Drug Detail:Benzaclin (Benzoyl peroxide and clindamycin topical [ ben-zoyl-per-ox-ide-and-clin-da-mye-sin ])
Drug Class: Topical acne agents
Topical Gel: clindamycin (1%) as clindamycin phosphate, benzoyl peroxide (5%)
For Dermatological Use Only - Not for Ophthalmic Use
*Reconstitute Before Dispensing*
BenzaClin Description
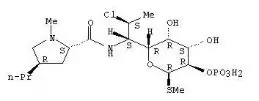
BenzaClin® Topical Gel contains clindamycin phosphate (7(S)-chloro-7-deoxylincomycin-2-phosphate). Clindamycin phosphate is a water soluble ester of the semi-synthetic antibiotic produced by a 7(S)-chloro-substitution of the 7(R)-hydroxyl group of the parent antibiotic lincomycin.
Chemically clindamycin phosphate is C18H34CIN2O8PS. The structural formula for clindamycin is represented below:
Clindamycin phosphate has a molecular weight of 504.97 and its chemical name is Methyl 7-chloro-6,7,8-trideoxy-6-(1-methyl-trans-4-propyl-L-2-pyrrolidinecarboxamido)-1-thio-L-threo-alpha-D-galacto-octopyranoside 2-(dihydrogen phosphate).
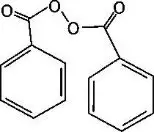
BenzaClin Topical Gel also contains benzoyl peroxide for topical use.
Chemically benzoyl peroxide is C14H10O4. It has the following structural formula:
Benzoyl peroxide has a molecular weight of 242.23.
Each gram of BenzaClin Topical Gel contains, as dispensed, 10 mg (1%) clindamycin as phosphate and 50 mg (5%) benzoyl peroxide in a base of carbomer, sodium hydroxide, dioctyl sodium sulfosuccinate, and purified water.
BenzaClin - Clinical Pharmacology
An in vitro percutaneous penetration study comparing BenzaClin Topical Gel and topical 1% clindamycin gel alone demonstrated there was no statistical difference in penetration between the two drugs. Mean systemic bioavailability of topical clindamycin in BenzaClin Topical Gel is suggested to be less than 1%.
Benzoyl peroxide has been shown to be absorbed by the skin where it is converted to benzoic acid. Less than 2% of the dose enters systemic circulation as benzoic acid. It is suggested that the lipophilic nature of benzoyl peroxide acts to concentrate the compound into the lipid-rich sebaceous follicle.
Pharmacokinetics: The pharmacokinetics (plasma and urine) of clindamycin from BenzaClin
Topical Gel was studied in male and female patients (n=13) with acne vulgaris. BenzaClin
Topical Gel (~2 g) was applied topically to the face and back twice daily for four and a half
(4.5) days. Quantifiable (>LOQ=1 ng/mL) clindamycin plasma concentrations were obtained
in six of thirteen subjects (46.2%) on Day 1 and twelve of thirteen subjects (92.3%) on Day 5.
Peak plasma concentrations (Cmax) of clindamycin ranged from 1.47 ng/mL to 2.77 ng/mL on Day 1 and 1.43 ng/mL to 7.18 ng/mL on Day 5. The AUC(0-12 h) ranged from 2.74 ng.h/mL to
12.86 ng.h/mL on Day 1 and 11.4 ng.h/mL to 69.7 ng.h/mL on Day 5.
The amount of clindamycin excreted in the urine during the 12-hour dosing interval increased
from a mean (SD) of 5745 (3130) ng on Day 1 to 12069 (7660) ng on Day 5. The mean % (SD) of
the administered dose that was excreted in the urine ranged from 0.03% (0.02) to 0.08% (0.04).
A comparison of the single (Day 1) and multiple (Day 5) dose plasma and urinary
concentrations of clindamycin indicates that there is accumulation of clindamycin following
multiple dosing of BenzaClin Topical Gel. The degree of accumulation calculated from the
plasma and urinary excretion data was ~ 2-fold.
Microbiology:
The clindamycin and benzoyl peroxide components individually have been shown to have in vitro activity against Propionibacterium acnes, an organism which has been associated with acne vulgaris; however, the clinical significance of this activity against P. acnes was not examined in clinical trials with this product.
Clinical Studies
In two adequate and well-controlled clinical studies of 758 patients, 214 used BenzaClin, 210 used benzoyl peroxide, 168 used clindamycin, and 166 used vehicle. BenzaClin applied twice daily for 10 weeks was significantly more effective than vehicle in the treatment of moderate to moderately severe facial acne vulgaris. Patients were evaluated and acne lesions counted at each clinical visit; Weeks 2, 4, 6, 8 and 10. The primary efficacy measures were the lesion counts and the investigator's global assessment evaluated at Week 10. Patients were instructed to wash the face with a mild soap, using only the hands. Fifteen minutes after the face was thoroughly dry, application was made to the entire face. Nonmedicated make-up could be applied at one hour after the BenzaClin application. If a moisturizer was required, the patients were provided a moisturizer to be used as needed. Patients were instructed to avoid sun exposure. Percent reductions in lesion counts after treatment for 10 weeks in these two studies are shown below:
| Study 1 | |||
|---|---|---|---|
|
BenzaClin |
Benzoyl peroxide |
Clindamycin |
Vehicle |
|
Mean percent reduction in inflammatory lesion counts |
|||
|
46% |
32% |
16% |
+ 3% |
|
Mean percent reduction in non-inflammatory lesion counts |
|||
|
22% |
22% |
9% |
+1% |
|
Mean percent reduction in total lesion counts |
|||
|
36% |
28% |
15% |
0.2% |
| Study 2 | |||
|---|---|---|---|
|
BenzaClin |
Benzoyl peroxide |
Clindamycin |
Vehicle |
|
Mean percent reduction in inflammatory lesion counts |
|||
|
63% |
53% |
45% |
42% |
|
Mean percent reduction in non-inflammatory lesion counts |
|||
|
54% |
50% |
39% |
36% |
|
Mean percent reduction in total lesion counts |
|||
|
58% |
52% |
42% |
39% |
The BenzaClin group showed greater overall improvement than the benzoyl peroxide, clindamycin and vehicle groups as rated by the investigator.
Indications and Usage for BenzaClin
BenzaClin Topical Gel is indicated for the topical treatment of acne vulgaris.
Contraindications
BenzaClin Topical Gel is contraindicated in those individuals who have shown hypersensitivity to any of its components or to lincomycin. It is also contraindicated in those having a history of regional enteritis, ulcerative colitis, or antibiotic-associated colitis.
Warnings
ORALLY AND PARENTERALLY ADMINISTERED CLINDAMYCIN HAS BEEN ASSOCIATED WITH SEVERE COLITIS WHICH MAY RESULT IN PATIENT DEATH. USE OF THE TOPICAL FORMULATION OF CLINDAMYCIN RESULTS IN ABSORPTION OF THE ANTIBIOTIC FROM THE SKIN SURFACE. DIARRHEA, BLOODY DIARRHEA, AND COLITIS (INCLUDING PSEUDOMEMBRANOUS COLITIS) HAVE BEEN REPORTED WITH THE USE OF TOPICAL AND SYSTEMIC CLINDAMYCIN. STUDIES INDICATE A TOXIN(S) PRODUCED BY CLOSTRIDIA IS ONE PRIMARY CAUSE OF ANTIBIOTIC-ASSOCIATED COLITIS. THE COLITIS IS USUALLY CHARACTERIZED BY SEVERE PERSISTENT DIARRHEA AND SEVERE ABDOMINAL CRAMPS AND MAY BE ASSOCIATED WITH THE PASSAGE OF BLOOD AND MUCUS. ENDOSCOPIC EXAMINATION MAY REVEAL PSEUDOMEMBRANOUS COLITIS. STOOL CULTURE FOR Clostridium Difficile AND STOOL ASSAY FOR C. difficile TOXIN MAY BE HELPFUL DIAGNOSTICALLY. WHEN SIGNIFICANT DIARRHEA OCCURS, THE DRUG SHOULD BE DISCONTINUED. LARGE BOWEL ENDOSCOPY SHOULD BE CONSIDERED TO ESTABLISH A DEFINITIVE DIAGNOSIS IN CASES OF SEVERE DIARRHEA. ANTIPERISTALTIC AGENTS SUCH AS OPIATES AND DIPHENOXYLATE WITH ATROPINE MAY PROLONG AND/OR WORSEN THE CONDITION. DIARRHEA, COLITIS, AND PSEUDOMEMBRANOUS COLITIS HAVE BEEN OBSERVED TO BEGIN UP TO SEVERAL WEEKS FOLLOWING CESSATION OF ORAL AND PARENTERAL THERAPY WITH CLINDAMYCIN.
Mild cases of pseudomembranous colitis usually respond to drug discontinuation alone. In moderate to severe cases, consideration should be given to management with fluids and electrolytes, protein supplementation and treatment with an antibacterial drug clinically effective against C. difficile colitis.
Precautions
General:
For dermatological use only; not for ophthalmic use. Concomitant topical acne therapy should be used with caution because a possible cumulative irritancy effect may occur, especially with the use of peeling, desquamating, or abrasive agents.
The use of antibiotic agents may be associated with the overgrowth of nonsusceptible organisms including fungi. If this occurs, discontinue use of this medication and take appropriate measures.
Avoid contact with eyes and mucous membranes.
Clindamycin and erythromycin containing products should not be used in combination. In vitro studies have shown antagonism between these two antimicrobials. The clinical significance of this in vitro antagonism is not known.
Information for Patients:
Patients using BenzaClin Topical Gel should receive the following information and instructions:
- 1.
- BenzaClin Topical Gel is to be used as directed by the physician. It is for external use only. Avoid contact with eyes, and inside the nose, mouth, and all mucous membranes, as this product may be irritating.
- 2.
- This medication should not be used for any disorder other than that for which it was prescribed.
- 3.
- Patients should not use any other topical acne preparation unless otherwise directed by physician.
- 4.
- Patients should minimize or avoid exposure to natural or artificial sunlight (tanning beds or UVA/B treatment) while using BenzaClin Topical Gel. To minimize exposure to sunlight, a wide-brimmed hat or other protective clothing should be worn, and a sunscreen with SPF 15 rating or higher should be used.
- 5.
- Patients who develop allergic symptoms such as severe swelling or shortness of breath should discontinue BenzaClin Topical Gel and contact their physician immediately. In addition, patients should report any signs of local adverse reactions to their physician.
- 6.
- BenzaClin Topical Gel may bleach hair or colored fabric.
- 7.
- BenzaClin Topical Gel can be stored at room temperature up to 25°C (77°F) for 3 months. Do not freeze. Discard any unused product after 3 months.
- 8.
- Before applying BenzaClin Topical Gel to affected areas, wash the skin gently, then rinse with warm water and pat dry.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
Benzoyl peroxide has been shown to be a tumor promoter and progression agent in a number of animal studies. The clinical significance of this is unknown.
Benzoyl peroxide in acetone at doses of 5 and 10 mg administered twice per week induced skin tumors in transgenic Tg.AC mice in a study using 20 weeks of topical treatment.
In a 52-week dermal photocarcinogenicity study in hairless mice, the median time to onset of skin tumor formation was decreased and the number of tumors per mouse increased following chronic concurrent topical administration of BenzaClin Topical Gel with exposure to ultraviolet radiation (40 weeks of treatment followed by 12 weeks of observation).
In a 2-year dermal carcinogenicity study in rats, treatment with BenzaClin Topical Gel at doses of 100, 500 and 2000 mg/kg/day caused a dose-dependent increase in the incidence of keratoacanthoma at the treated skin site of male rats. The incidence of keratoacanthoma at the treated site of males treated with 2000 mg/kg/day (8 times the highest recommended adult human dose of 2.5 g BenzaClin Topical Gel, based on mg/m2) was statistically significantly higher than that in the sham and vehicle controls.
Genotoxicity studies were not conducted with BenzaClin Topical Gel. Clindamycin phosphate was not genotoxic in Salmonella typhimurium or in a rat micronucleus test. Clindamycin phosphate sulfoxide, an oxidative degradation product of clindamycin phosphate and benzoyl peroxide, was not clastogenic in a mouse micronucleus test. Benzoyl peroxide has been found to cause DNA strand breaks in a variety of mammalian cell types, to be mutagenic in S. typhimurium tests by some but not all investigators, and to cause sister chromatid exchanges in Chinese hamster ovary cells. Studies have not been performed with BenzaClin Topical Gel or benzoyl peroxide to evaluate the effect on fertility. Fertility studies in rats treated orally with up to 300 mg/kg/day of clindamycin (approximately 120 times the amount of clindamycin in the highest recommended adult human dose of 2.5 g BenzaClin Topical Gel, based on mg/m2) revealed no effects on fertility or mating ability.
Pregnancy:
Teratogenic Effects:
Animal reproductive/developmental toxicity studies have not been conducted with BenzaClin Topical Gel or benzoyl peroxide. Developmental toxicity studies performed in rats and mice using oral doses of clindamycin up to 600 mg/kg/day (240 and 120 times amount of clindamycin in the highest recommended adult human dose based on mg/m2, respectively) or subcutaneous doses of clindamycin up to 250 mg/kg/day (100 and 50 times the amount of clindamycin in the highest recommended adult human dose based on mg/m2, respectively) revealed no evidence of teratogenicity.
There are no well-controlled trials in pregnant women treated with BenzaClin Topical Gel. It also is not known whether BenzaClin Topical Gel can cause fetal harm when administered to a pregnant woman.
Nursing Women:
It is not known whether BenzaClin Topical Gel is excreted in human milk after topical application. However, orally and parenterally administered clindamycin has been reported to appear in breast milk. Because of the potential for serious adverse reactions in nursing infants, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Adverse Reactions/Side Effects
During clinical trials, the most frequently reported adverse event in the BenzaClin treatment group was dry skin (12%). The table below lists local adverse events reported by at least 1% of patients in the BenzaClin and vehicle groups.
| Local Adverse Events - all causalities
in >/= 1% of patients |
||
|---|---|---|
|
BenzaClin |
Vehicle |
|
|
Application site reaction |
13 (3%) |
1 (<1%) |
|
Dry skin |
50 (12%) |
10 (6%) |
|
Pruritus |
8 (2%) |
1 (<1%) |
|
Peeling |
9 (2%) |
- |
|
Erythema |
6 (1%) |
1 (<1%) |
|
Sunburn |
5 (1%) |
- |
The actual incidence of dry skin might have been greater were it not for the use of a moisturizer in these studies.
Anaphylaxis, as well as allergic reactions leading to hospitalization, have been reported
during postmarketing use of clindamycin/benzoyl peroxide products. Because these reactions
are reported voluntarily from a population of uncertain size, it is not always possible to
reliably estimate their frequency or establish a causal relationship to drug exposure.
To report SUSPECTED ADVERSE REACTIONS, contact Valeant Pharmaceuticals North America LLC, at 1-800-321-4576 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
BenzaClin Dosage and Administration
BenzaClin Topical Gel should be applied twice daily, morning and evening, or as directed by a physician, to affected areas after the skin is gently washed, rinsed with warm water and patted dry.
How is BenzaClin supplied
| Size (Net Weight) | NDC 0187- | Benzoyl Peroxide Gel | Active Clindamycin Powder (in plastic vial) | Purified Water To Be Added to Each Vial |
|---|---|---|---|---|
|
25 grams |
5190-25 |
19.7 g |
0.3 g |
5 mL |
|
35 grams |
5190-35 |
27.6 g |
0.4 g |
7 mL |
|
50 grams |
5190-50 |
39.4 g |
0.6 g |
10 mL |
Prior to dispensing, tap the vial until powder flows freely. Add indicated amount of purified water to the vial (to the mark) and immediately shake to completely dissolve clindamycin. If needed, add additional purified water to bring level up to the mark. Add the solution in the vial to the gel and stir until homogeneous in appearance (1 to 1½ minutes). For the 35 and 50 gram pumps only, reassemble jar with pump dispenser. BenzaClin Topical Gel (as reconstituted) can be stored at room temperature up to 25°C (77°F) for 3 months. Place a 3-month expiration date on the label immediately following mixing.
Store at room temperature up to 25°C (77°F) [See USP].
Do not freeze. Keep tightly closed. Keep out of the reach of children.
Rx only
Manufactured for:
Valeant Pharmaceuticals North America LLC
Bridgewater, NJ 08807 USA
By:
Valeant Pharmaceuticals International, Inc.
Laval, Quebec H7L 4A8, Canada
BenzaClin is a trademark of Valeant Pharmaceuticals International, Inc. or its affiliates.
©Valeant Pharmaceuticals North America LLC
Rev. 02/17
50080835Q
9569601
PRINCIPAL DISPLAY PANEL - 25 gram jar
NDC 0187-5190-25
Rx only
BenzaClin®
(Clindamycin and Benzoyl Peroxide) gel 1%/5%
FOR TOPICAL USE ONLY
Reconstitute with Clinda-Pak™
One 25 g Jar (after reconstitution)
VALEANT
9568102
| BENZACLIN
clindamycin phosphate and benzoyl peroxide gel |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Bausch Health US LLC (831922468) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Bausch Health Companies Inc. | 245141858 | MANUFACTURE(0187-5190) | |