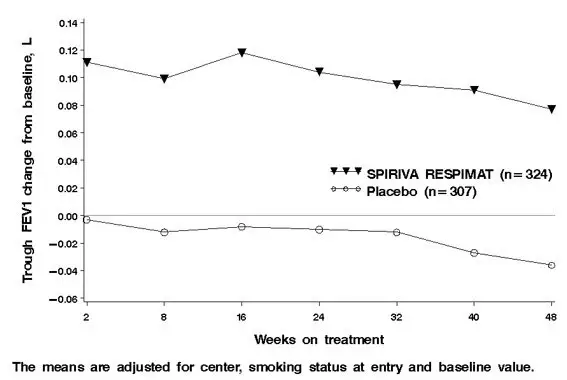
For clarification of the observed difference in fatal events, a long-term, randomized, double-blind, double dummy, active-controlled trial with an observation period up to 3 years was conducted to evaluate the risk of all-cause mortality associated with the use of SPIRIVA RESPIMAT compared to SPIRIVA HandiHaler (Trial 6). The objective of this trial was to rule out a relative excess mortality risk of 25% for SPIRIVA RESPIMAT versus SPIRIVA HandiHaler. The primary endpoints were all-cause mortality and time to first COPD exacerbation. Trial 6 also included a lung function sub-study which measured trough FEV1 measured every 24 weeks for 120 weeks (461 patients receiving SPIRIVA RESPIMAT 5 mcg, 445 patients receiving SPIRIVA HandiHaler).
In Trial 6, 5711 patients received SPIRIVA RESPIMAT 5 mcg and 5694 patients received SPIRIVA HandiHaler. All patients were followed for vital status (mortality) at the end of the trial. At baseline, patient characteristics were balanced between the two treatment arms. The mean age was 65 years and approximately 70% of subjects were male. Approximately, 82% of patients were Caucasian, 14% were Asian, and 2% were Black. Mean post-bronchodilator FEV1 was 1.34 L with a mean FEV1/FVC ratio of 50%. The majority of patients were GOLD II or III (48% and 40%, respectively).
The vital status was confirmed in 99.7% of patients. The median exposure to treatment was 835 days for both treatment groups. All-cause mortality was similar between SPIRIVA RESPIMAT 5 mcg and SPIRIVA HandiHaler with an estimated hazard ratio of 0.96 [(95% CI of (0.84 to 1.09), Table 5].
| SPIRIVA RESPIMAT 5 mcg (N = 5711) | SPIRIVA HandiHaler (N = 5694) |
|
|---|---|---|
| a Hazard ratios were estimated from a Cox proportional hazard model. | ||
| Number (%) of Deaths | 423 (7.4) | 439 (7.7) |
| Incidence Rate per 100 patient years | 3.22 | 3.36 |
| HR (95% CI)a | 0.96 (0.84, 1.09) | |
Cause of death was adjudicated by a blinded, independent committee. Cardiovascular deaths included cardiac death, sudden cardiac death, and sudden death; as well as fatal events caused by a cardiac disorder, vascular disorder, or stroke. There were 113 patients (2%) treated with SPIRIVA RESPIMAT 5 mcg who had cardiovascular deaths compared to 101 (2%) patients treated with SPIRIVA HandiHaler. Of the cardiovascular deaths, 11 (0.2%) and 3 (0.1%) deaths were due to myocardial infarction in SPIRIVA RESPIMAT 5 mcg patients and SPIRIVA HandiHaler patients, respectively. For cardiac deaths, sudden cardiac death, and sudden death, there were a total of 69 (1.2%) and 68 (1.2%) deaths in SPIRIVA RESPIMAT 5 mcg patients and SPIRIVA HandiHaler patients, respectively.
14.2 Asthma
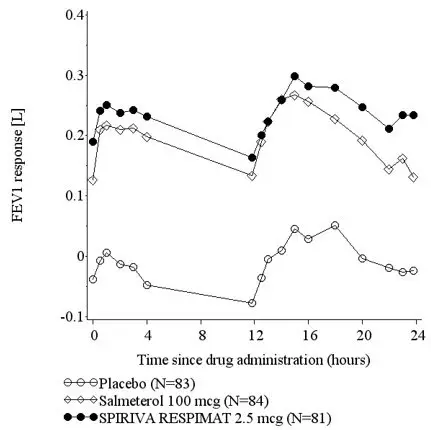
The SPIRIVA RESPIMAT clinical development program included six 4-week to 8-week cross-over design trials and ten 12-week to 48-week parallel-arm design trials in adult, adolescent (aged 12 to 17 years) and pediatric (aged 1 to 11 years) patients with asthma symptomatic on at least ICS. In all trials, SPIRIVA RESPIMAT was administered on a background of ICS therapy.
16. How is Spiriva Respimat supplied
SPIRIVA RESPIMAT Inhalation Spray is supplied in a carton containing one SPIRIVA RESPIMAT cartridge and one SPIRIVA RESPIMAT inhaler.
The SPIRIVA RESPIMAT cartridge is provided as an aluminum cylinder with a tamper protection seal on the cap. The SPIRIVA RESPIMAT cartridge is only intended for use with the SPIRIVA RESPIMAT inhaler and should not be interchanged with any other RESPIMAT device delivered product.
The SPIRIVA RESPIMAT inhaler is a cylindrical shaped plastic inhalation device with a gray colored body and a clear base. The clear base is removed to insert the cartridge. The inhaler contains a dose indicator. The written information on the label of the gray inhaler body indicates that it is labeled for use with the SPIRIVA RESPIMAT cartridge.
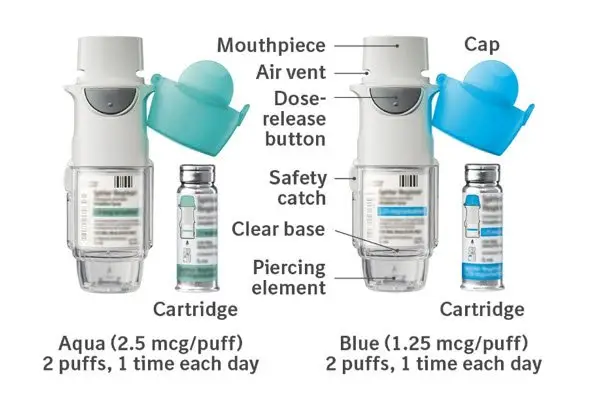
SPIRIVA RESPIMAT Inhalation Spray is available in two dosage strengths, identified by dose delivered per actuation and by the color of the cap and associated container label: aqua represents 2.5 mcg per actuation; blue represents 1.25 mcg per actuation.
To deliver the recommended dosage for COPD:
| 2.5 mcg/actuation | 60 metered actuations | (NDC 0597-0100-61) |
| 2.5 mcg/actuation | 10 metered actuations | (NDC 0597-0100-51) (institutional pack) |
To deliver the recommended dosage for asthma:
| 1.25 mcg/actuation | 60 metered actuations | (NDC 0597-0160-61) |
The SPIRIVA RESPIMAT cartridge for each strength has a net fill weight of 4 grams and when used with the SPIRIVA RESPIMAT inhaler, is designed to deliver the labeled number of metered actuations after preparation for use. Each actuation from the SPIRIVA RESPIMAT inhaler delivers 1.25 or 2.5 mcg of tiotropium (equivalent to 1.562 or 3.124 mcg, respectively, of tiotropium bromide monohydrate) from the mouthpiece.
When the labeled number of actuations has been dispensed from the inhaler, the RESPIMAT locking mechanism will be engaged and no more actuations can be dispensed.
After assembly, the SPIRIVA RESPIMAT inhaler should be discarded at the latest 3 months after first use or when the locking mechanism is engaged, whichever comes first.
Keep out of reach of children. Do not spray into eyes.
| SPIRIVA RESPIMAT
tiotropium bromide inhalation spray spray, metered |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| SPIRIVA RESPIMAT
tiotropium bromide inhalation spray spray, metered |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Boehringer Ingelheim Pharmaceuticals, Inc. (603175944) |
| Registrant - Boehringer Ingelheim Pharmaceuticals, Inc. (603175944) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Boehringer Ingelheim Pharma GmbH and Co. KG | 551147440 | API MANUFACTURE(0597-0100, 0597-0160) , LABEL(0597-0100, 0597-0160) , MANUFACTURE(0597-0100, 0597-0160) , PACK(0597-0100, 0597-0160) | |